Abstract
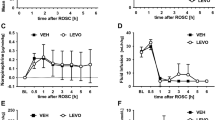
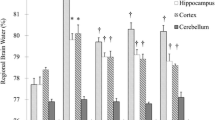
Brain injury is the most common cause of death for patients resuscitated from cardiac arrest. Magnesium is an attractive neuroprotective compound which protects neurons from ischemic injury by reducing neuronal calcium overload via NMDA receptor modulation and preventing calcium-induced mitochondrial permeability transition. Intramuscular (IM) delivery of MgSO4 during CPR has the potential to target these mechanisms within an early therapeutic window. We hypothesize that IM MgSO4 administrated during CPR could achieve therapeutic serum magnesium levels within 15 min after ROSC and improve neurologic outcomes in a rat model of asphyxial cardiac arrest. Male Long Evans rats were subjected to 8-min asphyxial cardiac arrest and block randomized to receive placebo, 107 mg/kg, 215 mg/kg, or 430 mg/kg MgSO4 IM at the onset of CPR. Serum magnesium concentrations increased rapidly with IM delivery during CPR, achieving twofold to fourfold increase by 15 min after ROSC in all magnesium dose groups. Rats subjected to cardiac arrest or sham surgery were block randomized to treatment groups for assessment of neurological outcomes. We found that IM MgSO4 during CPR had no effect on ROSC rate (p > 0.05). IM MgSO4 treatment had no statistically significant effect on 10-day survival with good neurologic function or hippocampal CA1 pyramidal neuron survival compared to placebo treatment. In conclusion, a single dose IM MgSO4 during CPR achieves up to fourfold baseline serum magnesium levels within 15 min after ROSC; however, this treatment strategy did not improve survival, recovery of neurologic function, or neuron survival. Future studies with repeated dosing or in combination with hypothermic targeted temperature management may be indicated.



Similar content being viewed by others
Data Availability
The data and materials used in the current study are available from the corresponding author on reasonable request.
References
Virani SS, Alonso A, Aparicio HJ, Benjamin EJ, Bittencourt MS, Callaway CW, Carson AP, Chamberlain AM et al (2021) Heart Disease and Stroke Statistics-2021 Update: a report from the American Heart Association. Circulation: CIR0000000000000950. https://doi.org/10.1161/CIR.0000000000000950
CARES. Cardiac Arrest Registry to Enhance Survival (CARES) 2019
Benjamin EJ, Virani SS, Callaway CW, Chamberlain AM, Chang AR, Cheng S, Chiuve SE, Cushman M et al (2018) Heart Disease and Stroke Statistics-2018 Update: a report From the American Heart Association. Circulation 137(12):e67–e492. https://doi.org/10.1161/CIR.0000000000000558
Laver S, Farrow C, Turner D, Nolan J (2004) Mode of death after admission to an intensive care unit following cardiac arrest. Intensive Care Med 30(11):2126–8. https://doi.org/10.1007/s00134-004-2425-z
Girotra S, van Diepen S, Nallamothu BK, Carrel M, Vellano K, Anderson ML, McNally B, Abella BS et al (2016) Regional variation in out-of-hospital cardiac arrest survival in the United States. Circulation 133(22):2159–68. https://doi.org/10.1161/CIRCULATIONAHA.115.018175
Elmer J, Rittenberger JC, Coppler PJ, Guyette FX, Doshi AA, Callaway CW, Pittsburgh Post-Cardiac Arrest S (2016) Long-term survival benefit from treatment at a specialty center after cardiac arrest. Resuscitation 108:48–53. https://doi.org/10.1016/j.resuscitation.2016.09.008
Reynolds IJ (1998) Intracellular calcium and magnesium: critical determinants of excitotoxicity? Prog Brain Res 116:225–243
Golshani-Hebroni S (2016) Mg(++) requirement for MtHK binding, and Mg(++) stabilization of mitochondrial membranes via activation of MtHK & MtCK and promotion of mitochondrial permeability transition pore closure: a hypothesis on mechanisms underlying Mg(++)’s antioxidant and cytoprotective effects. Gene 581(1):1–13. https://doi.org/10.1016/j.gene.2015.12.046
Szanda G, Rajki A, Gallego-Sandin S, Garcia-Sancho J, Spat A (2009) Effect of cytosolic Mg2+ on mitochondrial Ca2+ signaling. Pflugers Arch 457(4):941–54. https://doi.org/10.1007/s00424-008-0551-0
