Abstract
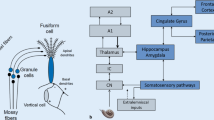
Increasing evidence shows that hearing loss is a risk factor for tinnitus and hyperacusis. Although both often coincide, a causal relationship between tinnitus and hyperacusis has not been shown. Currently, tinnitus and hyperacusis are assumed to be caused by elevated responsiveness in subcortical circuits. We examined both the impact of different degrees of cochlear damage and the influence of stress priming on tinnitus induction. We used (1) a behavioral animal model for tinnitus designed to minimize stress, (2) ribbon synapses in inner hair cells (IHCs) as a measure for deafferentation, (3) the integrity of auditory brainstem responses (ABR) to detect differences in stimulus-evoked neuronal activity, (4) the expression of the activity-regulated cytoskeletal protein, Arc, to identify long-lasting changes in network activity within the basolateral amygdala (BLA), hippocampal CA1, and auditory cortex (AC), and (5) stress priming to investigate the influence of corticosteroid on trauma-induced brain responses. We observed that IHC ribbon loss (deafferentation) leads to tinnitus when ABR functions remain reduced and Arc is not mobilized in the hippocampal CA1 and AC. If, however, ABR waves are functionally restored and Arc is mobilized, tinnitus does not occur. Both central response patterns were found to be independent of a profound threshold loss and could be shifted by the corticosterone level at the time of trauma. We, therefore, discuss the findings in the context of a history of stress that can trigger either an adaptive or nonadaptive brain response following injury.







Similar content being viewed by others
References
Lockwood AH, Salvi RJ, Burkard RF (2002) Tinnitus. N Engl J Med 347(12):904–910
Møller AR (2003) Pathophysiology of tinnitus. Otolaryngol Clin North Am 36(2):249–266, v-vi
Jastreboff PJ (2007) Tinnitus retraining therapy. Prog Brain Res 166:415–423
Zenner HP, Pfister M, Birbaumer N (2006) Tinnitus sensitization: sensory and psychophysiological aspects of a new pathway of acquired centralization of chronic tinnitus. Otol Neurotol 27(8):1054–1063
Puel JL, Guitton MJ (2007) Salicylate-induced tinnitus: molecular mechanisms and modulation by anxiety. Prog Brain Res 166:141–146
Leaver AM, Renier L, Chevillet MA, Morgan S, Kim HJ, Rauschecker JP (2011) Dysregulation of limbic and auditory networks in tinnitus. Neuron 69(1):33–43. doi:10.1016/j.neuron.2010.12.002
Meltser I, Tahera Y, Canlon B (2009) Glucocorticoid receptor and mitogen-activated protein kinases activity after restraint stress and acoustic trauma. J Neurotrauma 26(10):1835–1845
Langguth B, Salvi R, Elgoyhen AB (2009) Emerging pharmacotherapy of tinnitus. Expert Opin Emerg Drugs 14(4):687–702
Kim DK, Park SN, Kim HM, Son HR, Kim NG, Park KH, Yeo SW (2011) Prevalence and significance of high-frequency hearing loss in subjectively normal-hearing patients with tinnitus. Ann Otol Rhinol Laryngol 120(8):523–528
Roberts LE, Eggermont JJ, Caspary DM, Shore SE, Melcher JR, Kaltenbach JA (2010) Ringing ears: the neuroscience of tinnitus. J Neurosci 30(45):14972–14979. doi:10.1523/JNEUROSCI.4028-10.2010
Weisz N, Hartmann T, Dohrmann K, Schlee W, Noreña A (2006) High-frequency tinnitus without hearing loss does not mean absence of deafferentation. Hear Res 222(1–2):108–114
