Abstract
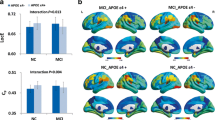
This study aims to explore the hippocampus-based functional connectivity patterns in young, healthy APP and/or presenilin-1/2 mutation carriers and APOE ε4 subjects. Seventy-eight healthy young adults (33 male, mean age 24.0 ± 2.2 years; 18 APP and/or presenilin1/2 mutation carriers [APP/presenilin-1/2 group], 30 APOE ε4 subjects [APOE ε4 group], and 30 subjects without the above-mentioned genes [control group]) underwent resting-state functional MR imaging and neuropsychological assessments. Bilateral hippocampus functional connectivity patterns were compared among three groups. The brain regions with statistical differences were then extracted, and correlation analyses were performed between Z values of the brain regions and neuropsychological results. Compared with control group, both APOE ε4 group and APP/presenilin-1/2 group showed increased functional connectivity in medial prefrontal cortex and precuneus for the seeds of bilateral hippocampi. The APOE ε4 group displayed increased functional connectivity from bilateral hippocampi to the left middle temporal gyrus compared with the control group. Moreover, compared with the APP/presenilin-1/2 group, the APOE ε4 group also had markedly increased functional connectivity in right hippocampus-left middle temporal gyrus. The Z values of right hippocampus-left middle temporal gyrus correlated with various neuropsychological results across all the subjects, as well as in APOE ε4 group. Young healthy adults carrying APOE ε4 and APP/presenilin-1/2 displayed different hippocampus functional connectivity patterns, which may underlie the discrepant mechanisms of gene-modulated cognitive dysfunction in Alzheimer’s disease.






Similar content being viewed by others
References
Kim DH, Yeo SH, Park J-M, Choi JY, Lee T-H, Park SY, Ock MS, Eo J et al (2014) Genetic markers for diagnosis and pathogenesis of Alzheimer’s disease. Gene 545:185–193
Caselli RJ, Reiman EM, Osborne D, Hentz JG, Baxter LC, Hernandez JL, Alexander GG (2004) Longitudinal changes in cognition and behavior in asymptomatic carriers of the APOE ɛ4 allele. Neurology 62:1990–1995
Sala-Llonch R, Fortea J, Bartrés-Faz D, Bosch B, Lladó A, Peña-Gómez C, Antonell A, Castellanos-Pinedo F (2013) Evolving brain functional abnormalities in psen1 mutation carriers: a resting and visual encoding FMRI study. J Alzheimers Dis 36:165–175
Kerchner GA, Berdnik D, Shen JC, Bernstein JD, Fenesy MC, Deutsch GK, Wyss-Coray T, Rutt BK (2014) APOE ɛ4 worsens hippocampal CA1 apical neuropil atrophy and episodic memory. Neurology 82:691–697
Godbolt a K, Beck J a, Collinge J, Garrard P, Warren JD, Fox NC, Rossor MN (2004) A presenilin 1 R278I mutation presenting with language impairment. Neurology 63:1702–1704
Guo Q, Wang Z, Li H, Wiese M, Zheng H (2012) APP physiological and pathophysiological functions: insights from animal models. Cell Res Nature Publishing Group 22:78–89
Zhu L, Zhong M, Elder GA, Sano M, Holtzman DM, Gandy S, Cardozo C, Haroutunian V et al (2015) Phospholipid dysregulation contributes to ApoE4-associated cognitive deficits in Alzheimer’s disease pathogenesis. Proc Natl Acad Sci U S A 112:11965–11970
Loy CT, Schofield PR, Turner AM, Kwok JBJ (2014) Genetics of dementia. Lancet 383:828–840
Jiang Y, Huang H, Abner E, Broster LS, Jicha GA, Schmitt FA, Kryscio R, Andersen A et al (2016) Alzheimer’s biomarkers are correlated with brain connectivity in older adults differentially during resting and task states. Front Aging Neurosci 8:15
Westlye ET, Lundervold A, Rootwelt H, Lundervold AJ, Westlye LT (2011) Increased hippocampal default mode synchronization during rest in middle-aged and elderly APOE ε4 carriers: relationships with memory performance. J Neurosci 31:7775–7783
Mufson EJ, Mahady L, Waters D, Counts SE, Perez SE, DeKosky S, Ginsberg SD, Ikonomovic MD et al (2015) Hippocampal plasticity during the progression of Alzheimer’s disease. Neuroscience IBRO 309:51–67
