Abstract
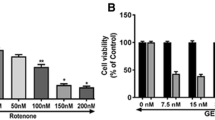
Oxidative stress causes cellular damage by (i) altering protein stability, (ii) impairing organelle function, or (iii) triggering the formation of 4-HNE protein aggregates. The catabolic process known as autophagy is an antioxidant cellular response aimed to counteract these stressful conditions. Therefore, autophagy might act as a cytoprotective response by removing impaired organelles and aggregated proteins. In the present study, we sought to understand the role of autophagy in the clearance of 4-HNE protein aggregates in ARPE-19 cells under rotenone exposure. Rotenone induced an overproduction of reactive oxygen species (ROS), which led to an accumulation of 4-HNE inclusions, and an increase in the number of autophagosomes. The latter resulted from a disturbed autophagic flux rather than an activation of the autophagic synthesis pathway. In compliance with this, rotenone treatment induced an increase in LC3-II while upstream autophagy markers such as Beclin- 1, Vsp34 or Atg5-Atg12, were decreased. Rotenone reduced the autophagosome-to-lysosome fusion step by increasing tubulin acetylation levels through a ROS-mediated pathway. Proof of this is the finding that the free radical scavenger, N-acetylcysteine, restored autophagy flux and reduced rotenone-induced tubulin hyperacetylation. Indeed, this dysfunctional autophagic response exacerbates cell death triggered by rotenone, since 3-methyladenine, an autophagy inhibitor, reduced cell mortality, while rapamycin, an inductor of autophagy, caused opposite effects. In summary, we shed new light on the mechanisms involved in the autophagic responses disrupted by oxidative stress, which take place in neurodegenerative diseases such as Huntington or Parkinson diseases, and age-related macular degeneration.







Similar content being viewed by others
Abbreviations
- 3-MA:
-
3-methyadenine
- 4-HNE:
-
4-hydroxynonenal
- ARPE-19:
-
Human Retinal Pigment Epithelial Cells
- CQ:
-
chloroquine
- DCF:
-
dichlorofluorescein
- GFP:
-
green fluorescent protein
- H2O2 :
-
hydrogen peroxide
- LC3:
-
microtubule-associated protein 1A/1B light chain 3
- NAC:
-
N-acetylcysteine
- PI:
-
propidium iodide
- RFP:
-
red fluorescent protein
- ROS:
-
reactive oxygen species
References
Geeraert C, Ratier A, Pfisterer SG, Perdiz D, Cantaloube I, Rouault A, Pattingre S, Proikas-Cezanne T et al (2010) Starvation-induced hyperacetylation of tubulin is required for the stimulation of autophagy by nutrient deprivation. J Biol Chem 285(31):24184–24194. doi:10.1074/jbc.M109.091553
Suzuki K, Ohsumi Y (2010) Current knowledge of the pre-autophagosomal structure (PAS). FEBS Lett 584(7):1280–1286. doi:10.1016/j.febslet.2010.02.001
Wirth M, Joachim J, Tooze SA (2013) Autophagosome formation--the role of ULK1 and Beclin1-PI3KC3 complexes in setting the stage. Semin Cancer Biol 23(5):301–309. doi:10.1016/j.semcancer.2013.05.007
Otomo C, Metlagel Z, Takaesu G, Otomo T (2012) Structure of the human ATG12 ~ ATG5 conjugate required for LC3 lipidation in autophagy. Nat Struct Mol Biol 20(1):59–66. doi:10.1038/nsmb.2431
Mackeh R, Perdiz D, Lorin S, Codogno P, Pous C (2013) Autophagy and microtubules—new story, old players. J Cell Sci 126(Pt 5):1071–1080. doi:10.1242/jcs.115626
Iwata A, Riley BE, Johnston JA, Kopito RR (2005) HDAC6 and microtubules are required for autophagic degradation of aggregated huntingtin. J Biol Chem 280(48):40282–40292. doi:10.1074/jbc.M508786200