Geven LI, de Kleine E, Free RH, van Dijk P (2011) Contralateral suppression of otoacoustic emissions in tinnitus patients. Otol Neurotol 32(2):315–321. doi:10.1097/MAO.0b013e3181fcf180
Bauer CA, Brozoski TJ, Myers K (2007) Primary afferent dendrite degeneration as a cause of tinnitus. J Neurosci Res 85(7):1489–1498
Knipper M, Müller M, Zimmermann U (2012) Molecular mechanism of tinnitus. In: Fay RR, Popper AN, Eggermont JJ (eds) Springer handbook of auditory research: neural correlates of tinnitus. Springer, New York
Knipper M, Zimmermann U, Müller M (2010) Molecular aspects of tinnitus. Hear Res 266(1–2):60–69. doi:10.1016/j.heares.2009.07.013
Baguley DM (2003) Hyperacusis. J R Soc Med 96(12):582–585
Gu JW, Halpin CF, Nam EC, Levine RA, Melcher JR (2010) Tinnitus, diminished sound-level tolerance, and elevated auditory activity in humans with clinically normal hearing sensitivity. J Neurophysiol 104(6):3361–3370. doi:10.1152/jn.00226.2010
Kujawa SG, Liberman MC (2009) Adding insult to injury: cochlear nerve degeneration after “temporary” noise-induced hearing loss. J Neurosci 29(45):14077–14085
Rauschecker JP, Leaver AM, Mühlau M (2010) Tuning out the noise: limbic–auditory interactions in tinnitus. Neuron 66(6):819–826. doi:10.1016/j.neuron.2010.04.032
Salvi RJ, Wang J, Ding D (2000) Auditory plasticity and hyperactivity following cochlear damage. Hear Res 147(1–2):261–274
Brozoski TJ, Bauer CA, Caspary DM (2002) Elevated fusiform cell activity in the dorsal cochlear nucleus of chinchillas with psychophysical evidence of tinnitus. J Neurosci 22(6):2383–2390
Kaltenbach JA (2007) The dorsal cochlear nucleus as a contributor to tinnitus: mechanisms underlying the induction of hyperactivity. Prog Brain Res 166:89–106
Middleton JW, Kiritani T, Pedersen C, Turner JG, Shepherd GM, Tzounopoulos T (2011) Mice with behavioral evidence of tinnitus exhibit dorsal cochlear nucleus hyperactivity because of decreased GABAergic inhibition. Proc Natl Acad Sci USA 108(18):7601–7606. doi:10.1073/pnas.1100223108
Yang S, Weiner BD, Zhang LS, Cho SJ, Bao S (2011) Homeostatic plasticity drives tinnitus perception in an animal model. Proc Natl Acad Sci USA 108(36):14974–14979. doi:10.1073/pnas.1107998108
Dehmel S, Pradhan S, Koehler S, Bledsoe S, Shore S (2012) Noise overexposure alters long-term somatosensory–auditory processing in the dorsal cochlear nucleus—possible basis for tinnitus-related hyperactivity? J Neurosci 32(5):1660–1671. doi:10.1523/JNEUROSCI.4608-11.2012
Schaette R, Kempter R (2012) Computational models of neurophysiological correlates of tinnitus. Front Syst Neurosci 6:34. doi:10.3389/fnsys.2012.00034
Cai S, Ma WL, Young ED (2009) Encoding intensity in ventral cochlear nucleus following acoustic trauma: implications for loudness recruitment. J Assoc Res Otolaryngol 10(1):5–22. doi:10.1007/s10162-008-0142-y
Szczepaniak WS, Møller AR (1996) Evidence of neuronal plasticity within the inferior colliculus after noise exposure: a study of evoked potentials in the rat. Electroencephalogr Clin Neurophysiol 100(2):158–164
Turner JG (2007) Behavioral measures of tinnitus in laboratory animals. Prog Brain Res 166:147–156
Rüttiger L, Ciuffani J, Zenner HP, Knipper M (2003) A behavioral paradigm to judge acute sodium salicylate-induced sound experience in rats: a new approach for an animal model on tinnitus. Hear Res 180(1–2):39–50
