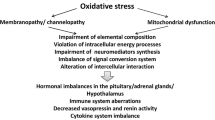
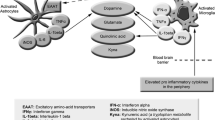
Abstract
There is evidence that immune-inflammatory and oxidative and nitrosative stress (O&NS) pathways play a role in the pathophysiology of myalgic encephalomyelitis (ME)/chronic fatigue syndrome (CFS). There is also evidence that these neuroimmune diseases are accompanied by hypothalamic-pituitary-adrenal (HPA) axis hypoactivity as indicated by lowered baseline glucocorticoid levels. This paper aims to review the bidirectional communications between immune-inflammatory and O&NS pathways and HPA axis hypoactivity in ME/CFS, considering two possibilities: (a) Activation of immune-inflammatory pathways is secondary to HPA axis hypofunction via attenuated negative feedback mechanisms, or (b) chronic activated immune-inflammatory and O&NS pathways play a causative role in HPA axis hypoactivity. Electronic databases, i.e., PUBMED, Scopus, and Google Scholar, were used as sources for this narrative review by using keywords CFS, ME, cortisol, ACTH, CRH, HPA axis, glucocorticoid receptor, cytokines, immune, immunity, inflammation, and O&NS. Findings show that activation of immune-inflammatory and O&NS pathways in ME/CFS are probably not secondary to HPA axis hypoactivity and that activation of these pathways may underpin HPA axis hypofunction in ME/CFS. Mechanistic explanations comprise increased levels of tumor necrosis factor-α, T regulatory responses with elevated levels of interleukin-10 and transforming growth factor-β, elevated levels of nitric oxide, and viral/bacterial-mediated mechanisms. HPA axis hypoactivity in ME/CFS is most likely a consequence and not a cause of a wide variety of activated immune-inflammatory and O&NS pathways in that illness.
Similar content being viewed by others
References
Maes M, Twisk FNM (2010) Chronic fatigue syndrome: Harvey and Wessely’s (bio)psychosocial model versus a bio(psychosocial) model based on inflammatory and oxidative and nitrosative stress pathways. BMC Med 8(1):35
Morris G, Maes M (2013) Myalgic encephalomyelitis/chronic fatigue syndrome and encephalomyelitis disseminata/multiple sclerosis show remarkable levels of similarity in phenomenology and neuroimmune characteristics. BMC Med 11(1):205
Fukuda K (1994) The chronic fatigue syndrome: a comprehensive approach to its definition and study. Ann Int Med 121(12):953
Maes M, Berk M, Goehler L, Song C, Anderson G, Galecki P (2012) Depression and sickness behavior are Janus-faced responses to shared inflammatory pathways. BMC Med 10:66
Carruthers B, van de Sande M, De Meirleir K, Klimas N, Broderick G, Mitchell T, Staines D, Powles A et al (2011) Myalgic encephalomyelitis: international consensus criteria. J Int Med 270:327–338
Maes M, Twisk FN, Johnson C (2012) Myalgic encephalomyelitis (ME), chronic fatigue syndrome (CFS), and chronic fatigue (CF) are distinguished accurately: results of supervised learning techniques applied on clinical and inflammatory data. Psychiatry Res 200(2–3):754–760
Anderson G, Berk M, Maes M (2014) Biological phenotypes underpin the physio-somatic symptoms of somatization, depression, and chronic fatigue syndrome. Acta Psychiatr Scand 129(2):83–97
Maes M, Twisk F, Kubera M, Ringel K (2012) Evidence for inflammation and activation of cell-mediated immunity in myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS): increased interleukin-1, tumor necrosis factor-alpha, PMN-elastase, lysozyme and neopteri. J Affect Disord 136:933–939
Fletcher M, Zeng X, Barnes Z, Levis S, Klimas NG (2009) Plasma cytokines in women with chronic fatigue syndrome. J Transl Med 7(1):96
Papadopoulos AS, Cleare AJ (2011) Hypothalamic–pituitary–adrenal axis dysfunction in chronic fatigue syndrome. Nat Rev Endocrinol 8(1):22–32
Tak LM, Cleare AJ, Ormel J, Manoharan A, Kok IC, Wessely S, Rosmalen JGM (2011) Meta-analysis and meta-regression of hypothalamic-pituitary-adrenal axis activity in functional somatic disorders. Biol Psychol 87(2):183–194
Gaillard RC (2003) Interactions between the hypothalamo-pituitary-adrenal axis and the immunological system. The ACTH Axis: Pathogenesis, Diagnosis and Treatment. Springer Science, Business Media. doi: 10.1007/978–1–4615-0501-3_6
Maes M, Anderson G, Kubera M, Berk M (2014) Targeting classical IL-6 signalling or IL-6 trans-signalling in depression? Exp Opin Ther Targets 18(5):495–512
