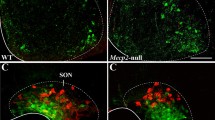
Abstract
Mating behavior in males and females is dependent on olfactory cues processed through both the main olfactory epithelium (MOE) and the vomeronasal organ (VNO). Signaling through the MOE is critical for the initiation of male mating behavior, and the loss of MOE signaling severely compromises this comportment. Here, we demonstrate that dosage of the homeodomain gene Six3 affects the degree of development of MOE but not the VNO. Anomalous MOE development in Six3 heterozygote mice leads to hyposmia, specifically disrupting male mounting behavior by impairing detection of volatile female estrus pheromones. Six3 is highly expressed in the MOE, main olfactory bulb (MOB), and hypothalamus; all regions essential in the proper migration of the gonadotropin-releasing hormone (GnRH) neurons, a key reproductive neuronal population that migrates along olfactory axons from the developing nose into the brain. Interestingly, we find that the reduction in Six3 expression in Six3 heterozygote mice compromises development of the MOE and MOB, resulting in mis-migration of GnRH neurons due to improper olfactory axon targeting. This reduction in the hypothalamic GnRH neuron population, by 45% in adulthood, leads to female subfertility, but does not impact male hormone levels, suggesting that male infertility is not related to GnRH neuron numbers, but exclusively linked to abnormal olfaction. We here determine that Six3 is haploinsufficient for MOE development, GnRH neuron migration, and fertility, and represents a novel candidate gene for Kallmann syndrome, a form of inherited infertility.








Similar content being viewed by others
References
Stowers L, Logan DW (2010) Olfactory mechanisms of stereotyped behavior: on the scent of specialized circuits. Curr Opin Neurobiol 20(3):274–280. https://doi.org/10.1016/j.conb.2010.02.013
Brown RE (1979) Mammalian social odors: a critical review. In: Rosenblat JS, Hinde RA, Beer C, Busnell M-C (eds) Advances in the study of behavior, vol 10. Academic Press, New York, pp. 107–161
Brennan PA, Zufall F (2006) Pheromonal communication in vertebrates. Nature 444(7117):308–315. https://doi.org/10.1038/nature05404
Thompson ML, Edwards DA (1972) Olfactory bulb ablation and hormonally induced mating in spayed female mice. Physiol Behav 8(6):1141–1146
Brennan PA, Keverne EB (1997) Neural mechanisms of mammalian olfactory learning. Prog Neurobiol 51(4):457–481
Dulac C, Torello AT (2003) Molecular detection of pheromone signals in mammals: from genes to behaviour. Nat Rev Neurosci 4(7):551–562. https://doi.org/10.1038/nrn1140
Pierman S, Douhard Q, Balthazart J, Baum MJ, Bakker J (2006) Attraction thresholds and sex discrimination of urinary odorants in male and female aromatase knockout (ArKO) mice. Horm Behav 49(1):96–104. https://doi.org/10.1016/j.yhbeh.2005.05.007
Ma D, Allen ND, Van Bergen YC, Jones CM, Baum MJ, Keverne EB, Brennan PA (2002) Selective ablation of olfactory receptor neurons without functional impairment of vomeronasal receptor neurons in OMP-ntr transgenic mice. Eur J Neurosci 16(12):2317–2323
Biellmann F, Henion TR, Burki K, Hennet T (2008) Impaired sexual behavior in male mice deficient for the beta1-3 N-acetylglucosaminyltransferase-I gene. Mol Reprod Dev 75(5):699–706. https://doi.org/10.1002/mrd.20828
Yoon H, Enquist LW, Dulac C (2005) Olfactory inputs to hypothalamic neurons controlling reproduction and fertility. Cell 123(4):669–682