Mackeh R, Lorin S, Ratier A, Mejdoubi-Charef N, Baillet A, Bruneel A, Hamai A, Codogno P et al (2014) Reactive oxygen species, AMP-activated protein kinase, and the transcription cofactor p300 regulate alpha-tubulin acetyltransferase-1 (alphaTAT-1/MEC-17)-dependent microtubule hyperacetylation during cell stress. J Biol Chem 289(17):11816–11828. doi:10.1074/jbc.M113.507400
Veldhoen RA, Banman SL, Hemmerling DR, Odsen R, Simmen T, Simmonds AJ, Underhill DA, Goping IS (2012) The chemotherapeutic agent paclitaxel inhibits autophagy through two distinct mechanisms that regulate apoptosis. Oncogene 32(6):736–746. doi:10.1038/onc.2012.92
Toops KA, Tan LX, Jiang Z, Radu RA, Lakkaraju A (2014) Cholesterol-mediated activation of acid sphingomyelinase disrupts autophagy in the retinal pigment epithelium. Mol Biol Cell 26(1):1–14. doi:10.1091/mbc.E14-05-1028
Kopito RR (2000) Aggresomes, inclusion bodies and protein aggregation. Trends Cell Biol 10(12):524–530
Miwa S, Brand MD (2005) The topology of superoxide production by complex III and glycerol 3-phosphate dehydrogenase in Drosophila mitochondria. Biochim Biophys Acta 1709(3):214–219. doi:10.1016/j.bbabio.2005.08.003
Kwong LK, Sohal RS (1998) Substrate and site specificity of hydrogen peroxide generation in mouse mitochondria. Arch Biochem Biophys 350(1):118–126. doi:10.1006/abbi.1997.0489
Leuner K, Schutt T, Kurz C, Eckert SH, Schiller C, Occhipinti A, Mai S, Jendrach M et al (2012) Mitochondrion-derived reactive oxygen species lead to enhanced amyloid beta formation. Antioxid Redox Signal 16(12):1421–1433. doi:10.1089/ars.2011.4173
Chu CT, Bayir H, Kagan VE (2013) LC3 binds externalized cardiolipin on injured mitochondria to signal mitophagy in neurons: implications for Parkinson disease. Autophagy 10(2):376–378. doi:10.4161/auto.27191
Sanders LH, Greenamyre JT (2013) Oxidative damage to macromolecules in human Parkinson disease and the rotenone model. Free Radic Biol Med 62:111–120. doi:10.1016/j.freeradbiomed.2013.01.003
Mitter SK, Song C, Qi X, Mao H, Rao H, Akin D, Lewin A, Grant M et al (2014) Dysregulated autophagy in the RPE is associated with increased susceptibility to oxidative stress and AMD. Autophagy 10(11):1989–2005. doi:10.4161/auto.36184
Lee SY, Oh JS, Rho JH, Jeong NY, Kwon YH, Jeong WJ, Ryu WY, Ahn HB et al. Retinal pigment epithelial cells undergoing mitotic catastrophe are vulnerable to autophagy inhibition. Cell Death Dis 5:e1303. doi:10.1038/cddis.2014.266
Beretta S, Wood JP, Derham B, Sala G, Tremolizzo L, Ferrarese C, Osborne NN (2006) Partial mitochondrial complex I inhibition induces oxidative damage and perturbs glutamate transport in primary retinal cultures. Relevance to Leber Hereditary Optic Neuropathy (LHON). Neurobiol Dis 24(2):308–317. doi:10.1016/j.nbd.2006.07.016
Chung WG, Miranda CL, Maier CS (2007) Epigallocatechin gallate (EGCG) potentiates the cytotoxicity of rotenone in neuroblastoma SH-SY5Y cells. Brain Res 1176:133–142. doi:10.1016/j.brainres.2007.07.083
Poli G, Schaur RJ, Siems WG, Leonarduzzi G (2008) 4-hydroxynonenal: a membrane lipid oxidation product of medicinal interest. Med Res Rev 28(4):569–631. doi:10.1002/med.20117
Butterfield DA, Lauderback CM (2002) Lipid peroxidation and protein oxidation in Alzheimer's disease brain: potential causes and consequences involving amyloid beta-peptide-associated free radical oxidative stress. Free Radic Biol Med 32(11):1050–1060
Yoritaka A, Hattori N, Uchida K, Tanaka M, Stadtman ER, Mizuno Y (1996) Immunohistochemical detection of 4-hydroxynonenal protein adducts in Parkinson disease. Proc Natl Acad Sci U S A 93(7):2696–2701
Nieva J, Shafton A, Altobell LJ 3rd, Tripuraneni S, Rogel JK, Wentworth AD, Lerner RA, Wentworth P Jr (2008) Lipid-derived aldehydes accelerate light chain amyloid and amorphous aggregation. Biochemistry 47(29):7695–7705. doi:10.1021/bi800333s