Wang L, Zang Y, He Y, Liang M, Zhang X, Tian L, Wu T, Jiang T et al (2006) Changes in hippocampal connectivity in the early stages of Alzheimer’s disease: evidence from resting state fMRI. NeuroImage 31:496–504
Farlow MR, He Y, Tekin S, Xu J, Lane R, Charles HC (2004) Impact of APOE in mild cognitive impairment. Neurology 63:1898–1901
Hostage CA, Choudhury KR, Doraiswamy PM, Petrella JR (2014) Mapping the effect of the apolipoprotein e genotype on 4-year atrophy rates in an Alzheimer disease-related brain. Radiology 271:211–219
Honea RA, Vidoni E, Harsha A, Burns JM (2009) Impact of APOE on the healthy aging brain: a voxel-based MRI and DTI study. J Alzheimers Dis 18:553–564
Wang J, Wang X, He Y, Yu X, Wang H, He Y (2015) Apolipoprotein ɛ4 modulates functional brain connectome in Alzheimer’s disease. Hum Brain Mapp 36:1828–1846
Quiroz YT, Budson AE, Celone K, Ruiz A, Newmark R, Castrillõn G, Lopera F, Stern CE (2010) Hippocampal hyperactivation in presymptomatic familial Alzheimer’s disease. Ann Neurol 68:865–875
Filippini N, Ebmeier KP, MacIntosh BJ, Trachtenberg AJ, Frisoni GB, Wilcock GK, Beckmann CF, Smith SM, Matthews PM, Mackay CE (2011) NeuroImage differential effects of the APOE genotype on brain function across the lifespan. Neuroimage. Elsevier Inc; 54: 602–10
Heise V, Filippini N, Trachtenberg AJ, Suri S, Ebmeier KP (2014) Mackay CE. Apolipoprotein E genotype, gender and age modulate connectivity of the hippocampus in healthy adults. Neuroimage. Elsevier Inc; 98: 23–30
Filippini N, Rao A, Wetten S, Gibson RA, Borrie M, Guzman D, Kertesz A, Loy-english I, Williams J, Nichols T, Whitcher B, Matthews PM (2009) NeuroImage anatomically-distinct genetic associations of APOE ɛ4 allele load with regional cortical atrophy in Alzheimer’s disease. Neuroimage. Elsevier Inc.; 44: 724–8
Kim J, Kim Y-H, Lee J-H (2013) Hippocampus-precuneus functional connectivity as an early sign of Alzheimer’s disease: a preliminary study using structural and functional magnetic resonance imaging data. Brain Res Elsevier 1495:18–29
Reiman EM, Quiroz YT, Fleisher AS, Chen K, Velez-Pardo C, Jimenez-Del-Rio M, Fagan AM, Shah AR et al (2007) Brain imaging and fluid biomarker analysis in young adults at genetic risk for autosomal dominant Alzheimer’s disease in the presenilin 1 E280A kindred: a case-control study. Neuroscience Elsevier Ltd 11:162–184
Filippini N, MacIntosh BJ, Hough MG, Goodwin GM, Frisoni GB, Smith SM, Matthews PM, Beckmann CF et al (2009) Distinct patterns of brain activity in young carriers of the APOE-epsilon4 allele. Proc Natl Acad Sci U S A 106:7209–7214
Dennis NA, Browndyke JN, Stokes J, Need A, Burke JR, Welsh-Bohmer KA, Cabeza R (2010) Temporal lobe functional activity and connectivity in young adult APOE-ɛ4 carriers. Alzheimer’s Dement Elsevier Ltd 6:303–311
Mayeux R, Stern Y (2012) Epidemiology of Alzheimer disease. Cold Spring Harb Perspect Med 1:2(8)
Galea M, Woodward M (2005) Mini-Mental State Examination (MMSE). Aust J Physiother 51:198
Nasreddine ZS, Phillips NA, Bédirian V, Charbonneau S, Whitehead V, Collin I, Cummings JL, Chertkow H (2005) The Montreal Cognitive Assessment, MoCA: a brief screening tool for mild cognitive impairment. J Am Geriatr Soc 53:695–699
Lehmann M, Ghosh PM, Madison C, Karydas A, Coppola G, O’Neil JP, Huang Y, Miller BL et al (2013) Greater medial temporal hypometabolism and lower cortical amyloid burden in ApoE4-positive AD patients. J Neurol Neurosurg Psychiatry 85:266–273
Wolk D a, Dickerson BC, Weiner M, Aiello M, Aisen P, Albert MS, Alexander G, Anderson HS et al (2010) Apolipoprotein E (APOE) genotype has dissociable effects on memory and attentional-executive network function in Alzheimer’s disease. Proc Natl Acad Sci 107:10256–10261