Oppong E, Cato AC (2015) Effects of glucocorticoids in the immune system. Adv Exp Med Biol 872:217–233
Franchimont D (2004) Overview of the actions of glucocorticoids on the immune response: a good model to characterize new pathways of Immunosuppression for new treatment strategies. Ann N Y Acad Sci 1024(1):124–137
Coutinho AE, Chapman KE (2011) The anti-inflammatory and immunosuppressive effects of glucocorticoids, recent developments and mechanistic insights. Mol Cell Endocrinol 335(1):2–13
Zhong H-J, Wang H-Y, Yang C, Zhou J-Y, Jiang J-X (2013) Low concentrations of corticosterone exert stimulatory effects on macrophage function in a manner dependent on glucocorticoid receptors. Int J Endocrinol 2013:1–9
Van Den Eede F, Moorkens G, Van Houdenhove B, Cosyns P, Claes SJ (2007) Hypothalamic-pituitary-adrenal Axis function in chronic fatigue syndrome. Neuropsychobiol 55(2):112–120
Silverman MN, Pearce BD, Biron CA, Miller AH (2005) Immune modulation of the hypothalamic-pituitary-adrenal (HPA) Axis during viral infection. Viral Immunol 18(1):41–78
Zunszain PA, Anacker C, Cattaneo A, Carvalho LA, Pariante CM (2011) Glucocorticoids, cytokines and brain abnormalities in depression. Progr Neuro-Psychopharmacol Biol Psychiatry 35(3):722–729
Roberts ADL (2004) Salivary cortisol response to awakening in chronic fatigue syndrome. Brit J Psychiatry 184(2):136–141
Jerjes WK, Cleare AJ, Wessely S, Wood PJ, Taylor NF (2005) Diurnal patterns of salivary cortisol and cortisone output in chronic fatigue syndrome. J Affect Disord 87(2–3):299–304
MacHale SM, Cavanagh JTO, Bennie J, Carroll S, Goodwin GM, Lawrie SM (1998) Diurnal variation of adrenocortical activity in chronic fatigue syndrome. Neuropsychobiol 38(4):213–217
Gur A, Cevik R, Nas K, Colpan L, Sarac S (2004) Cortisol and hypothalamic–pituitary–gonadal axis hormones in follicular-phase women with fibromyalgia and chronic fatigue syndrome and effect of depressive symptoms on these hormones. Arthr Res Ther 6(3):R232–R238
Demitrack MA, Dale JK, Straus SE, Laue L, Listwak SJ, Kruesi MJP, Chrousos GP, Gold PW (1991) Evidence for impaired activation of the hypothalamic-pituitary-adrenal Axis in patients with chronic fatigue syndrome. J Clin Endocrinol Metab 73(6):1224–1234
Roberts ADL, Charler ML, Papadopoulos A, Wessely S, Chalder T, Cleare AJ (2009) Does hypocortisolism predict a poor response to cognitive behavioural therapy in chronic fatigue syndrome? Psychol Med 40(03):515
Cleare AJ (2004) The HPA axis and the genesis of chronic fatigue syndrome. Trends Endocrinol Metabol 15(2):55–59
Crofford LJ, Young EA, Engleberg NC, Korszun A, Brucksch CB, McClure LA, Brown MB, Demitrack MA (2004) Basal circadian and pulsatile ACTH and cortisol secretion in patients with fibromyalgia and/or chronic fatigue syndrome. Brain Beh Imm 18(4):314–325
Torpy DJ, Ho JT (2007) Corticosteroid-binding globulin gene polymorphisms: clinical implications and links to idiopathic chronic fatigue disorders. Clin Endocrinol 67(2):161–167
Torres-Harding S, Sorenson M, Jason L, Reynolds N, Brown M, Maher K, Fletcher MA (2008) The associations between basal salivary cortisol and illness symptomatology in chronic fatigue syndrome. J Applied Biobehav Res 13(3):157–180
Scott LV, Medbak S, Dinan TG (1999) Desmopressin augments pituitary–adrenal responsivity to corticotropin-releasing hormone in subjects with chronic fatigue syndrome and in healthy volunteers. Biol Psychiatry 45(11):1447–1454
Kuratsune H, Yamaguti K, Sawada M, Kodate S, Machii T, Kanakura Y, Kitani T (1998) Dehydroepiandrosterone sulfate deficiency in chronic fatigue syndrome. Int J Mol Med. doi:10.3892/ijmm.1.1.143
Maes M, Mihaylova I, Leunis J (2005) In chronic fatigue syndrome, the decreased levels of omega-3 poly-unsaturated fatty acids are related to lowered serum zinc and defects in T cell activation. Neuro Endocrinol Lett 26:745–751
De Becker P, De Meirleir K, Joos E, Campine I, Van Steenberge E, Smitz J, Velkeniers B (1999) Dehydroepiandrosterone (DHEA) response to i.V. ACTH in patients with chronic fatigue syndrome. Horm Metabol Res 31(01):18–21
Himmel PB, Seligman TM (1999) A pilot study employing dehydroepiandrosterone (DHEA) in the treatment of chronic fatigue syndrome. J Clin Rheumatol 5(2):56–59
Scott LV, Salahuddin F, Cooney J, Svec F, Dinan TG (1999) Differences in adrenal steroid profile in chronic fatigue syndrome, in depression and in health. J Affect Disord 54(1–2):129–137