Matsumoto S, Yamazaki C, Masumoto KH, Nagano M, Naito M, Soga T, Hiyama H, Matsumoto M et al (2006) Abnormal development of the olfactory bulb and reproductive system in mice lacking prokineticin receptor PKR2. Proc Natl Acad Sci U S A 103(11):4140–4145. https://doi.org/10.1073/pnas.0508881103
Stowers L, Marton TF (2005) What is a pheromone? Mammalian pheromones reconsidered. Neuron 46(5):699–702
Lloyd-Thomas A, Keverne EB (1982) Role of the brain and accessory olfactory system in the block to pregnancy in mice. Neuroscience 7(4):907–913
Rattazzi L, Cariboni A, Poojara R, Shoenfeld Y, D'Acquisto F (2015) Impaired sense of smell and altered olfactory system in RAG-1(--) immunodeficient mice. Front Neurosci 9:318. https://doi.org/10.3389/fnins.2015.00318
Keverne EB (2004) Importance of olfactory and vomeronasal systems for male sexual function. Physiol Behav 83(2):177–187. https://doi.org/10.1016/j.physbeh.2004.08.013
Wray S (2010) From nose to brain: development of gonadotrophin-releasing hormone-1 neurones. J Neuroendocrinol 22(7):743–753. https://doi.org/10.1111/j.1365-2826.2010.02034.x
Forni PE, Wray S (2015) GnRH, anosmia and hypogonadotropic hypogonadism—where are we? Front Neuroendocrinol 36:165–177. https://doi.org/10.1016/j.yfrne.2014.09.004
Wray S (2002) Development of gonadotropin-releasing hormone-1 neurons. Front Neuroendocrinol 23(3):292–316
Schwanzel-Fukuda M, Pfaff DW (1989) Origin of luteinizing hormone-releasing hormone neurons. Nature 338:161–164
Schwanzel-Fukuda M (1999) Origin and migration of luteinizing hormone-releasing hormone neurons in mammals. Microsc Res Tech 44(1):2–10. https://doi.org/10.1002/(SICI)1097-0029(19990101)44:1<2::AID-JEMT2>3.0.CO;2-4
Wray S (2001) Development of luteinizing hormone releasing hormone neurones. J Neuroendocrinol 13(1):3–11
Tobet SA, Schwarting GA (2006) Minireview: recent progress in gonadotropin-releasing hormone neuronal migration. Endocrinology 147(3):1159–1165
Bless E, Raitcheva D, Henion TR, Tobet S, Schwarting GA (2006) Lactosamine modulates the rate of migration of GnRH neurons during mouse development. Eur J Neurosci 24(3):654–660. https://doi.org/10.1111/j.1460-9568.2006.04955.x
Cadman SM, Kim SH, Hu Y, Gonzalez-Martinez D, Bouloux PM (2007) Molecular pathogenesis of Kallmann’s syndrome. Horm Res 67(5):231–242
Balasubramanian R, Dwyer A, Seminara SB, Pitteloud N, Kaiser UB, Crowley WF Jr (2010) Human GnRH deficiency: a unique disease model to unravel the ontogeny of GnRH neurons. Neuroendocrinology 92(2):81–99. https://doi.org/10.1159/000314193
Pitteloud N, Quinton R, Pearce S, Raivio T, Acierno J, Dwyer A, Plummer L, Hughes V et al (2007) Digenic mutations account for variable phenotypes in idiopathic hypogonadotropic hypogonadism. J Clin Invest 117(2):457–463
Stamou MI, Cox KH, Crowley WF (2015) Discovering genes essential to the hypothalamic regulation of human reproduction using a human disease model: adjusting to life in the “-Omics” Era. Endocr Rev:er20151045. https://doi.org/10.1210/er.2015-1045
Chan YM, de Guillebon A, Lang-Muritano M, Plummer L, Cerrato F, Tsiaras S, Gaspert A, Lavoie HB et al (2009) GNRH1 mutations in patients with idiopathic hypogonadotropic hypogonadism. Proc Natl Acad Sci U S A 106(28):11703–11708
Diaczok D, DiVall S, Matsuo I, Wondisford FE, Wolfe AM, Radovick S (2011) Deletion of Otx2 in GnRH neurons results in a mouse model of hypogonadotropic hypogonadism. Mol Endocrinol 25(5):833–846. https://doi.org/10.1210/me.2010-0271
Hoffmann HM, Trang C, Gong P, Kimura I, Pandolfi EC, Mellon PL (2016) Deletion of Vax1 from GnRH neurons abolishes GnRH expression and leads to hypogonadism and infertility. J Neurosci 36(12):3506–3518. https://doi.org/10.1523/JNEUROSCI.2723-15.2016