Blomeyer CA, Bazil JN, Stowe DF, Dash RK, Camara AK (2016) Mg(2+) differentially regulates two modes of mitochondrial Ca(2+) uptake in isolated cardiac mitochondria: implications for mitochondrial Ca(2+) sequestration. J Bioenerg Biomembr 48(3):175–88. https://doi.org/10.1007/s10863-016-9644-1
Favaron M, Bernardi P (1985) Tissue-specific modulation of the mitochondrial calcium uniporter by magnesium ions. FEBS Lett 183(2):260–4. https://doi.org/10.1016/0014-5793(85)80789-4
Silver IA, Erecinska M (1990) Intracellular and extracellular changes of [Ca2+] in hypoxia and ischemia in rat brain in vivo. J Gen Physiol 95(5):837–66. https://doi.org/10.1085/jgp.95.5.837
Erecinska M, Silver IA (1992) Relationship between ions and energy metabolism: cerebral calcium movements during ischaemia and subsequent recovery. Can J Physiol Pharmacol 70(Suppl):S190-3. https://doi.org/10.1139/y92-262
Silver IA, Erecinska M (1992) Ion homeostasis in rat brain in vivo: intra- and extracellular [Ca2+] and [H+] in the hippocampus during recovery from short-term, transient ischemia. J Cereb Blood Flow Metab 12(5):759–72. https://doi.org/10.1038/jcbfm.1992.107
Dux E, Mies G, Hossmann KA, Siklos L (1987) Calcium in the mitochondria following brief ischemia of gerbil brain. Neurosci Lett 78(3):295–300
Izumi Y, Roussel S, Pinard E, Seylaz J (1991) Reduction of infarct volume by magnesium after middle cerebral artery occlusion in rats. J Cereb Blood Flow Metab 11(6):1025–30. https://doi.org/10.1038/jcbfm.1991.170
Marinov MB, Harbaugh KS, Hoopes PJ, Pikus HJ, Harbaugh RE (1996) Neuroprotective effects of preischemia intraarterial magnesium sulfate in reversible focal cerebral ischemia. J Neurosurg 85(1):117–24. https://doi.org/10.3171/jns.1996.85.1.0117
Schmid-Elsaesser R, Zausinger S, Hungerhuber E, Baethmann A, Reulen HJ (1999) Neuroprotective effects of combination therapy with tirilazad and magnesium in rats subjected to reversible focal cerebral ischemia. Neurosurgery 44(1):163–71. https://doi.org/10.1097/00006123-199901000-00100 (discussion 71-2)
Longstreth WT Jr, Fahrenbruch CE, Olsufka M, Walsh TR, Copass MK, Cobb LA (2002) Randomized clinical trial of magnesium, diazepam, or both after out-of-hospital cardiac arrest. Neurology 59(4):506–514
Sibai BM, Graham JM, McCubbin JH (1984) A comparison of intravenous and intramuscular magnesium sulfate regimens in preeclampsia. Am J Obstet Gynecol 150(6):728–733
Saha PK, Kaur J, Goel P, Kataria S, Tandon R, Saha L (2017) Safety and efficacy of low dose intramuscular magnesium sulphate (MgSO4) compared to intravenous regimen for treatment of eclampsia. J Obstet Gynaecol Res 43(10):1543–1549. https://doi.org/10.1111/jog.13424
Manorot M, Tongsong T, Khettglang T (1996) A comparison of serum magnesium sulfate levels in pregnant women with severe preeclampsia between intravenous and intramuscular magnesium sulfate regimens: a randomized controlled trial. J Med Assoc Thai = Chotmaihet thangphaet 79(2):76–82
Perkins GD, Ji C, Deakin CD, Quinn T, Nolan JP, Scomparin C, Regan S, Long J et al (2018) A randomized trial of epinephrine in out-of-hospital cardiac arrest. N Engl J Med 379(8):711–721. https://doi.org/10.1056/NEJMoa1806842
Mody P, Brown SP, Kudenchuk PJ, Chan PS, Khera R, Ayers C, Pandey A, Kern KB et al (2018) Intraosseous versus intravenous access in patients with out-of-hospital cardiac arrest: Insights from the resuscitation outcomes consortium continuous chest compression trial. Resuscitation 134:69–75. https://doi.org/10.1016/j.resuscitation.2018.10.031
Pritchard JA (1980) Management of preeclampsia and eclampsia. Kidney Int 18(2):259–66. https://doi.org/10.1038/ki.1980.134
Pritchard JA (1955) The use of the magnesium ion in the management of eclamptogenic toxemias. Surg Gynecol Obstet 100(2):131–140
Muir KW, Lees KR (1998) Dose optimization of intravenous magnesium sulfate after acute stroke. Stroke 29(5):918–923