Scott LV, Medbak S, Dinan TG (1998) Blunted adrenocorticotropin and cortisol responses to corticotropin-releasing hormone stimulation in chronic fatigue syndrome. Acta Psychiatr Scand 97(6):450–457
Scott LV, Teh J, Reznek R, Martin A, Sohaib A (1999) G. Dinan T. Small adrenal glands in chronic fatigue syndrome: a preliminary computer tomography study. Psychoneuroendocrinol 24(7):759–768
Gaab J, Hüster D, Peisen R, Engert V, Heitz V, Schad T, Schürmeyer TH, Ehlert U (2002) Hypothalamic-pituitary-adrenal axis reactivity in chronic fatigue syndrome and health under psychological, physiological, and pharmacological stimulation. Psychosom Med 64(6):951–962
Dinan TG, Majeed T, Lavelle E, Scott LV, Berti C, Behan P (1997) Blunted serotonin-mediated activation of the hypothalamic-pituitary-adrenal axis in chronic fatigue syndrome. Psychoneuroendocrinol 22(4):261–267
Segal TY, Hindmarsh PC, Viner RM (2005) Disturbed adrenal function in adolescents with chronic fatigue syndrome. J Pediatric Endocrinol Metabol 18(3):295–301
Jerjes WK, Taylor NF, Wood PJ, Cleare AJ (2007) Enhanced feedback sensitivity to prednisolone in chronic fatigue syndrome. Psychoneuroendocrinology 32(2):192–198
Visser J, Lentjes E, Haspels I, Graffelman W, Blauw B, de Kloet R, Nagelkerken L (2001) Increased sensitivity to glucocorticoids in peripheral blood mononuclear cells of chronic fatigue syndrome patients, without evidence for altered density or affinity of glucocorticoid receptors. J Investig Med 49(2):195–204
Visser J, Blauw B, Hinloopen B, Brommer E, de Kloet ER, Kluft C, Nagelkerken L (1998) CD4 T lymphocytes from patients with chronic fatigue syndrome have decreased interferon-γ production and increased sensitivity to dexamethasone. J Inf Dis 177(2):451–454
Visser JTJ, De Kloet ER, Nagelkerken LEX (2000) Altered glucocorticoid regulation of the immune response in the chronic fatigue syndrome. Ann N Y Acad Sci 917(1):868–875
Gaab J, Rohleder N, Heitz V, Engert V, Schad T, Schürmeyer TH, Ehlert U (2005) Stress-induced changes in LPS-induced pro-inflammatory cytokine production in chronic fatigue syndrome. Psychoneuroendocrinol 30(2):188–198
Fries E, Hesse J, Hellhammer J, Hellhammer DH (2005) A new view on hypocortisolism. Psychoneuroendocrinol 30(10):1010–1016
Horn CAC, Pietrzak RH, Corsi-Travali S, Neumeister A (2014) Linking plasma cortisol levels to phenotypic heterogeneity of posttraumatic stress symptomatology. Psychoneuroendocrinol 39:88–93
Erkut ZA, Endert E, Huitinga I, Swaab DF (2002) Cortisol is increased in postmortem cerebrospinal fluid of multiple sclerosis patients: relationship with cytokines and sepsis. Mult Scler 8(3):229–236
Carroll BJ, Cassidy F, Naftolowitz D, Tatham NE, Wilson WH, Iranmanesh A, Liu PY, Veldhuis JD (2007) Pathophysiology of hypercortisolism in depression. Acta Psychiatr Scand 115(s433):90–103
Sharma ST, Nieman LK (2011) Cushing’s syndrome: all variants, detection, and treatment. Endocrinol Metabol Clin North Am 40(2):379–391
Barnes PJ, Adcock IM (2009) Glucocorticoid resistance in inflammatory diseases. Lancet 373(9678):1905–1917
Wichers M, Maes M (2002) The psychoneuroimmuno-pathophysiology of cytokine-induced depression in humans. Int J Neuropsychopharmacol 5(4):375–388
Maes M, Ringel K, Kubera M, Anderson G, Morris G, Galecki P, Geffard M (2013) In myalgic encephalomyelitis/chronic fatigue syndrome, increased autoimmune activity against 5-HT is associated with immuno-inflammatory pathways and bacterial translocation. J Affect Disord 150(2):223–230
Maes M, Mihaylova I, Kubera M, Bosmans E (2007) Not in the mind but in the cell: increased production of cyclo-oxygenase-2 and inducible NO synthase in chronic fatigue syndrome. Neuro Endocrinol Lett 28:463–469
Kerr JR (2008) Gene profiling of patients with chronic fatigue syndrome/myalgic encephalomyelitis. Curr Rheumatol Rep 10(6):482–491
Cheney P, Dorman S, Bell D (1989) Interleukin-2 and the chronic fatigue syndrome. Ann Intern Med 110:321
Gow J, Hagan S, Herzyk P, Cannon C, Behan P, Chaudhuri A (2009) A gene signature for post-infectious chronic fatigue syndrome. BMC Med Genet 2:38
Brenu E, van Driel M, Staines D, Ashton K, Ramos S, Keane J (2011) Immunological abnormalities as potential biomarkers in chronic fatigue syndrome/myalgic encephalomyelitis. J Transl Med 9:81
