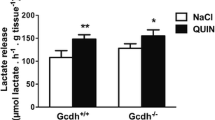
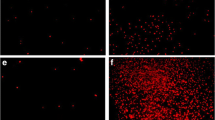
Abstract
Patients with glutaric aciduria type 1 (GA1), a neurometabolic disorder caused by deficiency of glutaryl-CoA dehydrogenase (GCDH) activity, commonly manifest acute encephalopathy associated with severe striatum degeneration and progressive cortical and striatal injury whose pathogenesis is still poorly known. We evaluated redox homeostasis, inflammatory response, mitochondrial biogenesis and dynamics, endoplasmic reticulum (ER)–mitochondria crosstalk, and ER stress in the brain of GCDH-deficient (Gcdh−/−) and wild-type (Gcdh+/+) mice fed a high Lys chow, which better mimics the human neuropathology mainly characterized by striatal lesions. Increased lipid peroxidation and altered antioxidant defenses, including decreased concentrations of reduced glutathione and increased activities of superoxide dismutase, catalase, and glutathione transferase, were observed in the striatum and cerebral cortex of Gcdh−/− mice. Augmented Iba-1 staining was also found in the dorsal striatum and neocortex, whereas the nuclear content of NF-κB was increased, and the cytosolic content of IκBα decreased in the striatum of the mutant animals, indicating a pro-inflammatory response. Noteworthy, in vivo treatment with the pan-PPAR agonist bezafibrate normalized these alterations. It was also observed that the ER–mitochondria crosstalk proteins VDAC1 and IP3R were reduced, whereas the ER stress protein DDIT3 was augmented in Gcdh−/− striatum, signaling disturbances of these processes. Finally, DRP1 content was elevated in the striatum of Gcdh−/− mice, indicating activated mitochondrial fission. We presume that some of these novel pathomechanisms may be involved in GA1 neuropathology and that bezafibrate should be tested as a potential adjuvant therapy for GA1.










Similar content being viewed by others
Data Availability
Data for this work is archived and publicly available upon request to the corresponding authors.
Abbreviations
- ANOVA:
-
Analysis of variance
- bez:
-
Bezafibrate
- CAT:
-
Catalase
- CSF:
-
Cerebrospinal fluid
- DDIT3:
-
DNA damage-inducible transcript 3
- DRP1:
-
Dynamin-1-like protein
- ER:
-
Endoplasmic reticulum
- GA1:
-
Glutaric acidemia 1
- GCDH:
-
Glutaryl-CoA dehydrogenase
- Grp75:
-
Glucose-regulated protein 75
- Grp78:
-
Glucose-regulated protein 78
- GSH:
-
Reduced glutathione
- GST:
-
Glutathione S-transferase
- Iba-1:
-
Ionized calcium-binding adapter molecule 1
- IκBα:
-
Nuclear factor of kappa light polypeptide gene enhancer in B-cells inhibitor
- IP3R:
-
Inositol trisphosphate receptor
- Lys:
-
Lysine
- MDA:
-
Malondialdehyde
- MFN1:
-
Mitofusin-1
- NF-κB:
-
Nuclear factor kappa B
- PBS:
-
Phosphate-buffered saline
- PGC-1α:
-
Peroxisome proliferator-activated receptor gamma coactivator 1-alpha
- ROS:
-
Reactive oxygen species
- SOD:
-
Superoxide dismutase
- VDAC1:
-
Voltage-dependent anion-selective channel 1
References
Goodman SI, Markey SP, Moe PG, Miles BS, Teng CC (1975) Glutaric aciduria; a “new” disorder of amino acid metabolism. Biochem Med 12:12–21. https://doi.org/10.1016/0006-2944(75)90091-5
Goodman SI, Kohlhoff JG (1975) Glutaric aciduria: inherited deficiency of glutaryl-CoA dehydrogenase activity. Biochem Med 13:138–140. https://doi.org/10.1016/0006-2944(75)90149-0