Oh JS, Tulasi J, Xiaodan R, Stacey WC, Neumar RW (2017) Valproic acid combined with postcardiac arrest hypothermic-targeted temperature management prevents delayed seizures and improves survival in a rat cardiac arrest model. Crit Care Med 45(11):e1149–e56. https://doi.org/10.1097/CCM.0000000000002690
Katz L, Ebmeyer U, Safar P, Radovsky A, Neumar R (1995) Outcome model of asphyxial cardiac arrest in rats. J Cereb Blood Flow Metab 15(6):1032–9. https://doi.org/10.1038/jcbfm.1995.129
Che D, Li L, Kopil CM, Liu Z, Guo W, Neumar RW (2011) Impact of therapeutic hypothermia onset and duration on survival, neurologic function, and neurodegeneration after cardiac arrest. Crit Care Med 39(6):1423–30. https://doi.org/10.1097/CCM.0b013e318212020a
Huang Q, Riviere JE (2014) The application of allometric scaling principles to predict pharmacokinetic parameters across species. Expert Opin Drug Metab Toxicol 10(9):1241–53. https://doi.org/10.1517/17425255.2014.934671
Sanderson TH, Wider JM (2013) 2-vessel occlusion/hypotension: a rat model of global brain ischemia. J Vis Exp JoVE (76). https://doi.org/10.3791/50173
Sanderson TH, Wider JM, Lee I, Reynolds CA, Liu J, Lepore B, Tousignant R, Bukowski MJ et al (2018) Inhibitory modulation of cytochrome c oxidase activity with specific near-infrared light wavelengths attenuates brain ischemia/reperfusion injury. Sci Rep 8(1):3481. https://doi.org/10.1038/s41598-018-21869-x
Lagina AT 3rd, Calo L, Deogracias M, Sanderson T, Kumar R, Wider J, Sullivan JM (2013) Combination therapy with insulin-like growth factor-1 and hypothermia synergistically improves outcome after transient global brain ischemia in the rat. Acad Emerg Med Off J Soc Acad Emerg Med 20(4):344–351. https://doi.org/10.1111/acem.12104
Chouchani ET, Pell VR, Gaude E, Aksentijevic D, Sundier SY, Robb EL, Logan A, Nadtochiy SM et al (2014) Ischaemic accumulation of succinate controls reperfusion injury through mitochondrial ROS. Nature 515(7527):431–435. https://doi.org/10.1038/nature13909
Benveniste H, Drejer J, Schousboe A, Diemer NH (1984) Elevation of the extracellular concentrations of glutamate and aspartate in rat hippocampus during transient cerebral ischemia monitored by intracerebral microdialysis. J Neurochem 43(5):1369–74. https://doi.org/10.1111/j.1471-4159.1984.tb05396.x
Hagberg H, Lehmann A, Sandberg M, Nystrom B, Jacobson I, Hamberger A (1985) Ischemia-induced shift of inhibitory and excitatory amino acids from intra- to extracellular compartments. J Cereb Blood Flow Metab 5(3):413–9. https://doi.org/10.1038/jcbfm.1985.56
White RE, Hartzell HC (1988) Effects of intracellular free magnesium on calcium current in isolated cardiac myocytes. Science 239(4841 Pt 1):778–80. https://doi.org/10.1126/science.2448878
Agus ZS, Morad M (1991) Modulation of cardiac ion channels by magnesium. Annu Rev Physiol 53:299–307. https://doi.org/10.1146/annurev.ph.53.030191.001503
Nowak L, Bregestovski P, Ascher P, Herbet A, Prochiantz A (1984) Magnesium gates glutamate-activated channels in mouse central neurones. Nature 307(5950):462–5. https://doi.org/10.1038/307462a0
Johnson JW, Ascher P (1990) Voltage-dependent block by intracellular Mg2+ of N-methyl-D-aspartate-activated channels. Biophys J 57(5):1085–90. https://doi.org/10.1016/S0006-3495(90)82626-6
Cotton DB, Hallak M, Janusz C, Irtenkauf SM, Berman RF (1993) Central anticonvulsant effects of magnesium sulfate on N-methyl-D-aspartate-induced seizures. Am J Obstet Gynecol 168(3 Pt 1):974–8. https://doi.org/10.1016/s0002-9378(12)90855-8
Smith DA, Connick JH, Stone TW (1989) Effect of changing extracellular levels of magnesium on spontaneous activity and glutamate release in the mouse neocortical slice. Br J Pharmacol 97(2):475–82. https://doi.org/10.1111/j.1476-5381.1989.tb11975.x
Muir KW (2002) Magnesium in stroke treatment. Postgrad Med J 78(925):641–5. https://doi.org/10.1136/pmj.78.925.641
Neumar RW (2000) Molecular mechanisms of ischemic neuronal injury. Ann Emerg Med 36(5):483–506. https://doi.org/10.1067/mem.2000.110995