Mawle AC, Nisenbaum R, Dobbins JG, Gary HE, Stewart JA, Reyes M, Steele L, Schmid DS et al (1997) Immune responses associated with chronic fatigue syndrome: a case-control study. J Inf Dis 175:136–141
Buchwald D, Wener M, Pearlman T, Kith P (1997) Markers of inflammation and immune activation in chronic fatigue and chronic fatigue syndrome. J Rheumatol 24:372–376
Skowera A, Cleare A, Blair D, Bevis L, Wessely SC, Peakman M (2004) High levels of type 2 cytokine-producing cells in chronic fatigue syndrome. Clin Exp Immunol 135(2):294–302
Lee Hardcastle S (2014) Analysis of the relationship between immune dysfunction and symptom severity in patients with chronic fatigue syndrome/myalgic encephalomyelitis (CFS/ME). J Clin Cell Immunol 5(01)
Maes M, Mihaylova I, Leunis JC (2006) Chronic fatigue syndrome is accompanied by an IgM-related immune response directed against neopitopes formed by oxidative or nitrosative damage to lipids and proteins. Neuro Endocrinol Lett 27(5):615–621
Maes M, Mihaylova I, Kubera M, Uytterhoeven M, Vrydags N, Bosmans E (2009) Coenzyme Q10 deficiency in myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS) is related to fatigue, autonomic and neurocognitive symptoms and is another risk factor explaining the early mortality in ME/CFS due to cardiovascular disorder. Neuro Endocrinol Lett 30:470–476
Maes M, Mihaylova I, Leunis J (2006) Chronic fatigue syndrome is accompanied by an IgM-related immune response directed against neopitopes formed by oxidative or nitrosative damage to lipids and proteins. Neuro Endocrinol Lett 27:615–621
Maes M, Twisk FNM, Kubera M, Ringel K (2012) Evidence for inflammation and activation of cell-mediated immunity in myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS): increased interleukin-1, tumor necrosis factor-α, PMN-elastase, lysozyme and neopterin. J Affect Disord 136(3):933–939
Maes M, Mihaylova I, Leunis J-C (2007) Increased serum IgA and IgM against LPS of enterobacteria in chronic fatigue syndrome (CFS): indication for the involvement of gram-negative enterobacteria in the etiology of CFS and for the presence of an increased gut–intestinal permeability. J Affect Disord 99(1–3):237–240
Maes M, Twisk FNM, Kubera M, Ringel K, Leunis J-C, Geffard M (2012) Increased IgA responses to the LPS of commensal bacteria is associated with inflammation and activation of cell-mediated immunity in chronic fatigue syndrome. J Affect Disord 136(3):909–917
Trivedi PP, Jena GB (2013) Melatonin reduces ulcerative colitis-associated local and systemic damage in mice: investigation on possible mechanisms. Dig Dis Sci 58(12):3460–3474
Van Houdenhove B, Eede FVD, Luyten P (2009) Does hypothalamic–pituitary–adrenal axis hypofunction in chronic fatigue syndrome reflect a ‘crash’ in the stress system? Med Hypotheses 72(6):701–705
Moutschen M, Triffaux JM, Demonty J, Legros JJ, Lefebvre PJ (1994) Pathogenic tracks in fatigue syndromes. Acta Clin Belg 49(6):274–289
O’Connor TM, O’Halloran DJ, Shanahan F (2000) The stress response and the hypothalamic-pituitary-adrenal axis: from molecule to melancholia. QJM 93(6):323–333
Heim C, Newport DJ, Bonsall R, Miller AH, Nemeroff CB (2003) Altered pituitary-adrenal Axis responses to provocative challenge tests in adult survivors of childhood abuse. FOC 1 3:282–289
Lucki NC, Li D, Sewer MB (2012) Sphingosine-1-phosphate rapidly increases cortisol biosynthesis and the expression of genes involved in cholesterol uptake and transport in H295R adrenocortical cells. Mol Cell Endocrinol 348(1):165–175
Dillon J (2005) Dehydroepiandrosterone, dehydroepiandrosterone sulfate and related steroids: their role in inflammatory, allergic and immunological disorders. CDTIA 4(3):377–385
Moss R, Mercandetti A, Vojdani A (1999) TNF-alpha and chronic fatigue syndrome. J Clin Immunol 19:314–316
Van Houdenhove B, Luyten P (2009) Treatment of chronic fatigue syndrome: how to find a ‘new equilibrium’? Patient Educ Couns 77(2):153–154
Van Houdenhove B (2006) What is the aim of cognitive behaviour therapy in patients with chronic fatigue syndrome? Psychother Psychosom 75(6):396–397
Parker AJR, Wessely S, Cleare AJ (2001) The neuroendocrinology of chronic fatigue syndrome and fibromyalgia. Psychol Med 31:08
Cleare AJ (2003) The neuroendocrinology of chronic fatigue syndrome. Endocrinol Rev 24(2):236–252