Larson A, Goodman S (2019) Glutaric acidemia type 1 summary genetic counseling. In: Adam MP, Ardinger HH, Pagon RA, Wallace SE, Bean LJH, Stephens K, Amemiya A (eds) GeneReviews®. University of Washington, Seattle, pp 1–27
Wajner M (2019) Neurological manifestations of organic acidurias. Nat Rev Neurol 15:253–271. https://doi.org/10.1038/s41582-019-0161-9
Goodman SI, Frerman FE (2014) Organic acidemias due to defects in lysine oxidation: 2-ketoadipic acidemia and glutaric acidemia. In: Valle D, Beaudet AL, Vogelstein B, Kinzler KW, Antonarakis SE, Ballabio A, Gibson K, Mitchell G (eds) The Online Metabolic and Molecular Bases of Inherited Disease. McGraw-Hill, New York, 2014. http://ommbid.mhmedical.com/content.aspx?bookid=971§ionid=62677296. Accessed 24 January 2022
Kölker S, Sauer SW, Okun JG, Hoffmann GF, Koeller DM (2006) Lysine intake and neurotoxicity in glutaric aciduria type I: towards a rationale for therapy? Brain 129:e54. https://doi.org/10.1093/brain/awl137
Jafari P, Braissant O, Bonafé L, Ballhausen D (2011) The unsolved puzzle of neuropathogenesis in glutaric aciduria type I. Mol Genet Metab 104:425–437. https://doi.org/10.1016/j.ymgme.2011.08.027
Koeller DM, Woontner M, Crnic LS, Kleinschmidt-DeMasters B, Stephens J, Hunt EL, Goodman SI (2002) Biochemical, pathologic and behavioral analysis of a mouse model of glutaric acidemia type I. Hum Mol Genet 11:347–357. https://doi.org/10.1093/hmg/11.4.347
Zinnanti WJ, Lazovic J, Wolpert EB, Antonetti DA, Smith MB, Connor JR, Woontner M, Goodman SI et al (2006) A diet-induced mouse model for glutaric aciduria type I. Brain 129:899–910. https://doi.org/10.1093/brain/awl009
Zinnanti WJ, Jacobs RE, Cheng KC, Zinnanti WJ, Lazovic J, Housman C, Lanoue K, Callaghan JPO et al (2007) Mechanism of age-dependent susceptibility and novel treatment strategy in glutaric acidemia type I. J Clin Invest 117:3258–3270. https://doi.org/10.1172/JCI31617.3258
Wajner M, Amaral AU, Leipnitz G, Seminotti B (2019) Pathogenesis of brain damage in glutaric acidemia type I: lessons from the genetic mice model. Int J Dev Neurosci 78:215–221. https://doi.org/10.1016/j.ijdevneu.2019.05.005
de Oliveira MF, Hagen ME, Pederzolli CD, Sgaravatti AM, Durigon K, Testa CG, Wannmacher CM, de Souza Wyse AT et al (2003) Glutaric acid induces oxidative stress in brain of young rats. Brain Res 964:153–158. https://doi.org/10.1016/S0006-8993(02)04118-5
Ferreira GC, Tonin A, Schuck PF, Viegas CM, Ceolato PC, Latini A, Perry ML, Wyse AT et al (2007) Evidence for a synergistic action of glutaric and 3-hydroxyglutaric acids disturbing rat brain energy metabolism. Int J Dev Neurosci 25(6):391–398. https://doi.org/10.1016/j.ijdevneu.2007.05.009
Latini A, Ferreira GC, Scussiato K, Schuck PF, Solano AF, Dutra-Filho CS, Vargas CR, Wajner M (2007) Induction of oxidative stress by chronic and acute glutaric acid administration to rats. Cell Mol Neurobiol 27:423–438. https://doi.org/10.1007/s10571-006-9134-9
Wajner M, Kölker S, Souza DO, Hoffmann GF, de Mello CF (2004) Modulation of glutamatergic and GABAergic neurotransmission in glutaryl-CoA dehydrogenase deficiency. J Inherit Metab Dis 27(6):825–828. https://doi.org/10.1023/B:BOLI.0000045765.37043.fb
Amaral AU, Seminotti B, Cecatto C, Fernandes CG, Busanello EN, Zanatta A, Kist LW, Bogo MR et al (2012) Reduction of Na+, K+-ATPase activity and expression in cerebral cortex of glutaryl-CoA dehydrogenase deficient mice: a possible mechanism for brain injury in glutaric aciduria type I. Mol Genet Metab 107:375–382. https://doi.org/10.1016/j.ymgme.2012.08.016