Susanto TAK, Pua EPK (2015) Zhou J, Alzheimer’s disease neuroimaging I. Cognition, brain atrophy, and cerebrospinal fluid biomarkers changes from preclinical to dementia stage of Alzheimer’s disease and the influence of apolipoprotein E. J Alzheimers Dis 45:253–268
Shu H, Shi Y, Chen G, Wang Z, Liu D, Yue C, Ward BD, Li W et al (2016) Opposite neural trajectories of apolipoprotein ɛ4 and 2 alleles with aging associated with different risks of Alzheimer’s disease. Cereb Cortex 26(4):1421–1429
Power JD, Barnes KA, Snyder AZ, Schlaggar BL, Petersen SE (2012) Spurious but systematic correlations in functional connectivity MRI networks arise from subject motion. NeuroImage 59:2142–2154
Yan C, Cheung B, Colcombe S, Craddock RC, Kelly C, Martino A Di, Li Q, Castellanos FX, Milham M (2012) The motion crisis in functional connectomics: damage assessment and control for resting-state fMRI. 3rd Bienn Conf Resting State Brain Connect. 2012; 15043
Saad ZS, Gotts SJ, Murphy K, Chen G, Jo HJ, Martin A, Cox RW (2012) Trouble at rest: how correlation patterns and group differences become distorted after global signal regression. Brain Connect 2:25–32
Maldjian JA, Laurienti PJ, Kraft RA, Burdette JH (2003) An automated method for neuroanatomic and cytoarchitectonic atlas-based interrogation of fMRI data sets. NeuroImage 19:1233–1239
Yan CG, Wang XD, Zuo XN, Zang YF (2016) DPABI: data processing & analysis for (resting-state) brain imaging. Neuroinformatics 14:339–351
Tiraboschi P, Hansen L, Masliah E, Alford M, Thal LJ, Corey-Bloom J (2004) Impact of APOE genotype on neuropathologic and neurochemical markers of Alzheimer disease. Neurology 62:1977–1983
Love S, Siew LK, Dawbarn D, Wilcock GK, Ben-Shlomo Y, Allen SJ (2006) Premorbid effects of APOE on synaptic proteins in human temporal neocortex. Neurobiol Aging 27:797–803
Lee GJ, Lu PH, Medina LD, Rodriguez-Agudelo Y, Melchor S, Coppola G, Braskie MN, Hua X et al (2013) Regional brain volume differences in symptomatic and presymptomatic carriers of familial Alzheimer’s disease mutations. J Neurol Neurosurg Psychiatry 84(2):154–162
Toga AW, Thompson PM (2003) Mapping brain asymmetry. Nat Rev Neurosci 4:37–48
Tang AC, Zou B, Reeb BC, Connor JA (2008) An epigenetic induction of a right-shift in hippocampal asymmetry: selectivity for short- and long-term potentiation but not post-tetanic potentiation. Hippocampus 18:5–10
Pedraza O, Bowers D, Gilmore R (2004) Asymmetry of the hippocampus and amygdala in MRI volumetric measurements of normal adults. J Int Neuropsychol Soc 10:664–678
Shi F, Liu B, Zhou Y, Yu C, Jiang T (2009) Hippocampal volume and asymmetry in mild cognitive impairment and Alzheimer’s disease: meta-analyses of MRI studies. 19(11):1055–64
Tohgi H, Takahashi S, Kato E, Homma A, Niina R (1997) Reduced size of right hippocampus in 39- to 80-year-old normal subjects carrying the apolipoprotein E e 4 allele. 236: 21–4
Geroldi C, Laakso MP, DeCarli C, Beltramello A, Bianchetti A, Soininen H, Trabucchi M, Frisoni GB (2000) Apolipoprotein E genotype and hippocampal asymmetry in Alzheimer’s disease: a volumetric MRI study. J Neurol Neurosurg Psychiatry 68:93–96
Eichenbaum H. (2004) Hippocampus: cognitive processes and neural representations that underlie declarative memory the hippocampus serves a critical role in declarative. 44: 109–20
Puthiyedth N, Riveros C, Berretta R, Moscato P (2016) Identification of differentially expressed genes through integrated study of Alzheimer’s disease affected brain regions. PLoS One 11:1–29
Subdivisions M. (2013) The angular gyrus: multiple functions and multiple subdivisions
Ye BS, Seo SW, Lee Y, Kim SY, Choi SH, Lee YM, Kim DH, Han HJ et al (2012) Neuropsychological performance and conversion to Alzheimer’s disease in early- compared to late-onset amnestic mild cognitive impairment: CREDOS study. Dement Geriatr Cogn Disord 34:156–166
