Abstract
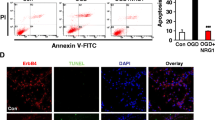
Ischemia is a condition associated with decreased blood supply to the brain, eventually leading to death of neurons. It is associated with a diverse cascade of responses involving both degenerative and regenerative mechanisms. At the cellular level, the changes are initiated prominently in the neuronal cytoskeleton. Cofilin, a cytoskeletal actin severing protein, is known to be involved in the early stages of apoptotic cell death. Evidence supports its intervention in the progression of disease states like Alzheimer’s and ischemic kidney disease. In the present study, we have hypothesized the possible involvement of cofilin in ischemia. Using PC12 cells and mouse primary cultures of cortical neurons, we investigated the potential role of cofilin in ischemia in two different in vitro ischemic models: chemical induced oxidative stress and oxygen-glucose deprivation/reperfusion (OGD/R). The expression profile studies demonstrated a decrease in phosphocofilin levels in all models of ischemia, implying stress-induced cofilin activation. Furthermore, calcineurin and slingshot 1L (SSH) phosphatases were found to be the signaling mediators of the cofilin activation. In primary cultures of cortical neurons, cofilin was found to be significantly activated after 1 h of OGD. To delineate the role of activated cofilin in ischemia, we knocked down cofilin by small interfering RNA (siRNA) technique and tested the impact of cofilin silencing on neuronal viability. Cofilin siRNA-treated neurons showed a significant reduction of cofilin levels in all treatment groups (control, OGD, and OGD/R). Additionally, cofilin siRNA-reduced cofilin mitochondrial translocation and caspase 3 cleavage, with a concomitant increase in neuronal viability. These results strongly support the active role of cofilin in ischemia-induced neuronal degeneration and apoptosis. We believe that targeting this protein mediator has a potential for therapeutic intervention in ischemic brain injury and stroke.







Similar content being viewed by others
References
Tissue plasminogen activator for acute ischemic stroke. The National Institute of Neurological Disorders and Stroke rt-PA Stroke Study Group (1995) N Engl J Med 333(24):1581–1587. doi:10.1056/NEJM199512143332401
Fuchs E, Cleveland DW (1998) A structural scaffolding of intermediate filaments in health and disease. Science 279(5350):514–519. doi:10.1126/science.279.5350.514
Bernstein BW, Bamburg JR (2010) ADF/cofilin: a functional node in cell biology. Trends Cell Biol 20(4):187–195. doi:10.1016/j.tcb.2010.01.001
Dos Remedios CG, Chhabra D, Kekic M, Dedova IV, Tsubakihara M, Berry DA, Nosworthy NJ (2003) Actin binding proteins: regulation of cytoskeletal microfilaments. Physiol Rev 83(2):433–473. doi:10.1152/physrev.00026.2002
Suurna MV, Ashworth SL, Hosford M, Sandoval RM, Wean SE, Shah BM, Bamburg JR, Molitoris BA (2006) Cofilin mediates ATP depletion-induced endothelial cell actin alterations. Am J Physiol Renal Physiol 290(6):F1398–1407. doi:10.1152/ajprenal.00194.2005
Bamburg JR, Wiggan OP (2002) ADF/cofilin and actin dynamics in disease. Trends Cell Biol 12(12):598–605
Minamide LS, Striegl AM, Boyle JA, Meberg PJ, Bamburg JR (2000) Neurodegenerative stimuli induce persistent ADF/cofilin-actin rods that disrupt distal neurite function. Nat Cell Biol 2(9):628–636
Klamt F, Zdanov S, Levine RL, Pariser A, Zhang Y, Zhang B, Yu LR, Veenstra TD, Shacter E (2009) Oxidant-induced apoptosis is mediated by oxidation of the actin-regulatory protein cofilin. Nat Cell Biol 11(10):1241–1246. doi:10.1038/ncb1968
Nada SE, Shah ZA (2012) Preconditioning with Ginkgo biloba (EGb 761(R)) provides neuroprotection through HO1 and CRMP2. Neurobiol Dis 46(1):180–189. doi:10.1016/j.nbd.2012.01.006
Olechnowicz SW, Fedele AO, Peet DJ (2012) Hypoxic induction of the regulator of G-protein signalling 4 gene is mediated by the hypoxia-inducible factor pathway. PLoS ONE 7(9):e44564. doi:10.1371/journal.pone.0044564PONE-D-12-18040
Nada SE, Tulsulkar J, Raghavan A, Hensley K, Shah ZA (2012) A derivative of the CRMP2 binding compound lanthionine ketimine provides neuroprotection in a mouse model of cerebral ischemia. Neurochem Int 61(8):1357–1363. doi:10.1016/j.neuint.2012.09.013
Dimauro I, Pearson T, Caporossi D, Jackson MJ (2012) A simple protocol for the subcellular fractionation of skeletal muscle cells and tissue. BMC Res Notes 5:513. doi:10.1186/1756-0500-5-513