Tan J, Rüttiger L, Panford-Walsh R, Singer W, Schulze H, Kilian SB, Hadjab S, Zimmermann U, Köpschall I, Rohbock K, Knipper M (2007) Tinnitus behavior and hearing function correlate with the reciprocal expression patterns of BDNF and Arg3.1/arc in auditory neurons following acoustic trauma. Neuroscience 145(2):715–726
Lin HW, Furman AC, Kujawa SG, Liberman MC (2011) Primary neural degeneration in the guinea pig cochlea after reversible noise-induced threshold shift. J Assoc Res Otolaryngol 12(5):605–616. doi:10.1007/s10162-011-0277-0
Melcher JR, Kiang NY (1996) Generators of the brainstem auditory evoked potential in cat. III: identified cell populations. Hear Res 93(1–2):52–71
Zuccotti A, Kuhn S, Johnson SL, Franz C, Singer W, Hecker D, Geisler HS, Köpschall I, Rohbock K, Gutsche K, Dlugaiczyk J, Schick B, Marcotti W, Rüttiger L, Schimmang T, Knipper M (2012) Lack of brain-derived neurotrophic factor hampers inner hair cell synapse physiology, but protects against noise induced hearing loss. J Neurosci 32:8545–8553
Bramham CR, Alme MN, Bittins M, Kuipers SD, Nair RR, Pai B, Panja D, Schubert M, Soule J, Tiron A, Wibrand K (2010) The Arc of synaptic memory. Exp Brain Res 200(2):125–140. doi:10.1007/s00221-009-1959-2
Gao M, Sossa K, Song L, Errington L, Cummings L, Hwang H, Kuhl D, Worley P, Lee HK (2010) A specific requirement of Arc/Arg3.1 for visual experience-induced homeostatic synaptic plasticity in mouse primary visual cortex. J Neurosci 30(21):7168–7178. doi:10.1523/JNEUROSCI.1067-10.2010
Korb E, Finkbeiner S (2012) Arc in synaptic plasticity: from gene to behavior. Trends Neurosci 34(11):591–598. doi:10.1016/j.tins.2011.08.007[doi]
Kozlovsky N, Matar MA, Kaplan Z, Kotler M, Zohar J, Cohen H (2008) The immediate early gene Arc is associated with behavioral resilience to stress exposure in an animal model of posttraumatic stress disorder. Eur Neuropsychopharmacol 18(2):107–116. doi:10.1016/j.euroneuro.2007.04.009
Ons S, Rotllant D, Marin-Blasco IJ, Armario A (2010) Immediate–early gene response to repeated immobilization: Fos protein and arc mRNA levels appear to be less sensitive than c-fos mRNA to adaptation. Eur J Neurosci 31(11):2043–2052. doi:10.1111/j.1460-9568.2010.07242.x
Turner BB (1986) Tissue differences in the up-regulation of glucocorticoid-binding proteins in the rat. Endocrinology 118(3):1211–1216
Chavez CM, McGaugh JL, Weinberger NM (2009) The basolateral amygdala modulates specific sensory memory representations in the cerebral cortex. Neurobiol Learn Mem 91(4):382–392. doi:10.1016/j.nlm.2008.10.010
Knipper M, Zinn C, Maier H, Praetorius M, Rohbock K, Köpschall I, Zimmermann U (2000) Thyroid hormone deficiency before the onset of hearing causes irreversible damage to peripheral and central auditory systems. J Neurophysiol 83(5):3101–3112
Schimmang T, Tan J, Müller M, Zimmermann U, Rohbock K, Köpschall I, Limberger A, Minichiello L, Knipper M (2003) Lack of Bdnf and TrkB signalling in the postnatal cochlea leads to a spatial reshaping of innervation along the tonotopic axis and hearing loss. Development 130(19):4741–4750
Engel J, Braig C, Rüttiger L, Kuhn S, Zimmermann U, Blin N, Sausbier M, Kalbacher H, Münkner S, Rohbock K, Ruth P, Winter H, Knipper M (2006) Two classes of outer hair cells along the tonotopic axis of the cochlea. Neuroscience 143:837–849
Blanchard RJ, McKittrick CR, Blanchard DC (2001) Animal models of social stress: effects on behavior and brain neurochemical systems. Physiol Behav 73(3):261–271