Amaral AU, Cecatto C, Seminotti B, Zanatta A, Fernandes CG, Busanello EN, Braga LM, Ribeiro CA et al (2012) Marked reduction of Na(+), K(+)-ATPase and creatine kinase activities induced by acute lysine administration in glutaryl-CoA dehydrogenase deficient mice. Mol Genet Metab 107:81–86. https://doi.org/10.1016/j.ymgme.2012.04.015
Amaral AU, Cecatto C, Seminotti B, Ribeiro CA, Lagranha VL, Pereira CC, de Oliveira FH, de Souza DG et al (2015) Experimental evidence that bioenergetics disruption is not mainly involved in the brain injury of glutaryl-CoA dehydrogenase deficient mice submitted to lysine overload. Brain Res 1620:116–129. https://doi.org/10.1016/j.brainres.2015.05.013
Busanello EN, Fernandes CG, Martell RV, Lobato VG, Goodman S, Woontner M, de Souza DO, Wajner M (2014) Disturbance of the glutamatergic system by glutaric acid in striatum and cerebral cortex of glutaryl-CoA dehydrogenase-deficient knockout mice: possible implications for the neuropathology of glutaric acidemia type I. J Neurol Sci 346:260–267. https://doi.org/10.1016/j.jns.2014.09.003
Rodrigues MD, Seminotti B, Amaral AU, Leipnitz G, Goodman SI, Woontner M, de Souza DO, Wajner M (2015) Experimental evidence that overexpression of NR2B glutamate receptor subunit is associated with brain vacuolation in adult glutaryl-CoA dehydrogenase deficient mice: a potential role for glutamatergic-induced excitotoxicity in GA I neuropathology. J Neurol Sci 359:133–140. https://doi.org/10.1016/j.jns.2015.10.043
Seminotti B, da Rosa MS, Fernandes CG, Amaral AU, Braga LM, Leipnitz G, de Souza DO, Woontner M et al (2012) Induction of oxidative stress in brain of glutaryl- CoA dehydrogenase deficient mice by acute lysine administration. Mol Genet Metab 106:31–38. https://doi.org/10.1016/j.ymgme.2012.03.002
Seminotti B, da Rosa MS, Fernandes CG, Leipnitz G, Olivera-Bravo S, Barbeito L, Ribeiro CA, de Souza DO et al (2013) Disruption of brain redox momeostasis in glutaryl-CoA dehydrogenase deficient mice treated with high dietary lysine supplementation. Mol Genet Metab 108:30–39. https://doi.org/10.1016/j.ymgme.2012.11.001
Seminotti B, Ribeiro RT, Amaral AU, da Rosa MS, Pereira CC, Leipnitz G, Koeller DM, Goodman S et al (2014) Acute lysine overload provokes protein oxidative damage and reduction of antioxidant defenses in the brain of infant glutaryl-CoA dehydrogenase deficient mice: a role for oxidative stress in GA I neuropathology. J Neurol Sci 344:105–113. https://doi.org/10.1016/j.jns.2014.06.034
Corona JC, Duchen MR (2015) PPARγ and PGC-1α as therapeutic targets in Parkinson’s. Neurochem Res 40:308–316. https://doi.org/10.1007/s11064-014-1377-0
Desvergne B, Wahli W (1999) Peroxisome proliferator-activated receptors: nuclear control of metabolism. Endocr Rev 20(5):649–688. https://doi.org/10.1210/edrv.20.5.0380
Manickam R, Wahli W (2017) Roles of peroxisome proliferator-activated receptor β/δ in skeletal muscle physiology. Biochimie 136:42–48. https://doi.org/10.1016/j.biochi.2016.11.010
Seminotti B, Amaral AU, Ribeiro RT, Rodrigues MD, Colin-Gonzalez AL, Leipnitz G, Santamaria A, Wajner M (2016) Oxidative stress, disrupted energy metabolism, and altered signaling pathways in glutaryl-CoA dehydrogenase knockout mice: potential implications of quinolinic acid toxicity in the neuropathology of glutaric acidemia type I. Mol Neurobiol 53:6459–6475. https://doi.org/10.1007/s12035-015-9548-9
