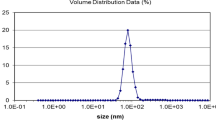
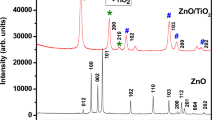
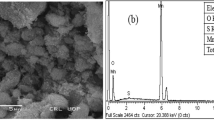
Abstract
Malachite green (MG) is a synthetic cationic dye extensively utilized in the textile industry for colouring purposes. It is a mutagenic agent and potentially carcinogenic for living organisms in nature. Therefore, it is necessary to treat and discharge the industrial effluents covering MG to avoid irreversible and serious effects on both living organisms and the environment. The photocatalyst SnO2 was synthesized by a new method by complexation of sodium diethyldithiocarbamate with tin dichloride pentahydrate, followed by calcination at 450, 600 and 750°C and utilized for photocatalytic degradation of MG dye. The better degradation was obtained at calcination temperature 600°C. The fluorescent light exposure resulted better degradation % of MG dye within 20 min, and the absorbance measurements were noted after every 5 min. The degradation of MG dye improved with an increase in the dose of SnO2 photocatalyst up to 20 mg. The highest decomposition of MG dye was found at 10 pH which may be due to deprotonation. The effects of Na+, HCO3−, Ca2+, Cl−, K+, SO42− and Mg2+ on dye degradation were examined and results showed that the salts had no effect on degradation. The kinetics and isothermic studies were carried out where the kinetic model pseudo-second-order and Langmuir isotherm were highly fitted. The factorial design was also drawn to checked the combined effects of variables, such as pH, dose, concentration and irradiation time on the degradation of MG dye.













Similar content being viewed by others
References
Gupta V K, Ali I and Saini V K 2007 Water Res. 41 3307
Zhu H, Jiang R, Fu Y, Guan Y, Yao J, Xiao L and Zeng G 2012 Desalination 286 41
Crini G 2006 Bio tech 97 1061
Daneshvar N, Salari D and Khataee A R 2003 J. Photochem. Photobiol. A: Chem. 157 111
Haque E, Jun J W and Jhung S H 2011 J. Haz. Mater. 185 507
Shi B, Li G, Wang D, Feng C and Tang H 2007 J. Haz. Mater. 143 567
Haque A, Shamsuzzoha M, Hussain F and Dean D 2003 J. Comp. Mater. 37 1821
Lee Y Y, Moon J H, Choi Y S, Park G O, Jin M, Jin L Y et al 2017 J. Phys. Chem. C 121 5137
Chu X, Han Y, Zhou S and Shui H 2010 Cer. Inter. 36 2175
Wang Z, Zhu L, Sun S, Wang J and Yan W 2021 Chemosensors 9 198
Zhang S, Yin B, Jiao Y, Liu Y, Qu F and Wu X 2014 Appl. Surf. Sci. 305 626
Hieda K, Hyodo T, Shimizu Y and Egashira M 2008 Sens. Actuators B Chem. 133 144
Vuong D D, Hien V X, Trung K Q and Chien N D 2011 Physica E: Low-Dimens. Syst. Nanostruct. 44 345
George A, Raj D M A, Venci X, Raj A D, Irudayaraj A A, Josephine R L et al 2022 Environ. Res. 203 111880
Abbasi S 2019 Environ. Monit. Assess. 191 (4) 1
Taghavi M, Ghaneian M T, Ehrampoush M H, Tabatabaee M, Afsharnia M, Alami A et al 2018 E M and A 190 1
Sabouri Z, Akbari A, Hosseini H A and Darroudi M 2018 J. Mol. Struct. 1173 931
Mishra S and Maiti A 2019 E M and A 191 1
Sudova E, Machova J, Svobodova Z and Vesely T 2007 Vet. Med. 52 527
Rathore R, Ameta R and Ameta S C 2014 Acta Chim. Pharm. Indica 4 213
Srivastava S, Sinha R and Roy D 2004 Aquat. Toxicol. 66 319
Verdon E and Andersen W C 2016 Chem. Anal. Non-antimicrob. Vet. Drug Residues Food 11 497
Zhang H, Liu D, Ren S and Zhang H 2017 Res. Chem. Intermed. 43 1529
Lanjwani M F, Khuhawar M Y, Khuhawar T M J, Lanjwani A H, Memon S Q et al 2022 J. Chem. Sci. 33 1
Tomaszewska E, Soliwoda K, Kadziola K, Tkacz-Szczesna B, Celichowski G, Cichomski M et al 2013 J. Nanomater. 2013 1
Hoo C M, Starostin N, West P and Mecartney M L 2008 J. Nano. Res. 10 89
Xu R 2008 Particuology 6 112
Groiss S, Selvaraj R, Varadavenkatesan T and Vinayagam R 2017 J. Mol. Struct. 1128 572
Janaki A C, Sailatha E and Gunasekaran S 2015 Spectrochim. Acta A Mol. Biomol. Spectrosc. 144 17
Honarmand M, Golmohammadi M and Naeimi A 2019 Adv. Powder Technol. 30 1551
Luque P A, Nava O, Soto-Robles C A, Chinchillas-Chinchillas M J, Garrafa-Galvez H E, Baez-Lopez Y A et al 2020 Optik 206 164299
Naje A N, Norry A S and Suhail A M 2013 IJIRSET 2 7068
Altunay N, Elik A, Lanjwani M F and Tuzen M 2022 Microchem. J. 179 107541
Lanjwani M F, Altunay N and Tuzen M 2023 Food Chem. 400 134085
Bingol D, Tekin N and Alkan M 2010 Appl. Clay Sci. 50 315
Taymaz B H, Kamis H and Yoldas O 2022 Environ. Eng. Res. 27 153
Elkady M F and Hassan H S 2021 Polymers 13 2033
Devi S A, Singh K J and Devi K N 2020 Inte. Ferroelectrics 205 38
Yang J, Xu X, Liu Y, Gao Y, Chen H and Li H 2019 Colloids Surf. A: Phys. Eng. Asp. 582 123858
Medidi S, Markapurapu S, Kotupalli M R, Chinnam R K R, Susarla V M, Gandham H B et al 2018 Modern Res. Catalysis 7 17
Hussein A S and Fairooz N Y 2016 J. Uni. Babylon 24 9
Baeissa E S 2016 J. Alloy Comp. 672 564
Pérez-Alvarez J, Solis Casados D A, Romero S and Escobar-Alarcón L 2014 Adv. Mater. Res. 976 212
Yang Y C, Wang X Z and Qu J 2014 Adv. Mater. Mater. 522 411
Tolia J, Chakraborty M and Murthy Z V P 2012 Pol. J. Chem. Technol. 14 16
Chen Y, Zhang Y, Liu C, Lu A and Zhang W 2011 Int. J. Photoenergy 2012 1
Chen C C, Lu C S, Chung Y C and Jan J L 2007 J. Haz. Mater. 141 520
Acknowledgements
Authors said thanks to University of Sindh for provided laboratory space.
Author information
Authors and Affiliations
Corresponding author
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lanjwani, M.F., Khuhawar, M.Y., Lanjwani, A.H. et al. Photocatalytic degradation capability of SnO2 nanoparticles as a catalyst vs. malachite green dye: factorial design, kinetic and isotherm models. Bull Mater Sci 47, 1 (2024). https://doi.org/10.1007/s12034-023-03070-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12034-023-03070-7