Gaab J, Engert V, Heitz V, Schad T, Schürmeyer TH, Ehlert U (2004) Associations between neuroendocrine responses to the insulin tolerance test and patient characteristics in chronic fatigue syndrome. J Psychosom Res 56(4):419–424
Hickie I (2006) Post-infective and chronic fatigue syndromes precipitated by viral and non-viral pathogens: prospective cohort study. BMJ 333(7568):575–570
Candy B, Chalder T, Cleare AJ, Peakman A, Skowera A, Wessely S, Weinman J, Zuckerman M et al (2003) Predictors of fatigue following the onset of infectious mononucleosis. Psychol Med 33(5):847–855
Whiting P, Bagnall A-M, Sowden AJ, Cornell JE, Mulrow CD, Ramírez G (2001) Interventions for the treatment and management of chronic fatigue syndrome. JAMA 286(11):1360
McKenzie R, O’Fallon A, Dale J et al (1998) Low-dose hydrocortisone for treatment of chronic fatigue syndrome: a randomized controlled trial. JAMA 280(12):1061–1066
Gupta S, Aggarwal S, See D, Starr A (1997) Cytokine production by adherent and non-adherent mononuclear cells in chronic fatigue syndrome. J Psychiatr Res 31(1):149–156
Nas K, Cevik R, Batum S, Sarac AJ, Acar S, Kalkanli S (2011) Immunologic and psychosocial status in chronic fatigue syndrome. Bratislavske Lekarske Listy 112(4):208–212
Fang H, Xie Q, Boneva R, Fostel J, Perkins R, Tong W (2006) Gene expression profile exploration of a large dataset on chronic fatigue syndrome. Pharmacogenomics 7(3):429–440
Turnbull AV, Lee S, Rivier C (1998) Mechanisms of hypothalamic-pituitary-adrenal axis stimulation by immune signals in the adult rata. Ann N Y Acad Sci 840(1):434–443
Reincke M, Heppner C, Petzke F, Allolio B, Arlt W, Mbulamberi D, Siekmann L, Vollmer D et al (1994) Impairment of adrenocortical function associated with increased plasma tumor necrosis factor-alpha and interleukin-6 concentrations in African Trypanosomiasis. Neuroimmunomodulation 1(1):14–22
Jäättelä M, Ilvesmaki V, Voutilainen R, Stenman ULFH, Saksela E (1991) Tumor necrosis factor as a potent inhibitor of adrenocorticotropin-induced cortisol production and steroidogenic P450 enzyme gene expression in cultured human fetal adrenal cells. Endocrinol 128(1):623–629
Jäättelä M, Carpén O, Stenman UH, Saksela E (1990) Regulation of ACTH-induced steroidogenesis in human fetal adrenals by rTNF-α. Mol Cell Endocrinol 68(2–3):R31–R36
Barney M, Call GB, McIlmoil CJ, Husein OF, Adams A, Balls AG, Oliveira GK, Miner EC et al (2000) Stimulation by interleukin-6 and inhibition by tumor necrosis factor of cortisol release from bovine adrenal zona fasciculata cells through their receptors. ENDO 13(3):369–377
Mikhaylova IV, Kuulasmaa T, Jääskeläinen J, Voutilainen R (2007) Tumor necrosis factor-α regulates steroidogenesis, apoptosis, and cell viability in the human adrenocortical cell line NCI-H295R. Endocrinol 148:386–392
Ozbay T, Merrill AH, Sewer MB (2004) ACTH regulates steroidogenic gene expression and cortisol biosynthesis in the human adrenal cortex via sphingolipid metabolism. Endocr Res 30:787–794
White AT, Light AR, Hughen RW, Bateman L, Martins TB, Hill HR, Light KC (2010) Severity of symptom flare after moderate exercise is linked to cytokine activity in chronic fatigue syndrome. Psychophysiology. doi:10.1111/j.1469-8986.2010.00978.x
Nakamura T, Schwander SK, Donnelly R, Ortega F, Togo F, Broderick G, Yamamoto Y, Cherniack NS et al (2010) Cytokines across the night in chronic fatigue syndrome with and without fibromyalgia. Clin Vaccine Immunol 17:582–587
Zhang H-Y, Liu Z-D, Hu C-J, Wang D-X, Zhang Y-B, Li Y-Z (2011) Up-regulation of TGF-β1 mRNA expression in peripheral blood mononuclear cells of patients with chronic fatigue syndrome. J Formosan Med Ass 110:701–704
White PD, Nye KE, Pinching AJ, Yap TM, Power N, Vleck V, Bentley DJ, Thomas JM et al (2008) Immunological changes after both exercise and activity in chronic fatigue syndrome. J Chron Fatigue Syndr 12:51–66
Chao C, Janoff E, Hu S, Thomas K, Gallagher M, Tsang M, Peterson P (1991) Altered cytokine release in peripheral blood mononuclear cell cultures from patients with the chronic fatigue syndrome. Cytokine 3:292–298
Bennett AL, Chao CC, Hu S, Buchwald D, Fagioli LR, Schur PH, Peterson PK, Komaroff AL (1997) Elevation of bioactive transforming growth factor-beta in serum from patients with chronic fatigue syndrome. J Clin Immunol 17(2):160–166
Murray PJ (2005) The primary mechanism of the IL-10-regulated antiinflammatory response is to selectively inhibit transcription. Proc Nat Acad Sci USA 102(24):8686–8691