Grings M, Moura AP, Parmeggiani B, Pletsch JT, Cardoso GMF, August PM, Matté C, Wyse ATS et al (2017) Bezafibrate prevents mitochondrial dysfunction, antioxidant system disturbance, glial reactivity and neuronal damage induced by sulfite administration in striatum of rats: Implications for a possible therapeutic strategy for sulfite oxidase deficiency. Biochim Biophys Acta Mol Basis Dis 1863(9):2135–2148. https://doi.org/10.1016/j.bbadis.2017.05.019
Evelson P, Travacio M, Repetto M, Escobar J, Llesuy S, Lissi EA (2001) Evaluation of total reactive antioxidant potential (TRAP) of tissue homogenates and their cytosols. Arch Biochem Biophys 388:261–266. https://doi.org/10.1006/abbi.2001.2292
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Yagi K (1998) Simple procedure for specific assay of lipid hydroperoxides in serum or plasma. Methods Mol Biol 108:107–110. https://doi.org/10.1385/0-89603-472-0:107
Browne RW, Armstrong D (1998) Reduced glutathione and glutathione disulfide. Methods Mol Biol 108:347–352. https://doi.org/10.1385/0-89603-472-0:347
Marklund SL (1985) Pyrogallol autoxidation. Handbook for Oxygen Radical Research. CRC Press, Boca Raton, pp 243–247
Aebi H (1984) Catalase in vitro. Methods Enzymol 105:121–126. https://doi.org/10.1016/s0076-6879(84)05016-3
Guthenberg C, Mannervik B (1981) Glutathione S-transferase (transferase pi) from human placenta is identical or closely related to glutathione S-transferase (transferase rho) from erythrocytes. Biochim Biophys Acta 661:255–260. https://doi.org/10.1016/0005-2744(81)90012-7
Seminotti B, Zanatta Â, Ribeiro RT, da Rosa MS, Wyse ATS, Leipnitz G, Wajner M (2019) Disruption of brain redox homeostasis, microglia activation and neuronal damage induced by intracerebroventricular administration of S-adenosylmethionine to developing rats. Mol Neurobiol 56:2760–2773. https://doi.org/10.1007/s12035-018-1275-6
Olivera-Bravo S, Seminotti B, Isasi E, Ribeiro CA, Leipnitz G, Woontner M, Goodman SI, Souza D et al (2019) Long lasting high lysine diet aggravates white matter injury in glutaryl-CoA dehydrogenase deficient (Gcdh-/-) mice. Mol Neurobiol 56(1):648–657. https://doi.org/10.1007/s12035-018-1077-x
Ribeiro RT, Seminotti B, Zanatta Â, de Oliveira FH, Amaral AU, Leipnitz G, Wajner M (2021) Neuronal death, glial reactivity, microglia activation, oxidative stress and bioenergetics impairment caused by intracerebroventricular administration of D-2-hydroxyglutaric acid to neonatal rats. Neuroscience 471:115–132. https://doi.org/10.1016/j.neuroscience.2021.07.024
Amaral AU, Seminotti B, da Silva JC, de Oliveira FH, Ribeiro RT, Leipnitz G, Souza DO, Wajner M (2019) Acute lysine overload provokes marked striatum injury involving oxidative stress signaling pathways in glutaryl-CoA dehydrogenase deficient mice. Neurochem Int 129:104467. https://doi.org/10.1016/j.neuint.2019.104467
Seminotti B, Amaral AU, Grings M, Ribeiro CAJ, Leipnitz G, Wajner M (2020) Lipopolysaccharide-elicited systemic inflammation induces selective vulnerability of cerebral cortex and striatum of developing glutaryl-CoA dehydrogenase deficient (Gcdh-/-) mice to oxidative stress. Neurotox Res 38(4):1024–1036. https://doi.org/10.1007/s12640-020-00291-0
Soffer D, Amir N, Elpeleg ON, Gomori JM, Shalev RS, Gottschalk-Sabag S (1992) Striatal degeneration and spongy myelinopathy in glutaric acidemia. J Neurol Sci 107:199–204. https://doi.org/10.1016/0022-510X(92)90289-W