Hoffmann HM, Tamrazian A, Xie H, Perez-Millan MI, Kauffman AS, Mellon PL (2014) Heterozygous deletion of ventral anterior homeobox (Vax1) causes subfertility in mice. Endocrinology 155(6):4043–4053. https://doi.org/10.1210/en.2014-1277
Larder R, Kimura I, Meadows J, Clark DD, Mayo S, Mellon PL (2013) Gene dosage of Otx2 is important for fertility in male mice. Mol Cell Endocrinol 377(1–2):16–22. https://doi.org/10.1016/j.mce.2013.06.026
Lagutin OV, Zhu CC, Kobayashi D, Topczewski J, Shimamura K, Puelles L, Russell HR, McKinnon PJ et al (2003) Six3 repression of Wnt signaling in the anterior neuroectoderm is essential for vertebrate forebrain development. Genes Dev 17(3):368–379
Oliver G, Mailhos A, Wehr R, Copeland NG, Jenkins NA, Gruss P (1995) Six3, a murine homologue of the sine oculis gene, demarcates the most anterior border of the developing neural plate and is expressed during eye development. Development 121(12):4045–4055
Anderson AM, Weasner BM, Weasner BP, Kumar JP (2012) Dual transcriptional activities of SIX proteins define their roles in normal and ectopic eye development. Development 139(5):991–1000. https://doi.org/10.1242/dev.077255
Dubourg C, Lazaro L, Pasquier L, Bendavid C, Blayau M, Le Duff F, Durou MR, Odent S et al (2004) Molecular screening of SHH, ZIC2, SIX3, and TGIF genes in patients with features of holoprosencephaly spectrum: Mutation review and genotype-phenotype correlations. Hum Mutat 24(1):43–51. https://doi.org/10.1002/humu.20056
Pasquier L, Dubourg C, Blayau M, Lazaro L, Le Marec B, David V, Odent S (2000) A new mutation in the six-domain of SIX3 gene causes holoprosencephaly. Eur J Hum Genet 8(10):797–800. https://doi.org/10.1038/sj.ejhg.5200540
Pasquier L, Dubourg C, Gonzales M, Lazaro L, David V, Odent S, Encha-Razavi F (2005) First occurrence of aprosencephaly/atelencephaly and holoprosencephaly in a family with a SIX3 gene mutation and phenotype/genotype correlation in our series of SIX3 mutations. J Med Genet 42(1):e4. https://doi.org/10.1136/jmg.2004.023416
Lewandoski M, Wassarman KM, Martin GR (1997) Zp3-cre, a transgenic mouse line for the activation or inactivation of loxP-flanked target genes specifically in the female germ line. Curr Biol 7(2):148–151
Wolfe A, Divall S, Singh SP, Nikrodhanond AA, Baria AT, Le WW, Hoffman GE, Radovick S (2008) Temporal and spatial regulation of CRE recombinase expression in gonadotrophin-releasing hormone neurones in the mouse. J Neuroendocrinol 20(7):909–916
Clark DD, Gorman MR, Hatori M, Meadows JD, Panda S, Mellon PL (2013) Aberrant development of the suprachiasmatic nucleus and circadian rhythms in mice lacking the homeodomain protein six6. J Biol Rhythm 28(1):15–25. https://doi.org/10.1177/0748730412468084
Paxinos G, Franklin KBJ (2004) The mouse brain in stereotxic coordinates. Academic Press,
Srinivas S, Watanabe T, Lin CS, William CM, Tanabe Y, Jessell TM, Costantini F (2001) Cre reporter strains produced by targeted insertion of EYFP and ECFP into the ROSA26 locus. BMC Dev Biol 1:4
Yang M, Crawley JN (2009) Simple behavioral assessment of mouse olfaction. Current protocols in neuroscience/editorial board, Jacqueline N Crawley [et al] Chapter 8:Unit 8 24. https://doi.org/10.1002/0471142301.ns0824s48
Arakawa H, Blanchard DC, Arakawa K, Dunlap C, Blanchard RJ (2008) Scent marking behavior as an odorant communication in mice. Neurosci Biobehav Rev 32(7):1236–1248. https://doi.org/10.1016/j.neubiorev.2008.05.012