Sanjabi S, Zenewicz LA, Kamanaka M, Flavell RA (2009) Anti-inflammatory and pro-inflammatory roles of TGF-β, IL-10, and IL-22 in immunity and autoimmunity. Curr Opin Pharmacol 9(4):447–453
Roque S, Correia-Neves M, Mesquita AR, Palha JA, Sousa N (2009) Interleukin-10: a key cytokine in depression? Cardiovasc Psychiatr Neurol 2009:1–5
Koldzic-Zivanovic N, Tu H, Juelich TL, Rady PL, Tyring SK, Hudnall SD, Smith EM, Hughes TK (2006) Regulation of adrenal glucocorticoid synthesis by interleukin-10: a preponderance of IL-10 receptor in the adrenal zona fasciculata. Brain Beh Imm 20:460–468
Tu H, Juelich T, Smith EM, Tyring SK, Rady PL, Hughes TK (2003) Evidence for endogenous interleukin-10 during nociception. J Neuroimmunol 139(1–2):145–149
Stefano GB, Prevot V, Beauvillain J-C, Hughes TK (1998) Interleukin-10 stimulation of corticotrophin releasing factor median eminence in rats: evidence for dependence upon nitric oxide production. Neurosci Lett 256(3):167–170
Rainey WE, Naville D, Saez JM, Carr BR, Byrd W, Magness RR, Mason JI (1990) Transforming growth factor-β inhibits steroid 17α-hydroxylase cytochrome P-450 expression in ovine adrenocortical cells. Endocrinol 127(4):1910–1915
Creed TJ, Lee RW, Newcomb PV, di Mambro AJ, Raju M, Dayan CM (2009) The effects of cytokines on suppression of lymphocyte proliferation by dexamethasone. J Immunol 183(1):164–171
Franchimont D, Martens H, Hagelstein M-T, Louis E, Dewe W, Chrousos GP, Belaiche J, Geenen V (1999) Tumor necrosis factor α decreases, and interleukin-10 increases, the sensitivity of human monocytes to dexamethasone: potential regulation of the glucocorticoid receptor. J Clin Endocrinol Metab 84(8):2834–2839
Perrin A, Pascal O, Defaye G, Feige J-J, Chambaz EM (1991) Transforming growth factor β 1 is a negative regulator of steroid 17α-hydroxylase expression in bovine adrenocortical cells. Endocrinol 128(1):357–362
Liakos P (2003) Transforming growth factor beta1 inhibits aldosterone and cortisol production in the human adrenocortical cell line NCI-H295R through inhibition of CYP11B1 and CYP11B2 expression. J Endocrinol 176(1):69–82
Stankovic AK, Dion LD, Parker CR (1994) Effects of transforming growth factor-β on human fetal adrenal steroid production. Mol Cell Endocrinol 99(2):145–151
Parker CR, Stankovic AK, Faye-petersen O, Falany CN, Li H, Jian M (1998) Effects of acth and cytokines on dehydroepiandrosterone sulfotransferase messenger RNA in human adrenal cells. Endocr Res 24(3–4):669–673
Li T, Iwaki H, Fu R, Hasegawa Y, Zhang H, Liu A (2006) Alpha-amino-SZ-carboxymuconate-epsilon-semialdehyde decarboxylase (ACMSD) is a new member of the amidohydrolase superfamily. Biochemist 45:6628–6634
Bugajski J, Gadek-Michalska A, Bugajski AJ (2004) Nitric oxide and prostaglandin systems in the stimulation of hypothalamic-pituitary-adrenal axis by neurotransmitters and neurohormones. J Physiol Pharmacol 55(4):679–703
Givalois L, Li S, Pelletier G (2002) Central nitric oxide regulation of the hypothalamic–pituitary–adrenocortical axis in adult male rats. Mol Brain Res 102(1–2):1–8
Weidenfeld J, Feldman S, DeKeyser FG, Ovadia H (1999) Effect of exogenous nitric oxide and inhibitors of nitric oxide synthase on the hypothalamic pituitary adrenal axis responses to neural stimuli. Neuroendocrinol 70(3):153–159
Ducsay CA, Myers DA (2011) eNOS activation and NO function: differential control of steroidogenesis by nitric oxide and its adaptation with hypoxia. J Endocrinol 210(3):259–269
Palacios M, Knowles RG, Palmer RMJ, Moncada S (1989) Nitric oxide from L-arginine stimulates the soluble guanylate cyclase in adrenal glands. Biochem Biophysic Res Comm 165(2):802–809
Drewett JG, Adams-Hays RL, Ho BY, Hegge DJ (2002) Nitric oxide potently inhibits the rate-limiting enzymatic step in steroidogenesis. Mol Cell Endocrinol 194(1–2):39–50
Cymeryng CB, Lotito SP, Colonna C, Finkielstein C, Pomeraniec Y, Grión N, Gadda L, Maloberti P et al (2002) Expression of nitric oxide synthases in rat adrenal zona fasciculata cells. Endocrinology 143(4):1235–1242
Repetto EM (2006) Characterization of L-arginine transport in adrenal cells: effect of ACTH. AJP: Endocrinol Metabol 291(2):E291–E297
Zimmerman PA, Betz JA, Poth M (1993) Nitric oxide: a potential modulator of adrenal hormone secretion. Pediatr Res 33:S21–S21