Latini A, Borba Rosa R, Scussiato K, Llesuy S, Belló-Klein A, Wajner M (2002) 3-Hydroxyglutaric acid induces oxidative stress and decreases the antioxidant defenses in cerebral cortex of young rats. Brain Res 956:367–373. https://doi.org/10.1016/S0006-8993(02)03573-4
Latini A, Scussiato K, Leipnitz G, Dutra-Filho CS, Wajner M (2005) Promotion of oxidative stress by 3-hydroxyglutaric acid in rat stria- tum. J Inherit Metab Dis 28:57–67. https://doi.org/10.1007/s10545-005-3677-7
Seminotti B, Grings M, Tucci P, Leipnitz G, Saso L (2021) Nuclear factor erythroid-2-related factor 2 signaling in the neuropathophysiology of inherited metabolic disorders. Front Cell Neurosci 38:1024–1036. https://doi.org/10.3389/fncel.2021.785057
Guerreiro G, Faverzani J, Jacques CED, Marchetti DP, Sitta A, de Moura CD, Kayser A, Kok F et al (2018) Oxidative damage in glutaric aciduria type I patients and the protective effects of l-carnitine treatment. J Cell Biochem 119(12):10021–10032. https://doi.org/10.1002/jcb.27332
Guerreiro G, Amaral AU, Ribeiro RT, Faverzani J, Groehs AC, Sitta A, Deon M, Wajner M et al (2019) L-Carnitine prevents oxidative stress in striatum of glutaryl-CoA dehydrogenase deficient mice submitted to lysine overload. Biochim Biophys Acta Mol Basis Dis 1865(9):2420–2427. https://doi.org/10.1016/j.bbadis.2019.06.007
Guerreiro G, Faverzani J, Moura AP, Volfart V, Gome Dos Reis B, Sitta A, Gonzalez EA, de Lima RG et al (2021) Protective effects of L-carnitine on behavioral alterations and neuroinflammation in striatum of glutaryl-COA dehydrogenase deficient mice. Arch Biochem Biophys 709:108970. https://doi.org/10.1016/j.abb.2021.108970
Halliwell B (2006) Oxidative stress and neurodegeneration: where are we now? J Neurochem 97(6):1634–1658. https://doi.org/10.1111/j.1471-4159.2006.03907.x
Halliwell B, Gutteridge JMC (2015) Free radicals in biology and medicine, 5th edn. Oxford University Press. https://doi.org/10.1093/acprof:oso/9780198717478.001.0001
Ionescu-Tucker A, Cotman CW (2021) Emerging roles of oxidative stress in brain aging and Alzheimer’s disease. Neurobiol Aging 25(107):86–95. https://doi.org/10.1016/j.neurobiolaging.2021.07.014
Leipnitz G, Vargas CR, Wajner M (2015) Disturbance of redox homeostasis as a contributing underlying pathomechanism of brain and liver alterations in 3-hydroxy-3-methylglutaryl-CoA lyase deficiency. J Inherit Metab Dis 38:1021–1028. https://doi.org/10.1007/s10545-015-9863-3
Mc Guire PJ, Parikh A, Diaz GA (2009) Profiling of oxidative stress in patients with inborn errors of metabolism. Mol Genet Metab 98:173–80. https://doi.org/10.1016/j.ymgme.2009.06.007
Parmeggiani B, Vargas CR (2018) Oxidative stress in urea cycle disorders: findings from clinical and basic research. Clin Chim Acta 477:121–126. https://doi.org/10.1016/j.cca.2017.11.041
Ribas GS, Vargas CR, Wajner M (2014) L-carnitine supplementation as a potential antioxidant therapy for inherited neurometabolic disorders. Gene 533:469–476. https://doi.org/10.1016/j.gene.2013.10.017
Wajner M, Latini A, Wyse AT, Dutra-Filho CS (2004) The role of oxidative damage in the neuropathology of organic acidurias: insights from animal studies. J Inherit Metab Dis 27:427–448. https://doi.org/10.1023/B:BOLI.0000037353.13085.e2
Wajner M, Vargas CR, Amaral AU (2020) Disruption of mitochondrial functions and oxidative stress contribute to neurologic dysfunction in organic acidurias. Arch Biochem Biophys 696:108646. https://doi.org/10.1016/j.abb.2020.108646