Lemasters JJ, Theruvath TP, Zhong Z, Nieminen AL (2009) Mitochondrial calcium and the permeability transition in cell death. Biochim Biophys Acta 1787(11):1395–1401. doi:10.1016/j.bbabio.2009.06.009
Wang Y, Shibasaki F, Mizuno K (2005) Calcium signal-induced cofilin dephosphorylation is mediated by Slingshot via calcineurin. J Biol Chem 280(13):12683–12689. doi:10.1074/jbc.M411494200
Chua BT, Volbracht C, Tan KO, Li R, Yu VC, Li P (2003) Mitochondrial translocation of cofilin is an early step in apoptosis induction. Nat Cell Biol 5(12):1083–1089. doi:10.1038/ncb1070ncb1070
Posadas I, Perez-Martinez FC, Guerra J, Sanchez-Verdu P, Cena V (2012) Cofilin activation mediates Bax translocation to mitochondria during excitotoxic neuronal death. J Neurochem 120(4):515–527. doi:10.1111/j.1471-4159.2011.07599.x
Huang TY, Minamide LS, Bamburg JR, Bokoch GM (2008) Chronophin mediates an ATP-sensing mechanism for cofilin dephosphorylation and neuronal cofilin-actin rod formation. Dev Cell 15(5):691–703. doi:10.1016/j.devcel.2008.09.017
Cichon J, Sun C, Chen B, Jiang M, Chen XA, Sun Y, Wang Y, Chen G (2012) Cofilin aggregation blocks intracellular trafficking and induces synaptic loss in hippocampal neurons. J Biol Chem 287(6):3919–3929. doi:10.1074/jbc.M111.301911
Gurniak CB, Perlas E, Witke W (2005) The actin depolymerizing factor n-cofilin is essential for neural tube morphogenesis and neural crest cell migration. Dev Biol 278(1):231–241. doi:10.1016/j.ydbio.2004.11.010
Bellenchi GC, Gurniak CB, Perlas E, Middei S, Ammassari-Teule M, Witke W (2007) N-cofilin is associated with neuronal migration disorders and cell cycle control in the cerebral cortex. Genes Dev 21(18):2347–2357. doi:10.1101/gad.434307
Takahashi M, Tsujioka Y, Yamada T, Tsuboi Y, Okada H, Yamamoto T, Liposits Z (1999) Glycosylation of microtubule-associated protein tau in Alzheimer’s disease brain. Acta Neuropathol 97(6):635–641
Lipton SA (1999) Neuronal protection and destruction by NO. Cell Death Differ 6(10):943–951. doi:10.1038/sj.cdd.4400580
Schwartz N, Hosford M, Sandoval RM, Wagner MC, Atkinson SJ, Bamburg J, Molitoris BA (1999) Ischemia activates actin depolymerizing factor: role in proximal tubule microvillar actin alterations. Am J Physiol 276(4 Pt 2):F544–551
Woo JA, Jung AR, Lakshmana MK, Bedrossian A, Lim Y, Bu JH, Park SA, Koo EH, Mook-Jung I, Kang DE (2012) Pivotal role of the RanBP9-cofilin pathway in Abeta-induced apoptosis and neurodegeneration. Cell Death Differ 19(9):1413–1423. doi:10.1038/cdd.2012.14
Lim D, Iyer A, Ronco V, Grolla AA, Canonico PL, Aronica E, Genazzani AA (2013) Amyloid beta deregulates astroglial mGluR5-mediated calcium signaling via calcineurin and Nf-kB. Glia 61(7):1134–1145. doi:10.1002/glia.22502
Kim JE, Ryu HJ, Kim MJ, Kim DW, Kwon OS, Choi SY, Kang TC (2010) Pyridoxal-5′-phosphate phosphatase/chronophin induces astroglial apoptosis via actin-depolymerizing factor/cofilin system in the rat brain following status epilepticus. Glia 58(16):1937–1948. doi:10.1002/glia.21063
Rasmussen I, Pedersen LH, Byg L, Suzuki K, Sumimoto H, Vilhardt F (2010) Effects of F/G-actin ratio and actin turn-over rate on NADPH oxidase activity in microglia. BMC Immunol 11:44. doi:10.1186/1471-2172-11-44
Hadas S, Spira M, Hanisch UK, Reichert F, Rotshenker S (2012) Complement receptor-3 negatively regulates the phagocytosis of degenerated myelin through tyrosine kinase Syk and cofilin. J Neuroinflammation 9:166. doi:10.1186/1742-2094-9-166
Gitik M, Kleinhaus R, Hadas S, Reichert F, Rotshenker S (2014) Phagocytic receptors activate and immune inhibitory receptor SIRPalpha inhibits phagocytosis through paxillin and cofilin. Front Cell Neurosci 8:104. doi:10.3389/fncel.2014.00104
Acknowledgments
The study was partly funded by a grant from NIH (R00AT004197) and start-up funds from The University of Toledo to ZAS. Qasim Alhadidi was supported by Higher Committee for Education Development in Iraq (www.hcediraq.org). The authors would like to thank Charisse N. Montgomery for her assistance in the manuscript editing.
Author information
Authors and Affiliations
Corresponding author
Additional information
A. Madineni and Q. Alhadidi contributed equally.
Rights and permissions
About this article
Cite this article
Madineni, A., Alhadidi, Q. & Shah, Z.A. Cofilin Inhibition Restores Neuronal Cell Death in Oxygen–Glucose Deprivation Model of Ischemia. Mol Neurobiol 53, 867–878 (2016). https://doi.org/10.1007/s12035-014-9056-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-014-9056-3