Haller J, Fuchs E, Halasz J, Makara GB (1999) Defeat is a major stressor in males while social instability is stressful mainly in females: towards the development of a social stress model in female rats. Brain Res Bull 50(1):33–39
Heidrych P, Zimmermann U, Kuhn S, Franz C, Engel J, Duncker SV, Hirt B, Pusch CM, Ruth P, Pfister M, Marcotti W, Blin N, Knipper M (2009) Otoferlin interacts with myosin VI: implications for maintenance of the basolateral synaptic structure of the inner hair cell. Hum Mol Genet 18:2779–2790
Panford-Walsh R, Singer W, Rüttiger L, Hadjab S, Tan J, Geisler HS, Zimmermann U, Köpschall I, Rohbock K, Vieljans A, Oestreicher E, Knipper M (2008) Midazolam reverses salicylate-induced changes in brain-derived neurotrophic factor and arg3.1 expression: implications for tinnitus perception and auditory plasticity. Mol Pharmacol 74(3):595–604. doi:10.1124/mol.108.046375
Ramirez-Amaya V, Vazdarjanova A, Mikhael D, Rosi S, Worley PF, Barnes CA (2005) Spatial exploration-induced Arc mRNA and protein expression: evidence for selective, network-specific reactivation. J Neurosci 25(7):1761–1768
Müller M (1991) Frequency representation in the rat cochlea. Hear Res 51(2):247–254
Doron NN, Ledoux JE, Semple MN (2002) Redefining the tonotopic core of rat auditory cortex: physiological evidence for a posterior field. J Comp Neurol 453(4):345–360
Sarro EC, Kotak VC, Sanes DH, Aoki C (2008) Hearing loss alters the subcellular distribution of presynaptic GAD and postsynaptic GABAA receptors in the auditory cortex. Cereb Cortex 18(12):2855–2867. doi:10.1093/cercor/bhn044
Yilmaz-Rastoder E, Miyamae T, Braun AE, Thiels E (2011) LTP- and LTD-inducing stimulations cause opposite changes in arc/arg3.1 mRNA level in hippocampal area CA1 in vivo. Hippocampus 21(12):1290–1301. doi:10.1002/hipo.20838
Wang H, Brozoski TJ, Turner JG, Ling L, Parrish JL, Hughes LF, Caspary DM (2009) Plasticity at glycinergic synapses in dorsal cochlear nucleus of rats with behavioral evidence of tinnitus. Neuroscience 164(2):747–759. doi:10.1016/j.neuroscience.2009.08.026
Vogler DP, Robertson D, Mulders WH (2011) Hyperactivity in the ventral cochlear nucleus after cochlear trauma. J Neurosci 31(18):6639–6645. doi:10.1523/JNEUROSCI.6538-10.2011
Glowatzki E, Fuchs PA (2002) Transmitter release at the hair cell ribbon synapse. Nat Neurosci 5(2):147–154
Buran BN, Strenzke N, Neef A, Gundelfinger ED, Moser T, Liberman MC (2010) Onset coding is degraded in auditory nerve fibers from mutant mice lacking synaptic ribbons. J Neurosci 30(22):7587–7597. doi:10.1523/JNEUROSCI.0389-10.2010
Johnson DH, Kiang NY (1976) Analysis of discharges recorded simultaneously from pairs of auditory nerve fibers. Biophys J 16(7):719–734
Schaette R, Kempter R (2009) Predicting tinnitus pitch from patients’ audiograms with a computational model for the development of neuronal hyperactivity. J Neurophysiol 101(6):3042–3052
Qiu C, Salvi R, Ding D, Burkard R (2000) Inner hair cell loss leads to enhanced response amplitudes in auditory cortex of unanesthetized chinchillas: evidence for increased system gain. Hear Res 139(1–2):153–171
Zeng FG (2012) An active loudness model suggesting tinnitus as increased central noise and hyperacusis as increased nonlinear gain. Hear Res. doi:10.1016/j.heares.2012.05.009
Schaette R, McAlpine D (2011) Tinnitus with a normal audiogram: physiological evidence for hidden hearing loss and computational model. J Neurosci 31(38):13452–13457. doi:10.1523/JNEUROSCI.2156-11.2011