Wong ES, Tan JM, Soong WE, Hussein K, Nukina N, Dawson VL, Dawson TM, Cuervo AM et al (2008) Autophagy-mediated clearance of aggresomes is not a universal phenomenon. Hum Mol Genet 17(16):2570–2582. doi:10.1093/hmg/ddn157
Harris H, Rubinsztein DC (2011) Control of autophagy as a therapy for neurodegenerative disease. Nat Rev Neurol 8(2):108–117. doi:10.1038/nrneurol.2011.200
McCray BA, Taylor JP (2008) The role of autophagy in age-related neurodegeneration. Neurosignals 16(1):75–84. doi:10.1159/000109761
Palacino JJ, Sagi D, Goldberg MS, Krauss S, Motz C, Wacker M, Klose J, Shen J (2004) Mitochondrial dysfunction and oxidative damage in parkin-deficient mice. J Biol Chem 279(18):18614–18622. doi:10.1074/jbc
Manning-Bog AB, Caudle WM, Perez XA, Reaney SH, Paletzki R, Isla MZ, Chou VP, McCormack AL et al (2007) Increased vulnerability of nigrostriatal terminals in DJ-1-deficient mice is mediated by the dopamine transporter. Neurobiol Dis 27(2):141–150. doi:10.1016/j.nbd.2007.03.014
Kim RH, Smith PD, Aleyasin H, Hayley S, Mount MP, Pownall S, Wakeham A, You-Ten AJ et al (2005) Hypersensitivity of DJ-1-deficient mice to 1-methyl-4-phenyl-1,2,3,6-tetrahydropyrindine (MPTP) and oxidative stress. Proc Natl Acad Sci U S A 102(14):5215–5220. doi:10.1073/pnas.0501282102
Frank-Cannon TC, Tran T, Ruhn KA, Martinez TN, Hong J, Marvin M, Hartley M, Trevino I et al (2008) Parkin deficiency increases vulnerability to inflammation-related nigral degeneration. J Neurosci 28(43):10825–10834. doi:10.1523/JNEUROSCI.3001-08.2008
Bonet-Ponce L, Saez-Atienzar S, da Casa C, Flores-Bellver M, Barcia JM, Sancho-Pelluz J, Romero FJ, Jordan J et al (2015) On the mechanism underlying ethanol-induced mitochondrial dynamic disruption and autophagy response. Biochim Biophys Acta 1852(7):1400–1409. doi:10.1016/j.bbadis.2015.03.006
Ghislat G, Aguado C, Knecht E (2012) Annexin A5 stimulates autophagy and inhibits endocytosis. J Cell Sci 125(Pt 1):92–107. doi:10.1242/jcs.086728
Klionsky DJ, Abdalla FC, Abeliovich H, Abraham RT, Acevedo-Arozena A, Adeli K et al (2012) Guidelines for the use and interpretation of assays for monitoring autophagy. Autophagy 8(4):445–544
Saez-Atienzar S, Bonet-Ponce L, Blesa JR, Romero FJ, Murphy MP, Jordan J, Galindo MF (2014) The LRRK2 inhibitor GSK2578215A induces protective autophagy in SH-SY5Y cells: involvement of Drp-1-mediated mitochondrial fission and mitochondrial-derived ROS signaling. Cell Death Dis 5:e1368. doi:10.1038/cddis.2014.320
Ding WX, Li M, Chen X, Ni HM, Lin CW, Gao W, Lu B, Stolz DB et al (2010) Autophagy reduces acute ethanol-induced hepatotoxicity and steatosis in mice. Gastroenterology 139(5):1740–1752. doi:10.1053/j.gastro.2010.07.041
Wu F, Xu HD, Guan JJ, Hou YS, Gu JH, Zhen XC, Qin ZH (2014) Rotenone impairs autophagic flux and lysosomal functions in Parkinson's disease. Neuroscience 284:900–911. doi:10.1016/j.neuroscience.2014.11.004
Raben N, Shea L, Hill V, Plotz P (2009) Monitoring autophagy in lysosomal storage disorders. Methods Enzymol 453:417–449. doi:10.1016/S0076-6879(08)04021-4
Zhou J, Tan SH, Nicolas V, Bauvy C, Yang ND, Zhang J, Xue Y, Codogno P et al (2013) Activation of lysosomal function in the course of autophagy via mTORC1 suppression and autophagosome-lysosome fusion. Cell Res 23(4):508–523. doi:10.1038/cr.2013.11
Lee J, Kosaras B, Del Signore SJ, Cormier K, McKee A, Ratan RR, Kowall NW, Ryu H (2010) Modulation of lipid peroxidation and mitochondrial function improves neuropathology in Huntington's disease mice. Acta Neuropathol 121(4):487–498. doi:10.1007/s00401-010-0788-5
