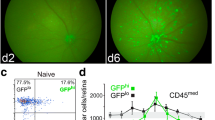
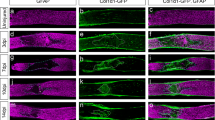
Abstract
Although glial scar formation has been extensively studied after optic nerve injury, the existence and characteristics of traumatic optic nerve fibrotic scar formation have not been previously characterized. Recent evidence suggests infiltrating macrophages are involved in pathological processes after optic nerve crush (ONC), but their role in fibrotic scar formation is unknown. Using wild-type and transgenic mouse models with optic nerve crush injury, we show that macrophages infiltrate and associate with fibroblasts in the traumatic optic nerve lesion fibrotic scar. We dissected the role of hematogenous and resident macrophages, labeled with Dil liposomes intravenously administered, and observed that hematogenous macrophages (Dil+ cells) specifically accumulate in the center of traumatic fibrotic scar while Iba-1+ cells reside predominantly at the margins of optic nerve fibrotic scar. Depletion of hematogenous macrophages results in reduced fibroblast density and decreased extracellular matrix deposition within the fibrotic scar area following ONC. However, retinal ganglion cell degeneration and function loss after optic nerve crush remain unaffected after hematogenous macrophage depletion. We present new and previously not characterized evidence that hematogenous macrophages are selectively recruited into the fibrotic core of the optic nerve crush site and critical for this fibrotic scar formation.






Similar content being viewed by others
Data Availability
Please contact the corresponding author for data requests.
Abbreviations
- CD3:
-
Cluster of differentiation 3
- CD68:
-
Cluster of differentiation 68
- CNS:
-
Central nervous system
- GCL:
-
Ganglion cell layer
- GFP:
-
Green fluorescent protein
- GFAP:
-
Glial fibrillary acidic protein
- Iba-1:
-
Ionized calcium binding adaptor molecule 1
- IL-6:
-
Interleukin-6
- IL-13:
-
Interleukin-13
- ONC:
-
Optic nerve crush
- PBS:
-
Phosphate buffered saline
- PDGF:
-
Platelet derived growth factor
- P-ERG:
-
Pattern electroretinogram
- PFA:
-
Paraformaldehyde
- RBPMS:
-
RNA-binding protein with multiple splicing
- RGC:
-
Retinal ganglion cells
- SCI:
-
Spinal cord injury
- TGF-β1:
-
Transforming growth factor-beta1
- Tmem119:
-
Transmembrane protein 119
- TON:
-
Traumatic optic neuropathy
References
Weber AJ, Harman CD, Viswanathan S (2008) Effects of optic nerve injury, glaucoma, and neuroprotection on the survival, structure, and function of ganglion cells in the mammalian retina. J Physiol 586(18):4393–4400. https://doi.org/10.1113/jphysiol.2008.156729
Kawano H, Kimura-Kuroda J, Komuta Y, Yoshioka N, Li HP, Kawamura K, Li Y, Raisman G (2012) Role of the lesion scar in the response to damage and repair of the central nervous system. Cell Tissue Res 349(1):169–180. https://doi.org/10.1007/s00441-012-1336-5
Monnier PP, D’Onofrio PM, Magharious M, Hollander AC, Tassew N, Szydlowska K, Tymianski M, Koeberle PD (2011) Involvement of caspase-6 and caspase-8 in neuronal apoptosis and the regenerative failure of injured retinal ganglion cells. J Neurosci 31(29):10494–10505. https://doi.org/10.1523/JNEUROSCI.0148-11.2011
Soderblom C, Luo X, Blumenthal E, Bray E, Lyapichev K, Ramos J, Krishnan V et al (2013) Perivascular fibroblasts form the fibrotic scar after contusive spinal cord injury. J Neurosci 33(34):13882–13887. https://doi.org/10.1523/JNEUROSCI.2524-13.2013
Orr MB, Gensel JC (2018) Spinal cord injury scarring and inflammation: therapies targeting glial and inflammatory responses. Neurotherapeutics 15(3):541–553. https://doi.org/10.1007/s13311-018-0631-6