Nicolson G, Nasralla M, De Meirleir K, Gan R, Haier J (2003) Evidence for bacterial (mycoplasma, Chlamydia) and viral (HHV-6) co-infections in chronic fatigue syndrome patients. JCFS 11:7–20
Beishuizen A, Thijs LG (2003) Review: endotoxin and the hypothalamo-pituitary-adrenal (HPA) axis. J Endotoxin Res 9(1):3–24
Vallès A, Martı́ O, Harbuz MS, Armario A (2002) A single lipopolysaccharide administration is sufficient to induce a long-term desensitization of the hypothalamic–pituitary–adrenal axis. Neuroscience 112(2):383–389
Linde A, Andersson B, Svenson SB, Ahrne H, Carlsson M, Forsberg P, Hugo H, Karstorp A et al (1992) Serum levels of lymphokines and soluble cellular receptors in primary Epstein-Barr virus infection and in patients with chronic fatigue syndrome. J Infect Dis 165(6):994–1000
Grinevich V, Ma XM, Herman JP, Jezova D, Akmayev I, Aguilera G (2001) Effect of repeated lipopolysaccharide administration on tissue cytokine expression and hypothalamic-pituitary-adrenal axis activity in rats. J Neuroendocrinol 13(8):711–723
Eskan MA, Rose BG, Benakanakere MR, Zeng Q, Fujioka D, Martin MH, Lee M-J, Kinane DF (2008) TLR4 and S1P receptors cooperate to enhance inflammatory cytokine production in human gingival epithelial cells. Eur J Immunol 38(4):1138–1147
Hughes JE, Srinivasan S, Lynch KR, Proia RL, Ferdek P, Hedrick CC (2008) Sphingosine-1-phosphate induces an antiinflammatory phenotype in macrophages. Circ Res 102(8):950–958
Sheng R, Xu X, Tang Q, Bian D, Li Y, Qian C, He X, Gao X et al (2011) Polysaccharide of radix Pseudostellariae improves chronic fatigue syndrome induced by poly I:C in mice. Evid-Bas Compl Alternat Med 2011:1–9
Benitez BA, Cooper B, Pastor P, Jin S-C, Lorenzo E, Cervantes S, Cruchaga C TREM2 is associated with the risk of Alzheimer’s disease in Spanish population. Neurobiol Aging 34(6):1711.e1715–1711.e1717
Johnson EO, Kostandi M, Moutsopoulos HM (2006) Hypothalamic-pituitary-adrenal axis function in Sjogren’s syndrome: mechanisms of neuroendocrine and immune system homeostasis. Ann N Y Acad Sci 1088(1):41–51
Tzioufas AG, Tsonis J, Moutsopoulos HM (2008) Neuroendocrine dysfunction in Sjogren’s syndrome. Neuroimmunomodulation 15(1):37–45
Shah D, Kiran R, Wanchu A, Bhatnagar A (2009) Relationship between T lymphocyte subsets and cortisol in systemic lupus erythematosus. Kathmandu University Med J (KUMJ) 7(27):213–219
Liu SY, Han LS, Guo JY, Zheng ZH, Li H, Zhang L, Zhang X, He YJ et al (2013) Metabolic syndrome in Chinese patients with systemic lupus erythematosus: no association with plasma cortisol level. Lupus 22(5):519–526
van der Goes MC, Bossema ER, Hartkamp A, Godaert GLR, Jacobs JWG, Kruize AA, Derksen RHWM, Bijlsma JWJ et al (2010) Cortisol during the day in patients with systemic lupus erythematosus or primary Sjogren’s syndrome. J Rheumatol 38(2):285–288
Sawalha AH, Kovats S (2008) Dehydroepiandrosterone in systemic lupus erythematosus. Curr Rheumatol Rep 10(4):286–291
Pool AJ (2004) Serum cortisol reduction and abnormal prolactin and CD4+/CD8+ T-cell response as a result of controlled exercise in patients with rheumatoid arthritis and systemic lupus erythematosus despite unaltered muscle energetics. Rheumatology 43(1):43–48
Miller BE, Deutsch O, Redlich M, Konttinen YT, Benoliel R, Zaks B, Davidovich E, Palmon A et al (2011) Sialochemistry and cortisol levels in patients with Sjogren’s syndrome. Oral Dis 18(3):255–259
Johnson EO, Moutsopoulos HM (2000) Neuroendocrine manifestations in Sjogren’s syndrome. Relation to the neurobiology of stress. Ann N Y Acad Sci 917:797–808
Mavragani CP, Fragoulis GE, Moutsopoulos HM (2012) Endocrine alterations in primary Sjogren’s syndrome: an overview. J Autoimmun 39(4):354–358
Straub RH (2004) Renal clearance and daily excretion of cortisol and adrenal androgens in patients with rheumatoid arthritis and systemic lupus erythematosus. Ann Rheum Dis 63(8):961–968
Alten R, Doring G, Cutolo M, Gromnica-Ihle E, Witte S, Straub R, Buttgereit F (2010) Hypothalamus-pituitary-adrenal axis function in patients with rheumatoid arthritis treated with nighttime-release prednisone. J Rheumatol 37(10):2025–2031
Jessop DS (2005) A defect in cortisol production in rheumatoid arthritis: why are we still looking? Rheumatol 44(9):1097–1100
Eijsbouts AMM, Kempers MJE, Kramer RSA, Hopman MTE, van den Hoogen FHJ, Laan RFJM, Hermus ARMM, Sweep FCGJ et al (2009) Effect of naproxen on the hypothalamic-pituitary-adrenal axis in healthy volunteers. Br J Clin Pharmacol 67(1):22–28