Shibata N, Inose Y, Toi S, Hiroi A, Yamamoto T, Kobayashi M (2010) Involvement of 4-hydroxy-2-nonenal accumulation in multiple system atrophy. Acta Histochem Cytochem 43(2):69–75. doi:10.1267/ahc.10005
Oyamada R, Hayashi M, Katoh Y, Tsuchiya K, Mizutani T, Tominaga I, Kashima H (2006) Neurofibrillary tangles and deposition of oxidative products in the brain in cases of myotonic dystrophy. Neuropathology 26(2):107–114
Sekigawa A, Fujita M, Sekiyama K, Takamatsu Y, Hatano T, Rockenstein E, La Spada AR, Masliah E et al (2012) Distinct mechanisms of axonal globule formation in mice expressing human wild type alpha-synuclein or dementia with Lewy bodies-linked P123H beta-synuclein. Mol Brain 5:34. doi:10.1186/1756-6606-5-34
Shimamoto K, Hayashi H, Taniai E, Morita R, Imaoka M, Ishii Y, Suzuki K, Shibutani M et al (2011) Antioxidant N-acetyl-L-cysteine (NAC) supplementation reduces reactive oxygen species (ROS)-mediated hepatocellular tumor promotion of indole-3-carbinol (I3C) in rats. J Toxicol Sci 36(6):775–786
Hambright HG, Meng P, Kumar AP, Ghosh R (2015) Inhibition of PI3K/AKT/mTOR axis disrupts oxidative stress-mediated survival of melanoma cells. Oncotarget 6(9):7195–7208
Kawaguchi Y, Kovacs JJ, McLaurin A, Vance JM, Ito A, Yao TP (2003) The deacetylase HDAC6 regulates aggresome formation and cell viability in response to misfolded protein stress. Cell 115(6):727–738
Yamamoto A, Simonsen A (2010) The elimination of accumulated and aggregated proteins: a role for aggrephagy in neurodegeneration. Neurobiol Dis 43(1):17–28. doi:10.1016/j.nbd.2010.08.015
Mizushima N, Yoshimori T (2007) How to interpret LC3 immunoblotting. Autophagy 3(6):542–545
Mizushima N, Yoshimori T, Levine B (2010) Methods in mammalian autophagy research. Cell 140(3):313–326. doi:10.1016/j.cell.2010.01.028
Baas PW, Ahmad FJ (2013) Beyond taxol: microtubule-based treatment of disease and injury of the nervous system. Brain 136(Pt 10):2937–2951. doi:10.1093/brain/awt153
Sanchez de Diego A, Alonso Guerrero A, Martinez AC, van Wely KH (2014) Dido3-dependent HDAC6 targeting controls cilium size. Nat Commun 5:3500. doi:10.1038/ncomms4500
Zuco V, De Cesare M, Cincinelli R, Nannei R, Pisano C, Zaffaroni N, Zunino F (2011) Synergistic antitumor effects of novel HDAC inhibitors and paclitaxel in vitro and in vivo. PLoS One 6(12):e29085. doi:10.1371/journal.pone
Marcus AI, Zhou J, O'Brate A, Hamel E, Wong J, Nivens M, El-Naggar A, Yao TP et al (2005) The synergistic combination of the farnesyl transferase inhibitor lonafarnib and paclitaxel enhances tubulin acetylation and requires a functional tubulin deacetylase. Cancer Res 65(9):3883–3893. doi:10.1158/0008-5472.CAN-04-3757
Dowdy SC, Jiang S, Zhou XC, Hou X, Jin F, Podratz KC, Jiang SW (2006) Histone deacetylase inhibitors and paclitaxel cause synergistic effects on apoptosis and microtubule stabilization in papillary serous endometrial cancer cells. Mol Cancer Ther 5(11):2767–2776. doi:10.1158/1535-7163.MCT-06-0209
Radad K, Rausch WD, Gille G (2006) Rotenone induces cell death in primary dopaminergic culture by increasing ROS production and inhibiting mitochondrial respiration. Neurochem Int 49(4):379–386. doi:10.1016/j.neuint.2006.02.003
Li N, Ragheb K, Lawler G, Sturgis J, Rajwa B, Melendez JA, Robinson JP (2003) Mitochondrial complex I inhibitor rotenone induces apoptosis through enhancing mitochondrial reactive oxygen species production. J Biol Chem 278(10):8516–8525. doi:10.1074/jbc.M210432200