Miles AN, Majda BT, Meloni BP, Knuckey NW (2001) Postischemic intravenous administration of magnesium sulfate inhibits hippocampal CA1 neuronal death after transient global ischemia in rats. Neurosurgery 49(6):1443–50. https://doi.org/10.1097/00006123-200112000-00025 (discussion 50-1)
Sameshima H, Ikenoue T (2001) Long-term magnesium sulfate treatment as protection against hypoxic-ischemic brain injury in seven-day-old rats. Am J Obstet Gynecol 184(2):185–90. https://doi.org/10.1067/mob.2001.108343
Zhu H, Meloni BP, Moore SR, Majda BT, Knuckey NW (2004) Intravenous administration of magnesium is only neuroprotective following transient global ischemia when present with post-ischemic mild hypothermia. Brain Res 1014(1–2):53–60. https://doi.org/10.1016/j.brainres.2004.03.073
Zhu H, Meloni BP, Bojarski C, Knuckey MW, Knuckey NW (2005) Post-ischemic modest hypothermia (35 degrees C) combined with intravenous magnesium is more effective at reducing CA1 neuronal death than either treatment used alone following global cerebral ischemia in rats. Exp Neurol 193(2):361–8. https://doi.org/10.1016/j.expneurol.2005.01.022
Kilgannon JH, Roberts BW, Jones AE, Mittal N, Cohen E, Mitchell J, Chansky ME, Trzeciak S (2014) Arterial blood pressure and neurologic outcome after resuscitation from cardiac arrest*. Crit Care Med 42(9):2083–91. https://doi.org/10.1097/CCM.0000000000000406
Xu K, Puchowicz MA, LaManna JC (2018) Post-resuscitation arterial blood pressure on survival and change of capillary density following cardiac arrest and resuscitation in rats. Adv Exp Med Biol 1072:77–82. https://doi.org/10.1007/978-3-319-91287-5_13
Young MN, Hollenbeck RD, Pollock JS, Giuseffi JL, Wang L, Harrell FE, McPherson JA (2015) Higher achieved mean arterial pressure during therapeutic hypothermia is not associated with neurologically intact survival following cardiac arrest. Resuscitation 88:158–64. https://doi.org/10.1016/j.resuscitation.2014.12.008
Muir KW (2001) Magnesium for neuroprotection in ischaemic stroke: rationale for use and evidence of effectiveness. CNS Drugs 15(12):921–30. https://doi.org/10.2165/00023210-200115120-00002
Saver JL, Starkman S, Eckstein M, Stratton SJ, Pratt FD, Hamilton S, Conwit R, Liebeskind DS et al (2015) Prehospital use of magnesium sulfate as neuroprotection in acute stroke. N Engl J Med 372(6):528–36. https://doi.org/10.1056/NEJMoa1408827
Funding
This work was supported by the American Heart Association, Strategically Focused Research Network grant (19SFRN34830008) (RWN, THS). This work has also been supported in part by NIH R01 NS091242 and NS120322 (THS), NIH T32HL007853 (TB), and the China Scholarship Council (201806220219) (RZ).
Author information
Authors and Affiliations
Contributions
Robert W. Neumar and Thomas H. Sanderson designed the research. Rui Zhang, Timothy D. Bryson, Garrett M. Fogo, Jinhui Liao, Sarita Raghunayakula, Jennifer Mathieu, Joseph M. Wider, Xiaodan Ren, Kathleen J. Maheras, Katlynn J. Emaus, and Erin Gruley performed the research. Rui Zhang, Timothy D. Bryson, Garrett M. Fogo, Robert W. Neumar, Thomas H. Sanderson, and Yuguo Chen analyzed and interpreted data. Rui Zhang, Timothy D. Bryson, Thomas H. Sanderson, and Robert W. Neumar drafted and revised the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics Approval
This study was approved by the University of Michigan Institutional Animal Care and Use Committee.
Consent to Participate
Not applicable.
Consent for Publication
All authors approved the final manuscript and submission.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Thomas H. Sanderson and Robert W. Neumar are co-senior authors.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhang, R., Bryson, T.D., Fogo, G.M. et al. Rapid Treatment with Intramuscular Magnesium Sulfate During Cardiopulmonary Resuscitation Does Not Provide Neuroprotection Following Cardiac Arrest. Mol Neurobiol 59, 1872–1881 (2022). https://doi.org/10.1007/s12035-021-02645-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-021-02645-x