Liu X, Liu Y, Jin H, Khodeiry MM, Kong W, Wang N, Lee JK, Lee RK (2021) Reactive fibroblasts in response to optic nerve crush injury. Mol Neurobiol 58(4):1392–1403. https://doi.org/10.1007/s12035-020-02199-4
Pakshir P, Hinz B (2018) The big five in fibrosis: macrophages, myofibroblasts, matrix, mechanics, and miscommunication. Matrix Biol 68–69:81–93. https://doi.org/10.1016/j.matbio.2018.01.019
Duffield JS, Forbes SJ, Constandinou CM, Clay S, Partolina M, Vuthoori S, Wu S, Lang R, Iredale JP (2005) Selective depletion of macrophages reveals distinct, opposing roles during liver injury and repair. J Clin Invest 115(1):56–65. https://doi.org/10.1172/JCI22675
Christophi GP, Massa PT (2009) Central neuroinvasion and demyelination by inflammatory macrophages after peripheral virus infection is controlled by SHP-1. Viral Immunol 22(6):371–387. https://doi.org/10.1089/vim.2009.0052
Zhu Y, Soderblom C, Krishnan V, Ashbaugh J, Bethea JR, Lee JK (2015) Hematogenous macrophage depletion reduces the fibrotic scar and increases axonal growth after spinal cord injury. Neurobiol Dis 74:114–125. https://doi.org/10.1016/j.nbd.2014.10.024
Yazawa N, Hamaguchi Y, Poe JC, Tedder TF (2005) Immunotherapy using unconjugated CD19 monoclonal antibodies in animal models for B lymphocyte malignancies and autoimmune disease. Proc Natl Acad Sci USA 102(42):15178–15183. https://doi.org/10.1073/pnas.0505539102
Shoulders H, Garner KH, Singla DK (2019) Macrophage depletion by clodronate attenuates bone morphogenetic protein-7 induced M2 macrophage differentiation and improved systolic blood velocity in atherosclerosis. Transl Res 203:1–14. https://doi.org/10.1016/j.trsl.2018.07.006
Ritter MR, Banin E, Moreno SK, Aguilar E, Dorrell MI, Friedlander M (2006) Myeloid progenitors differentiate into microglia and promote vascular repair in a model of ischemic retinopathy. J Clin Invest 116(12):3266–3276. https://doi.org/10.1172/JCI29683
Kataoka K, Nishiguchi KM, Kaneko H, van Rooijen N, Kachi S, Terasaki H (2011) The roles of vitreal macrophages and circulating leukocytes in retinal neovascularization. Invest Ophthalmol Vis Sci 52(3):1431–1438. https://doi.org/10.1167/iovs.10-5798
Yata Y, Scanga A, Gillan A, Yang L, Reif S, Breindl M, Brenner DA, Rippe RA (2003) DNase I-hypersensitive sites enhance alpha1(I) collagen gene expression in hepatic stellate cells. Hepatology 37(2):267–276. https://doi.org/10.1053/jhep.2003.50067
Liu Y, Tapia ML, Yeh J, He RC, Pomerleu D, Lee RK (2020) Differential gamma-synuclein expression in acute and chronic retinal ganglion cell death in the retina and optic nerve. Mol Neurobiol 57(2):698–709. https://doi.org/10.1007/s12035-019-01735-1
Porciatti V (2007) The mouse pattern electroretinogram. Doc Ophthalmol 115(3):145–153. https://doi.org/10.1007/s10633-007-9059-8
Ahmed Z, Shaw G, Sharma VP, Yang C, McGowan E, Dickson DW (2007) Actin-binding proteins coronin-1a and IBA-1 are effective microglial markers for immunohistochemistry. J Histochem Cytochem 55(7):687–700. https://doi.org/10.1369/jhc.6A7156.2007
Fischer D, Leibinger M (2012) Promoting optic nerve regeneration. Prog Retin Eye Res 31(6):688–701. https://doi.org/10.1016/j.preteyeres.2012.06.005
Varol C, Mildner A, Jung S (2015) Macrophages: development and tissue specialization. Annu Rev Immunol 33:643–675. https://doi.org/10.1146/annurev-immunol-032414-112220
Mildner A, Mack M, Schmidt H, Brück W, Djukic M, Zabel MD, Hille A, Priller J, Prinz M (2009) CCR2+Ly-6Chi monocytes are crucial for the effector phase of autoimmunity in the central nervous system. Brain 132(Pt 9):2487–2500. https://doi.org/10.1093/brain/awp144