Bulua AC, Simon A, Maddipati R, Pelletier M, Park H, Kim KY, Sack MN, Kastner DL et al (2011) Mitochondrial reactive oxygen species promote production of proinflammatory cytokines and are elevated in TNFR1-associated periodic syndrome (TRAPS). J Exp Med 208(3):519–533. doi:10.1084/jem.20102049
Lazaro-Dieguez F, Aguado C, Mato E, Sanchez-Ruiz Y, Esteban I, Alberch J, Knecht E, Egea G (2008) Dynamics of an F-actin aggresome generated by the actin-stabilizing toxin jasplakinolide. J Cell Sci 121(Pt 9):1415–1425. doi:10.1242/jcs.017665
Heath CM, Windsor M, Wileman T (2001) Aggresomes resemble sites specialized for virus assembly. J Cell Biol 153(3):449–455
Zaarur N, Meriin AB, Bejarano E, Xu X, Gabai VL, Cuervo AM, Sherman MY (2014) Proteasome failure promotes positioning of lysosomes around the aggresome via local block of microtubule-dependent transport. Mol Cell Biol 34(7):1336–1348. doi:10.1128/MCB.00103-14
Grune T, Jung T, Merker K, Davies KJ (2004) Decreased proteolysis caused by protein aggregates, inclusion bodies, plaques, lipofuscin, ceroid, and ‘aggresomes’ during oxidative stress, aging, and disease. Int J Biochem Cell Biol 36(12):2519–2530. doi:10.1016/j.biocel.2004.04.020S1357272504001670
Lin TK, Chen SD, Chuang YC, Lin HY, Huang CR, Chuang JH, Wang PW, Huang ST et al (2014) Resveratrol partially prevents rotenone-induced neurotoxicity in dopaminergic SH-SY5Y cells through induction of heme oxygenase-1 dependent autophagy. Int J Mol Sci 15(1):1625–1646. doi:10.3390/ijms15011625
Mader BJ, Pivtoraiko VN, Flippo HM, Klocke BJ, Roth KA, Mangieri LR, Shacka JJ (2012) Rotenone inhibits autophagic flux prior to inducing cell death. ACS Chem Neurosci 3(12):1063–1072. doi:10.1021/cn300145z
Wong E, Bejarano E, Rakshit M, Lee K, Hanson HH, Zaarur N, Phillips GR, Sherman MY et al (2012) Molecular determinants of selective clearance of protein inclusions by autophagy. Nat Commun 3:1240. doi:10.1038/ncomms2244
Liu S, Sarkar C, Dinizo M, Faden AI, Koh EY, Lipinski MM, Wu J (2015) Disrupted autophagy after spinal cord injury is associated with ER stress and neuronal cell death. Cell Death Dis 6:e1582. doi:10.1038/cddis.2014.527
Guan J, Mishra S, Qiu Y, Shi J, Trudeau K, Las G, Liesa M, Shirihai OS et al (2014) Lysosomal dysfunction and impaired autophagy underlie the pathogenesis of amyloidogenic light chain-mediated cardiotoxicity. EMBO Mol Med 6(11):1493–1507. doi:10.15252/emmm.201404190
Ma X, Liu H, Foyil SR, Godar RJ, Weinheimer CJ, Hill JA, Diwan A (2012) Impaired autophagosome clearance contributes to cardiomyocyte death in ischemia/reperfusion injury. Circulation 125(25):3170–3181. doi:10.1161/CIRCULATIONAHA.111.041814
Zhang X, Li L, Chen S, Yang D, Wang Y, Wang Z, Le W (2011) Rapamycin treatment augments motor neuron degeneration in SOD1(G93A) mouse model of amyotrophic lateral sclerosis. Autophagy 7(4):412–425
Giordano S, Darley-Usmar V, Zhang J (2014) Autophagy as an essential cellular antioxidant pathway in neurodegenerative disease. Redox Biol 2:82–90. doi:10.1016/j.redox.2013.12.013.S2213-2317(13)00101-8
Acknowledgments
We thank Carlos Garrido for his technical help and Dr E. Knecht for providing the mRFP-GFP-LC3 vector. This work was supported by a grant from the Ministerio de Ciencia e Innovación, Spain (SAF2010-21317), funds from the Universidad Católica de Valencia to FJR, and JCCM to JJ.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bonet-Ponce, L., Saez-Atienzar, S., da Casa, C. et al. Rotenone Induces the Formation of 4-Hydroxynonenal Aggresomes. Role of ROS-Mediated Tubulin Hyperacetylation and Autophagic Flux Disruption. Mol Neurobiol 53, 6194–6208 (2016). https://doi.org/10.1007/s12035-015-9509-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-015-9509-3