Malmierca M, Merchan M (2004) The auditory system. In: Paxinos G (ed) The rat nervous system. Academic, San Diego, pp 997–1082
Shepherd JD, Rumbaugh G, Wu J, Chowdhury S, Plath N, Kuhl D, Huganir RL, Worley PF (2006) Arc/Arg3.1 mediates homeostatic synaptic scaling of AMPA receptors. Neuron 52(3):475–484
Guzowski JF, McNaughton BL, Barnes CA, Worley PF (1999) Environment-specific expression of the immediate–early gene Arc in hippocampal neuronal ensembles. Nat Neurosci 2(12):1120–1124
Pinaud R, Penner MR, Robertson HA, Currie RW (2001) Upregulation of the immediate early gene arc in the brains of rats exposed to environmental enrichment: implications for molecular plasticity. Brain Res Mol Brain Res 91(1–2):50–56
Beique JC, Na Y, Kuhl D, Worley PF, Huganir RL (2010) Arc-dependent synapse-specific homeostatic plasticity. Proc Natl Acad Sci U S A 108(2):816–821. doi:10.1073/pnas.1017914108
Kotak VC, Fujisawa S, Lee FA, Karthikeyan O, Aoki C, Sanes DH (2005) Hearing loss raises excitability in the auditory cortex. J Neurosci 25(15):3908–3918
Peebles CL, Yoo J, Thwin MT, Palop JJ, Noebels JL, Finkbeiner S (2010) Arc regulates spine morphology and maintains network stability in vivo. Proc Natl Acad Sci U S A 107(42):18173–18178. doi:10.1073/pnas.1006546107
Eggermont JJ, Roberts LE (2004) The neuroscience of tinnitus. Trends Neurosci 27(11):676–682
Noreña AJ, Tomita M, Eggermont JJ (2003) Neural changes in cat auditory cortex after a transient pure-tone trauma. J Neurophysiol 90(4):2387–2401
Schecklmann M, Vielsmeier V, Steffens T, Landgrebe M, Langguth B, Kleinjung T (2012) Relationship between audiometric slope and tinnitus pitch in tinnitus patients: insights into the mechanisms of tinnitus generation. PLoS One 7(4):e34878. doi:10.1371/journal.pone.0034878
Mangiamele LA, Thomson CJ, Lebonville CL, Burmeister SS (2010) Characterization of the plasticity-related gene, Arc, in the frog brain. Dev Neurobiol 70(12):813–825. doi:10.1002/dneu.20817
Hawley ML, Melcher JR, Fullerton BC (2005) Effects of sound bandwidth on fMRI activation in human auditory brainstem nuclei. Hear Res 204(1–2):101–110. doi:10.1016/j.heares.2005.01.005
Rajan R (1998) Receptor organ damage causes loss of cortical surround inhibition without topographic map plasticity. Nat Neurosci 1(2):138–143. doi:10.1038/388
Diesch E, Struve M, Rupp A, Ritter S, Hulse M, Flor H (2004) Enhancement of steady-state auditory evoked magnetic fields in tinnitus. Eur J Neurosci 19(4):1093–1104
Langers DR, de Kleine E, van Dijk P (2012) Tinnitus does not require macroscopic tonotopic map reorganization. Front Syst Neurosci 6:2. doi:10.3389/fnsys.2012.00002
Campeau S, Watson SJ (1997) Neuroendocrine and behavioral responses and brain pattern of c-fos induction associated with audiogenic stress. J Neuroendocrinol 9(8):577–588
Karst H, Berger S, Erdmann G, Schutz G, Joëls M (2010) Metaplasticity of amygdalar responses to the stress hormone corticosterone. Proc Natl Acad Sci U S A 107(32):14449–14454. doi:10.1073/pnas.0914381107
Tasker JG, Di S, Malcher-Lopes R (2006) Minireview: rapid glucocorticoid signaling via membrane-associated receptors. Endocrinology 147(12):5549–5556. doi:10.1210/en.2006-0981
Groeneweg FL, Karst H, de Kloet ER, Joëls M (2011) Rapid non-genomic effects of corticosteroids and their role in the central stress response. J Endocrinol 209(2):153–167. doi:10.1530/JOE-10-0472