Sivandzade F, Prasad S, Bhalerao A, Cucullo L (2019) NRF2 and NF-қB interplay in cerebrovascular and neurodegenerative disorders: molecular mechanisms and possible therapeutic approaches. Redox Biol 21:101059. https://doi.org/10.1016/j.redox.2018.11.017
Rodrigues FS, de Zorzi VN, Funghetto MP, Haupental F, Cardoso AS, Marchesan S, Cardoso AM, Schinger MRC et al (2019) Involvement of the cholinergic parameters and glial cells in learning delay induced by glutaric acid: protection by N-acetylcysteine. Mol Neurobiol 56:4945–4959. https://doi.org/10.1007/s12035-018-1395-z
Rodrigues FS, França AP, Broetto N, Furian AF, Oliveira MS, Santos ARS, Royes LFF, Fighera MR (2020) Sustained glial reactivity induced by glutaric acid may be the trigger to learning delay in early and late phases of development: involvement of p75NTR receptor and protection by N-acetylcysteine. Brain Res 1749:147145. https://doi.org/10.1016/j.brainres.2020.147145
Olivera S, Fernandez A, Latini A, Rosillo JC, Casanova G, Wajner M, Cassina P, Barbeito L (2008) Astrocytic proliferation and mitochondrial dysfunction induced by accumulated glutaric acidemia I (GAI) metabolites: possible implications for GAI pathogenesis. Neurobiol Dis 32:528–534. https://doi.org/10.1016/j.nbd.2008.09.011
Gonzalez Melo M, Remacle N, Cudré-Cung HP, Roux C, Poms M, Cudalbu C, Barroso M, Gersting SW et al (2021) The first knock-in rat model for glutaric aciduria type I allows further insights into pathophysiology in brain and periphery. Mol Genet Metab 133:157–181. https://doi.org/10.1016/j.ymgme.2021.03.017
Guerreiro G, Diaz Jaques CE, Wajner M, Vargas CR (2020) Elevated levels of BDNF and cathepsin-d as possible peripheral markers of neurodegeneration in plasma of patients with glutaric acidemia type I. Int J Dev Neurosci 80(1):42–49. https://doi.org/10.1002/jdn.10006
Arantes-Rodrigues R, Henriques A, Pinto-Leite R, Faustino-Rocha A, Pinho-Oliveira J, Teixeira-Guedes C, Seixas F, Gama A et al (2012) The effects of repeated oral gavage on the health of male CD-1 mice. Lab Anim (NY) 41:129–134. https://doi.org/10.1038/laban0512-129
Bonnefont JP, Bastin J, Laforêt P, Aubey F, Mogenet A, Romano S, Ricquier D, Gobin-Limballe S et al (2010) Long-term follow-up of bezafibrate treatment in patients with the myopathic form of carnitine palmitoyltransferase 2 deficiency. Clin Pharmacol Ther 88:101–108. https://doi.org/10.1038/clpt.2010.55
Djouadi F, Aubey F, Schlemmer D, Ruiter JP, Wanders RJ, Strauss AW, Bastin J (2005) Bezafibrate increases very-long-chain acyl-CoA dehydrogenase protein and mRNA expression in deficient fibroblasts and is a potential therapy for fatty acid oxidation disorders. Hum Mol Genet 14:2695–2703. https://doi.org/10.1093/hmg/ddi303
Djouadi F, Habarou F, Le Bachelier C, Ferdinandusse S, Schlemmer D, Benoist JF, Boutron A, Andresen BS et al (2016) Mitochondrial trifunctional protein deficiency in human cultured fibroblasts: effects of bezafibrate. J Inherit Metab Dis 39:47–58. https://doi.org/10.1007/s10545-015-9871-3
Gobin-Limballe S, Djouadi F, Aubey F, Olpin S, Andresen BS, Yamaguchi S, Mandel H, Fukao T et al (2007) Genetic basis for correction of very-long-chain acyl-coenzyme A dehydrogenase deficiency by bezafibrate in patient fibroblasts: toward a genotype-based therapy. Am J Hum Genet 81:1133–1143. https://doi.org/10.1086/522375