Straub RH, Paimela L, Peltomaa R, Schölmerich J, Leirisalo-Repo M (2002) Inadequately low serum levels of steroid hormones in relation to interleukin-6 and tumor necrosis factor in untreated patients with early rheumatoid arthritis and reactive arthritis. Arthr Rheum 46(3):654–662
Masi AT, Aldag JC (2005) Integrated neuroendocrine immune risk factors in relation to rheumatoid arthritis: should rheumatologists now adopt a model of a multiyear, presymptomatic phase? Scand J Rheumatol 34(5):342–352
Herrmann M, SchÖLmerich J, Straub RH (2002) Influence of cytokines and growth factors on distinct steroidogenic enzymes in vitro. Ann N Y Acad Sci 966(1):166–186
Straub RH, Härle P, Yamana S, Matsuda T, Takasugi K, Kishimoto T, Nishimoto N (2006) Anti–interleukin-6 receptor antibody therapy favors adrenal androgen secretion in patients with rheumatoid arthritis: a randomized, double-blind, placebo-controlled study. Arthritis Rheum 54(6):1778–1785
Straub RH, Cutolo M (2006) Further evidence for insufficient hypothalamic-pituitary-glandular axes in polymyalgia rheumatica. J Rheumatol 33(7):1219–1223
Imrich R, Vlcek M, Kerlik J, Vogeser M, Kirchhoff F, Penesova A, Radikova Z, Lukac J et al (2012) Adrenal function in rheumatoid arthritis: a correlation with disease activity. Arthr Res Ther 14(Suppl 1):P27
Bunn SJ, Ait-Ali D, Eiden LE (2012) Immune-neuroendocrine integration at the adrenal gland: cytokine control of the adrenomedullary transcriptome. J Mol Neurosci 48(2):413–419
Douglas SA, Sreenivasan D, Carman FH, Bunn SJ (2010) Cytokine interactions with adrenal medullary chromaffin cells. Cell Mol Neurobiol 30(8):1467–1475
Aringer M, Feierl E, Smolen J (2008) Cytokine blockade-a promising therapeutic option in SLE. Z Rheumatol 67:315–317
Youinou P, Pers J (2011) Disturbance of cytokine networks in Sjogrens syndrome. Arthritis Res Ther 13:227
Burmester GR, Feist E, Dörner T (2013) Emerging cell and cytokine targets in rheumatoid arthritis. Nature Rev Rheumatol 10(2):77–88
Stephens MAC, Wand G (2012) Stress and the HPA axis: role of glucocorticoids in alcohol dependence. Alcohol Res Curr Rev 34(4):468–483
Hunter HJ, Griffiths CE, Kleyn CE (2013) Does psychosocial stress play a role in the exacerbation of psoriasis? Br J Dermatol 169(5):965–974
Späth-Schwalbe E, Born J, Schrezenmeier H, Bornstein SR, Stromeyer P, Drechsler S, Fehm HL, Porzsolt F (1994) Interleukin-6 stimulates the hypothalamus-pituitary-adrenocortical axis in man. J Clin Endocrinol Metab 79(4):1212–1214
Chesnokova V (2002) Minireview: neuro-immuno-endocrine modulation of the hypothalamic-pituitary-adrenal (HPA) axis by gp130 signaling molecules. Endocrinol 143(5):1571–1574
Larsen PJ, Jessop D, Patel H, Lightman SL, Chowdrey HS (1993) Substance P inhibits the release of anterior pituitary adrenocorticotrophin via a central mechanism involving corticotrophin-releasing factor-containing neurons in the hypothalamic paraventricular nucleus. J Neuroendocrinol 5(1):99–105
Dube SR, Fairweather D, Pearson WS, Felitti VJ, Anda RF, Croft JB (2009) Cumulative childhood stress and autoimmune diseases in adults. Psychosom Med 71(2):243–250
Stojanovich L (2010) Stress and autoimmunity. Autoimm Rev 9(5):A271–A276
Stojanovich L, Marisavljevich D (2008) Stress as a trigger of autoimmune disease. Autoimm Rev 7(3):209–213
Contributions
The authors contributed equally to the writing up of the paper and the electronic database searches. The authors certify that they have seen and approved the final version of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
We confirm that we have read the journal’s position on issues involved in ethical publication and affirm that this report is consistent with those guidelines.
Conflict of Interest
The authors do not report any conflict of interest.
Funding
There was no specific funding for this specific study.
Rights and permissions
About this article
Cite this article
Morris, G., Anderson, G. & Maes, M. Hypothalamic-Pituitary-Adrenal Hypofunction in Myalgic Encephalomyelitis (ME)/Chronic Fatigue Syndrome (CFS) as a Consequence of Activated Immune-Inflammatory and Oxidative and Nitrosative Pathways. Mol Neurobiol 54, 6806–6819 (2017). https://doi.org/10.1007/s12035-016-0170-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-016-0170-2