Richter-Levin G, Maroun M (2010) Stress and amygdala suppression of metaplasticity in the medial prefrontal cortex. Cereb Cortex 20(10):2433–2441. doi:10.1093/cercor/bhp311
Kozlovsky N, Matar MA, Kaplan Z, Zohar J, Cohen H (2009) A distinct pattern of intracellular glucocorticoid-related responses is associated with extreme behavioral response to stress in an animal model of post-traumatic stress disorder. Eur Neuropsychopharmacol 19(11):759–771. doi:10.1016/j.euroneuro.2009.04.009
Abraham WC, Bear MF (1996) Metaplasticity: the plasticity of synaptic plasticity. Trends Neurosci 19(4):126–130
Wang Y, Liberman MC (2002) Restraint stress and protection from acoustic injury in mice. Hear Res 165(1–2):96–102
Terakado M, Kumagami H, Takahashi H (2011) Distribution of glucocorticoid receptors and 11 beta-hydroxysteroid dehydrogenase isoforms in the rat inner ear. Hear Res 280(1–2):148–156. doi:10.1016/j.heares.2011.05.006
Yao X, Rarey KE (1996) Localization of the mineralocorticoid receptor in rat cochlear tissue. Acta Otolaryngol 116(3):493–496
Oitzl MS, Champagne DL, van der Veen R, de Kloet ER (2010) Brain development under stress: hypotheses of glucocorticoid actions revisited. Neurosci Biobehav Rev 34(6):853–866. doi:10.1016/j.neubiorev.2009.07.006
Knapman A, Kaltwasser SF, Martins-de-Souza D, Holsboer F, Landgraf R, Turck CW, Czisch M, Touma C (2012) Increased stress reactivity is associated with reduced hippocampal activity and neuronal integrity along with changes in energy metabolism. Eur J Neurosci 35(3):412–422. doi:10.1111/j.1460-9568.2011.07968.x
Hebert S, Lupien SJ (2007) The sound of stress: blunted cortisol reactivity to psychosocial stress in tinnitus sufferers. Neurosci Lett 411(2):138–142
Eriksson TM, Delagrange P, Spedding M, Popoli M, Mathe AA, Ogren SO, Svenningsson P (2012) Emotional memory impairments in a genetic rat model of depression: involvement of 5-HT/MEK/Arc signaling in restoration. Mol Psychiatry 17(2):173–184. doi:10.1038/mp.2010.131
Palop JJ, Chin J, Bien-Ly N, Massaro C, Yeung BZ, Yu GQ, Mucke L (2005) Vulnerability of dentate granule cells to disruption of arc expression in human amyloid precursor protein transgenic mice. J Neurosci 25(42):9686–9693
Wegenast-Braun BM, Fulgencio Maisch A, Eicke D, Radde R, Herzig MC, Staufenbiel M, Jucker M, Calhoun ME (2009) Independent effects of intra- and extracellular Abeta on learning-related gene expression. Am J Pathol 175(1):271–282. doi:10.2353/ajpath.2009.090044
Goel A, Lee HK (2007) Persistence of experience-induced homeostatic synaptic plasticity through adulthood in superficial layers of mouse visual cortex. J Neurosci 27(25):6692–6700. doi:10.1523/JNEUROSCI.5038-06.2007
Acknowledgments
This work was supported by the Marie Curie Research Training Network CavNET MRTN-CT-2006-035367, Deutsche Forschungsgemeinschaft DFG-Kni-316-4-1, and Hahn Stiftung (Index AG).
Conflict of Interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Wibke Singer and Annalisa Zuccotti equally contributed to this work.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 81 kb)
Rights and permissions
About this article
Cite this article
Singer, W., Zuccotti, A., Jaumann, M. et al. Noise-Induced Inner Hair Cell Ribbon Loss Disturbs Central Arc Mobilization: A Novel Molecular Paradigm for Understanding Tinnitus. Mol Neurobiol 47, 261–279 (2013). https://doi.org/10.1007/s12035-012-8372-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-012-8372-8