Gobin-Limballe S, McAndrew RP, Djouadi F, Kim JJ, Bastin J (2010) Compared effects of missense mutations in Very-Long-Chain Acyl-CoA Dehydrogenase deficiency: combined analysis by structural, functional and pharmacological approaches. Biochim Biophys Acta 1802:478–484. https://doi.org/10.1016/j.bbadis.2010.01.001
da Rosa-Junior NT, Parmeggiani B, da Rosa MS, Glänzel NM, de Moura AL, Wajner M, Leipnitz G (2019) Bezafibrate in vivo administration prevents 3-methylglutaric acid-induced impairment of redox status, mitochondrial biogenesis, and neural injury in brain of developing rats. Neurotox Res 35:809–822. https://doi.org/10.1007/s12640-019-00019-9
da Rosa-Junior NT, Parmeggiani B, Glänzel NM, de Moura AL, Frusciante MR, Dutra Filho CS, Wajner M, Leipnitz G (2020) In vivo evidence that bezafibrate prevents oxidative stress and mitochondrial dysfunction caused by 3-methylglutaric acid in rat liver. Biochimie 171–172:187–196. https://doi.org/10.1016/j.biochi.2020.03.007
Parmeggiani B, Grings M, da Rosa-Junior NT, Britto R, Wajner M, Leipnitz G (2019) Bezafibrate prevents glycine-induced increase of antioxidant enzyme activities in rat striatum. Mol Neurobiol 56:29–38. https://doi.org/10.1007/s12035-018-1074-0
Johri A, Calingasan NY, Hennessey TM, Sharma A, Yang L, Wille E, Chandra A, Beal MF (2012) Pharmacologic activation of mitochondrial biogenesis exerts widespread beneficial effects in a transgenic mouse model of Huntington’s disease. Hum Mol Genet 21(5):1124–1137. https://doi.org/10.1093/hmg/ddr541
Suárez-Rivero JM, Villanueva-Paz M, de la Cruz-Ojeda P, de la Mata M, Cotán D, Oropesa-Ávila M, de Lavera I, Álvarez-Córdoba M et al (2016) Mitochondrial dynamics in mitochondrial diseases. Diseases 5(1):1. https://doi.org/10.3390/diseases5010001
van der Bliek AM, Shen Q, Kawajiri S (2013) Mechanisms of mitochondrial fission and fusion. Cold Spring Harb Perspect Biol 5:a011072. https://doi.org/10.1101/cshperspect.a011072
Westrate LM, Drocco JA, Martin KR, Hlavacek WS, MacKeigan JP (2014) Mitochondrial morphological features are associated with fission and fusion events. PLoS ONE 9:e95265. https://doi.org/10.1371/journal.pone.0095265
Dandekar A, Mendez R, Zhang K (2015) Cross talk between ER stress, oxidative stress, and inflammation in health and disease. Methods Mol Biol 1292:205–214. https://doi.org/10.1007/978-1-4939-2522-3_15
Filadi R, Theurey P, Pizzo P (2017) The endoplasmic reticulum-mitochondria coupling in health and disease: Molecules, functions and significance. Cell Calcium 62:1–15. https://doi.org/10.1016/j.ceca.2017.01.003
Lim JH, Lee HJ, Ho Jung M, Song J (2009) Coupling mitochondrial dysfunction to endoplasmic reticulum stress response: a molecular mechanism leading to hepatic insulin resistance. Cell Signal 21:169–177. https://doi.org/10.1016/j.cellsig.2008.10.004
Nakka VP, Prakash-Babu P, Vemuganti R (2016) Crosstalk between endoplasmic reticulum stress, oxidative stress, and autophagy: potential therapeutic targets for acute CNS injuries. Mol Neurobiol 53:532–544. https://doi.org/10.1007/s12035-014-9029-6
Veeresh P, Kaur H, Sarmah D, Mounica L, Verma G, Kotian V, Kesharwani R, Kalia K et al (2019) Endoplasmic reticulum-mitochondria crosstalk: from junction to function across neurological disorders. Ann N Y Acad Sci 1457:41–60. https://doi.org/10.1111/nyas.14212
Flippo KH, Strack S (2017) Mitochondrial dynamics in neuronal injury, development and plasticity. J Cell Sci 130:671–681. https://doi.org/10.1242/jcs.171017