Sapkota S, Wiebe SA, Small BJ, Dixon RA (2015) Apolipoprotein E and clusterin can magnify effects of personality vulnerability on declarative memory performance in non-demented older adults. Int J Geriatr Psychiatry. 502–9
Sheline YI, Raichle ME, Snyder AZ, Morris JC, Head D, Wang S, Mintun M (2011) Amyloid plaques disrupt resting state default mode network connectivity in cognitively normal elderly. Biol Psychiatry 67:584–587
Chhatwal JP, Schultz AP, Johnson K, Benzinger TLS, Jack C, Ances BM, Sullivan CA, Salloway SP et al (2013) Impaired default network functional connectivity in autosomal dominant Alzheimer disease. Neurology 81:736–744
Ryan NS, Biessels G-J, Kim L, Nicholas JM, Barber PA, Walsh P, Gami P, Morris HR et al (2015) Genetic determinants of white matter hyperintensities and amyloid angiopathy in familial Alzheimer’s disease. Neurobiol Aging 36:3140–3145
Ringman JM, Medina LD, Braskie M, Rodriguez-Agudelo Y, Geschwind DH, MacIas-Islas MA, Cummings JL, Bookheimer S (2011) Effects of risk genes on BOLD activation in presymptomatic carriers of familial Alzheimer’s disease mutations during a novelty encoding task. Cereb Cortex 21:877–883
Chao OY, Huston JP, Li JS, Wang AL, de Souza Silva MA (2016) The medial prefrontal cortex-lateral entorhinal cortex circuit is essential for episodic-like memory and associative object-recognition. Hippocampus 26(5):633–645
Jin J, Maren S (2015) Prefrontal-hippocampal interactions in memory and emotion. Front Syst Neurosci 9:170
de Souza SM (2016) A., Huston JP, Wang a.-L, Petri D, Chao OY-H. Evidence for a specific integrative mechanism for episodic memory mediated by AMPA/kainate receptors in a circuit involving medial prefrontal cortex and hippocampal CA3 region. Cereb Cortex 26(7):3000–3009
Cavanna AE, Trimble MR (2006) The precuneus: a review of its functional anatomy and behavioural correlates. Brain 129(Pt 3):564–583
Dean DC, Jerskey BA, Chen K, Protas H, Thiyyagura P, Roontiva A, O’Muircheartaigh J, Dirks H et al (2014) Brain differences in infants at differential genetic risk for late-onset Alzheimer disease: a cross-sectional imaging study. JAMA Neurol 71:11–22
Tao J, Liu J, Egorova N, Chen X, Sun S, Xue X, Huang J, Zheng G et al (2016) Increased hippocampus–medial prefrontal cortex resting state functional connectivity and memory function after Tai Chi Chuan practice in elder adults. Front Aging Neurosci 8:1–9
Ward B (2011) AlphaSim program documentation for AFNI, simultaneous inference for fMRI data: Medical College of Wisconsin, Milwaukee
Camchong J, Stenger A, Fein G (2013) Resting-state synchrony during early alcohol abstinence can predict subsequent relapse. Cereb Cortex 23:2086–2099
Acknowledgments
The authors gratefully acknowledge the altruism of all participants and contributions of this research and gratefully acknowledge the grants from Natural Scientific Foundation of China (81322020, 81230032, and 81171313 to L.J.Z. and Nos. 81671667 and 81471644 to G.Z.) and Program for New Century Excellent Talents in University (NCET-12-0260 to L.J.Z.).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflicts of Interest
UJS is a consultant for and/or receives research support from Astellas, Bayer, Bracco, GE, Guerbet, Medrad, and Siemens. All other authors have no conflict of interest to disclose.
Electronic Supplementary Material
Supplementary Table S1
(DOCX 14 kb).
Supplementary Table S2
(DOCX 14 Ckb).
Supplementary Table S3
(DOCX 14 kb).
Supplementary Table S4
(DOCX 14 kb).
Supplementary Table S5
(DOCX 14 kb).
Supplementary Table S6
(DOCX 14 kb).
Supplementary Table S7
(DOCX 14 kb).
Supplementary Table S8
(DOCX 14 kb).
Rights and permissions
About this article
Cite this article
Zheng, L.J., Su, Y.Y., Wang, Y.F. et al. Different Hippocampus Functional Connectivity Patterns in Healthy Young Adults with Mutations of APP/Presenilin-1/2 and APOEε4 . Mol Neurobiol 55, 3439–3450 (2018). https://doi.org/10.1007/s12035-017-0540-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-017-0540-4