Hilla AM, Diekmann H, Fischer D (2017) Microglia are irrelevant for neuronal degeneration and axon regeneration after acute injury. J Neurosci 37(25):6113–6124. https://doi.org/10.1523/JNEUROSCI.0584-17.2017
Ellis-Behnke RG, Jonas RA, Jonas JB (2013) The microglial system in the eye and brain in response to stimuli in vivo. J Glaucoma 22(Suppl 5):S32-35. https://doi.org/10.1097/IJG.0b013e3182934aca
Bennett ML, Bennett FC, Liddelow SA, Ajami B, Zamanian JL, Fernhoff NB, Mulinyawe SB, Bohlen CJ et al (2016) New tools for studying microglia in the mouse and human CNS. Proc Natl Acad Sci U S A 113(12):E1738-1746. https://doi.org/10.1073/pnas.1525528113
Satoh T, Nakagawa K, Sugihara F, Kuwahara R, Ashihara M, Yamane F, Minowa Y, Fukushima K et al (2017) Identification of an atypical monocyte and committed progenitor involved in fibrosis. Nature 541(7635):96–101. https://doi.org/10.1038/nature20611
Porter KE, Turner NA (2009) Cardiac fibroblasts: at the heart of myocardial remodeling. Pharmacol Ther 123(2):255–278. https://doi.org/10.1016/j.pharmthera.2009.05.002
Mould KJ, Barthel L, Mohning MP, Thomas SM, McCubbrey AL, Danhorn T, Leach SM, Fingerlin TE et al (2017) Cell origin dictates programming of resident versus recruited macrophages during acute lung injury. Am J Respir Cell Mol Biol 57(3):294–306. https://doi.org/10.1165/rcmb.2017-0061OC
Haan N, Zhu B, Wang J, Wei X, Song B (2015) Crosstalk between macrophages and astrocytes affects proliferation, reactive phenotype and inflammatory response, suggesting a role during reactive gliosis following spinal cord injury. J Neuroinflammation 12:109. https://doi.org/10.1186/s12974-015-0327-3
Wang X, Cao K, Sun X, Chen Y, Duan Z, Sun L, Guo L, Bai P et al (2015) Macrophages in spinal cord injury: phenotypic and functional change from exposure to myelin debris. Glia 63(4):635–651. https://doi.org/10.1002/glia.22774
Sanchez S, Lemmens S, Baeten P, Sommer D, Dooley D, Hendrix S, Gou Fabregas M (2018) HDAC3 inhibition promotes alternative activation of macrophages but does not affect functional recovery after spinal cord injury. Exp Neurobiol 27(5):437–452. https://doi.org/10.5607/en.2018.27.5.437
Funding
This work was supported by the National Institutes of Health Center Core Grant (P30EY014801) and an unrestricted Research to Prevent Blindness Unrestricted Grant to the Bascom Palmer Eye Institute. R.K.L. is partially supported by the Walter G. Ross Foundation. This work was partly supported by the Gutierrez Family Research Fund and the Camiener Foundation Glaucoma Research Fund.
Author information
Authors and Affiliations
Contributions
RK conceived and designed the research project. HJ, YL, and XL performed the experimental protocols. HJ and YL did the data collection. HJ, MM, and XL analyzed the data. HJ, RK, and JK wrote and revised the paper. RK obtained the funding and supervised the study. All authors have read and approved the final manuscript for publication.
Corresponding author
Ethics declarations
Ethics Approval
All animals were treated in compliance with the guidelines from the Association for Research in Vision and Ophthalmology (ARVO) Statement for the Use of Animals in Ophthalmic and Vision Research. All animal procedures were performed in accordance with the ethical standards of the institution or practice of the University of Miami Institutional Animal Care and Use Committee and NIH guidelines. No human participant was involved in this study.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jin, H., Liu, Y., Liu, X. et al. Hematogenous Macrophages Contribute to Fibrotic Scar Formation After Optic Nerve Crush. Mol Neurobiol 59, 7393–7403 (2022). https://doi.org/10.1007/s12035-022-03052-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-022-03052-6