Guillery O, Malka F, Frachon P, Milea D, Rojo M, Lombès A (2008) Modulation of mitochondrial morphology by bioenergetics defects in primary human fibroblasts. Neuromuscul Disord 18:319–330. https://doi.org/10.1016/j.nmd.2007.12.008
Yu T, Wang L, Yoon Y (2015) Morphological control of mitochondrial bioenergetics. Front Biosci (Landmark Ed) 20:229–246. https://doi.org/10.2741/4306
Csordás G, Weaver D, Hajnóczky G (2018) Endoplasmic reticulum-mitochondrial contactology: structure and signaling functions. Trends Cell Biol 28:523–540. https://doi.org/10.1016/j.tcb.2018.02.009
Hu H, Tian M, Ding C, Yu S (2019) The C/EBP homologous protein (CHOP) transcription factor functions in endoplasmic reticulum stress-induced apoptosis and microbial infection. Front Immunol 9:3083. https://doi.org/10.3389/fimmu.2018.03083
Tian F, Fu X, Gao J, Ying Y, Hou L, Liang Y, Ning Q, Luo X (2014) Glutaric acid-mediated apoptosis in primary striatal neurons. Biomed Res Int 2014:484731. https://doi.org/10.1155/2014/484731
Acknowledgements
We thank the Centro de Microscopia e Microanálise (UFRGS, RS, Brazil) for the support with the immunofluorescence analysis.
Funding
This work was supported by grants from Conselho Nacional de Desenvolvimento Científico e Tecnológico [CNPq, #425914/2016–0, MW and #427051/2018–5, BS], Fundação de Amparo à Pesquisa do Estado do Rio Grande do Sul [FAPERGS / PRONEX II, #16/0465–0, MW], Fundo de Incentivo à Pesquisa e Eventos/Hospital de Clínicas de Porto Alegre [FIPE/HCPA, #180538, MW], Financiadora de Estudos e Projetos/Rede Instituto Brasileiro de Neurociência [FINEP, #01.06.0842–00, MW], Instituto Nacional de Ciência e Tecnologia em Excitotoxicidade e Neuroproteção [INCT-EN, #465671/2014–4, MW], and a Post-Doctoral Researcher fellowship under the National Post-Doctoral Program (Programa Nacional de Pós Doutorado/PNPD, BS) from Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES).
Author information
Authors and Affiliations
Contributions
B. S. was responsible for study conceptualization, investigation, methodology, validation, visualization, data curation, formal analysis, and writing. M. B., R. T. R., G. L., and M. W. were responsible for investigation and data curation. M. W. was responsible for study conceptualization, investigation, data curation, funding acquisition, resources, and writing.
Corresponding author
Ethics declarations
Ethics Approval
All experimental procedures with animals were approved by the Ethical Committee for the Care and Use of Laboratory Animals of the Hospital de Clínicas de Porto Alegre (number 2018–0538), and all efforts were made to minimize animal suffering and stress and to reduce the number of animals necessary to produce consistent scientific data. The experiments were designed and performed in accordance with the “Guide for the Care and Use of Laboratory Animals” (National Institutes of Health, publication no. 80–23, revised 2011), the Directive 2010/63/EU, and the International Guiding Principles for Biomedical Research Involving Animals.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Seminotti, B., Brondani, M., Ribeiro, R.T. et al. Disturbance of Mitochondrial Dynamics, Endoplasmic Reticulum–Mitochondria Crosstalk, Redox Homeostasis, and Inflammatory Response in the Brain of Glutaryl-CoA Dehydrogenase-Deficient Mice: Neuroprotective Effects of Bezafibrate. Mol Neurobiol 59, 4839–4853 (2022). https://doi.org/10.1007/s12035-022-02887-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-022-02887-3