Keller M, Pierman S, Douhard Q, Baum MJ, Bakker J (2006) The vomeronasal organ is required for the expression of lordosis behaviour, but not sex discrimination in female mice. Eur J Neurosci 23(2):521–530. https://doi.org/10.1111/j.1460-9568.2005.04589.x
Pankevich DE, Baum MJ, Cherry JA (2004) Olfactory sex discrimination persists, whereas the preference for urinary odorants from estrous females disappears in male mice after vomeronasal organ removal. J Neurosci 24(42):9451–9457. https://doi.org/10.1523/JNEUROSCI.2376-04.2004
Stowers L, Holy TE, Meister M, Dulac C, Koentges G (2002) Loss of sex discrimination and male-male aggression in mice deficient for TRP2. Science 295(5559):1493–1500
Poling MC, Kauffman AS (2012) Sexually dimorphic testosterone secretion in prenatal and neonatal mice is independent of kisspeptin-Kiss1r and GnRH signaling. Endocrinology 153(2):782–793. https://doi.org/10.1210/en.2011-1838
Kimchi T, Xu J, Dulac C (2007) A functional circuit underlying male sexual behaviour in the female mouse brain. Nature 448(7157):1009–1014. https://doi.org/10.1038/nature06089
Matsuo T, Hattori T, Asaba A, Inoue N, Kanomata N, Kikusui T, Kobayakawa R, Kobayakawa K (2015) Genetic dissection of pheromone processing reveals main olfactory system-mediated social behaviors in mice. Proc Natl Acad Sci U S A 112(3):E311–E320. https://doi.org/10.1073/pnas.1416723112
Slotnick B, Restrepo D, Schellinck H, Archbold G, Price S, Lin W (2010) Accessory olfactory bulb function is modulated by input from the main olfactory epithelium. Eur J Neurosci 31(6):1108–1116. https://doi.org/10.1111/j.1460-9568.2010.07141.x
Suarez R, Garcia-Gonzalez D, de Castro F (2012) Mutual influences between the main olfactory and vomeronasal systems in development and evolution. Front Neuroanat 6:50. https://doi.org/10.3389/fnana.2012.00050
Der-Avakian A, Markou A (2012) The neurobiology of anhedonia and other reward-related deficits. Trends Neurosci 35(1):68–77. https://doi.org/10.1016/j.tins.2011.11.005
Boden MJ, Kennaway DJ (2006) Circadian rhythms and reproduction. Reproduction 132(3):379–392. https://doi.org/10.1530/rep.1.00614
Fueshko S, Wray S (1994) LHRH cells migrate on peripherin fibers in embryonic olfactory explant cultures: an in vitro model for neurophilic neuronal migration. Dev Biol 166(1):331–348
Mandiyan VS, Coats JK, Shah NM (2005) Deficits in sexual and aggressive behaviors in Cnga2 mutant mice. Nat Neurosci 8(12):1660–1662. https://doi.org/10.1038/nn1589
Baum MJ (2012) Contribution of pheromones processed by the main olfactory system to mate recognition in female mammals. Front Neuroanat 6:20. https://doi.org/10.3389/fnana.2012.00020
Keller M, Baum MJ, Bakker J (2008) Olfactory control of sex-recognition and sexual behavior in mice. In: Hurst J, Beynon RJ, Roberts SC, Wyatt T (eds) Chemical signals in vertebrates vol 11. Springer, New York, pp. 241–250
Herbison AE, Porteous R, Pape JR, Mora JM, Hurst PR (2008) Gonadotropin-releasing hormone (GnRH) neuron requirements for puberty, ovulation and fertility. Endocrinology 149(2):597–604
Pingault V, Bodereau V, Baral V, Marcos S, Watanabe Y, Chaoui A, Fouveaut C, Leroy C et al (2013) Loss-of-function mutations in SOX10 cause Kallmann syndrome with deafness. Am J Hum Genet 92(5):707–724. https://doi.org/10.1016/j.ajhg.2013.03.024
Givens ML, Rave-Harel N, Goonewardena VD, Kurotani R, Berdy SE, Swan CH, Rubenstein JL, Robert B et al (2005) Developmental regulation of gonadotropin-releasing hormone gene expression by the MSX and DLX homeodomain protein families. J Biol Chem 280:19156–19165
Ng KL, Li JD, Cheng MY, Leslie FM, Lee AG, Zhou QY (2005) Dependence of olfactory bulb neurogenesis on prokineticin 2 signaling. Science 308(5730):1923–1927. https://doi.org/10.1126/science.1112103
Dode C, Rondard P (2013) PROK2/PROKR2 signaling and Kallmann syndrome. Front Endocrinol 4:19. https://doi.org/10.3389/fendo.2013.00019
Layman WS, McEwen DP, Beyer LA, Lalani SR, Fernbach SD, Oh E, Swaroop A, Hegg CC et al (2009) Defects in neural stem cell proliferation and olfaction in Chd7 deficient mice indicate a mechanism for hyposmia in human CHARGE syndrome. Hum Mol Genet 18(11):1909–1923. https://doi.org/10.1093/hmg/ddp112
Walters KA, Allan CM, Jimenez M, Lim PR, Davey RA, Zajac JD, Illingworth P, Handelsman DJ (2007) Female mice haploinsufficient for an inactivated androgen receptor (AR) exhibit age-dependent defects that resemble the AR null phenotype of dysfunctional late follicle development, ovulation, and fertility. Endocrinology 148(8):3674–3684. https://doi.org/10.1210/en.2007-0248
Kim HG, Herrick SR, Lemyre E, Kishikawa S, Salisz JA, Seminara S, MacDonald ME, Bruns GA et al (2005) Hypogonadotropic hypogonadism and cleft lip and palate caused by a balanced translocation producing haploinsufficiency for FGFR1. J Med Genet 42(8):666–672. https://doi.org/10.1136/jmg.2004.026989
Edwards DA, Burge KG (1973) Olfactory control of the sexual behavior of male and female mice. Physiol Behav 11(6):867–872
Larder R, Clark DD, Miller NL, Mellon PL (2011) Hypothalamic dysregulation and infertility in mice lacking the homeodomain protein Six6. J Neurosci 31(2):426–438. https://doi.org/10.1523/JNEUROSCI.1688-10.2011
Zhu CC, Dyer MA, Uchikawa M, Kondoh H, Lagutin OV, Oliver G (2002) Six3-mediated auto repression and eye development requires its interaction with members of the Groucho-related family of co-repressors. Development 129(12):2835–2849
Acknowledgements
The authors thank Lauren D. Sitts and Jason D. Meadows for technical assistance on this project. This work was supported by National Institutes of Health grants R01 HD082567 and R01 HD072754 (to P.L.M.) and by National Institute of Child Health and Human Development/National Institutes of Health P50 HD012303 as part of the National Centers for Translational Research in Reproduction and Infertility (P.L.M.). P.L.M. was partially supported by P30 DK063491, P42 ES101337, and P30 CA023100. E.C.P. was partially supported by National Institutes of Health R25 GM083275 and National Institutes of Health F31 HD098652. H.M.H. was partially supported by K99 HD084759. E.L.S. was partially supported by T32 HD007203, The Lalor Foundation, P42 ES101337, and T32 DK007044.
Funding
This work was supported by NIH.
Author information
Authors and Affiliations
Contributions
E.C.P., H.M.H., E.L.S., M.R.G. and P.L.M. designed the experiments; E.C.P., H.M.H., and E.L.S performed the experiments; E.C.P., H.M.H., E.L.S., and P.L.M. analyzed the data; E.C.P., H.M.H., E.L.S., M.R.G., and P.L.M. discussed the results; and E.C.P., H.M.H., E.L.S., M.R.G., and P.L.M. wrote the manuscript.
Corresponding author
Ethics declarations
All animal procedures were performed in accordance with the UCSD Institutional Animal Care and Use Committee regulations.
Conflict of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Pandolfi, E.C., Hoffmann, H.M., Schoeller, E.L. et al. Haploinsufficiency of SIX3 Abolishes Male Reproductive Behavior Through Disrupted Olfactory Development, and Impairs Female Fertility Through Disrupted GnRH Neuron Migration. Mol Neurobiol 55, 8709–8727 (2018). https://doi.org/10.1007/s12035-018-1013-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-018-1013-0