Abstract
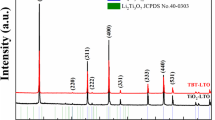
This research investigates the effect of different precursor’s pH values on the fabrication of lithium titanate (Li4Ti5O12) via sol–gel processing and its characteristics as an anode for lithium-ion batteries. The precursor solution of lithium acetate was dropped into titanium butoxide at various initial pH values of 0.14, 0.39 and 0.69. The study of synthesis of Li4Ti5O12 in the extreme acid environment is rarely investigated. Thermogravimetry-differential thermal analyzer was used to examine the thermal profile of the dried Li4Ti5O12 precursor to determine the required sintering temperature, while the crystal structure and morphology of the sintered samples were characterized using an X-ray diffractometer and scanning electron microscope. The thermal analysis shows that the precursor should be sintered at 850°C to acquire pure spinel Li4Ti5O12 crystal. An investigation of the crystal structure shows that a higher purity of Li4Ti5O12 phase with smaller crystallite is obtainable in a more acidic condition. The resultant products contain an agglomerated and irregular morphology with non-uniform size distribution. These results show that the sample with the highest purity of Li4Ti5O12 provides the best electrochemical performance, with a specific capacity of 100 mAh g−1 at 0.1 C scanning rate and fast charging characteristics up to 10 C scanning rate.







Similar content being viewed by others
References
Zhu G N, Liu H J, Zhuang J H, Wang C X, Wang Y G and Xia Y Y 2011 Energy Environ. Sci. 4 4016
Song H, Jeong T G, Yun S W, Lee E K, Park S A and Kim Y T 2017 Sci. Rep. 7 43335
Wu H B, Chen J S, Hng H H and Lou X W 2012 Nanoscale 4 2526
Zaghib K, Dontigny M, Guerfi A, Charest P, Rodrigues I, Mauger A et al 2011 J. Power Sour. 196 3949
Wang X, Liu D, Weng Q, Liu J, Liang Q and Zhang C 2015 NPG Asia Mater. 7 171
Xue X, Yan H and Fu Y 2019 Solid State Ion. 335 1
Zhang S, Ge X and Chen C 2017 Chin. Chem. Lett. 28 2274
Sun B, Wang R, Ni L, Qiu S and Zhang Z 2018 Inorg. Chem. Commun. 93 10
Shen L, Yuan C, Luo H, Zhang X, Xu K and Xia Y 2010 J. Mater. Chem. 20 6998
Drezen T, Kwon N H, Bowen P, Teerlinck I, Isono M and Exnar I 2007 J. Power Sour. 174 949
Yao W, Zhuang W, Ji X, Chen J, Lu X and Wang C 2016 Mater. Res. Bull. 75 204
Zhu J, Duan R, Zhang Y and Zhu J 2016 Ceram. Int. 42 334
Zhang E and Zhang H 2019 Ceram. Int. 45 7419
Sloovere D D, Marchal W, Ulu F, Vranken T, Verheijen M, Bael M K V et al 2017 RSC Adv. 7 18745
Nithya V D, Selvan R K, Vediappan K, Saminathan S and Lee C W 2012 Appl. Surf. Sci. 261 515
Kuo Y C, Peng H T, Xiao Y and Lin J Y 2016 J. Solid State Electrochem. 20 1625
Gockeln M, Pokhrel S, Meierhofer F, Glenneberg J and Kun R 2018 J. Power Sour. 374 97
Gockeln M, Glenneberg J, Busse M, Pokhrel S, Mädler L and Kun R 2018 Nano Energy 49 564
Meierhofer F, Li H, Gockeln M, Kun R, Grieb T, Rosenauer A et al 2017 ACS Appl. Mater. Interfaces 9 37760
Kuo Y C and Lin J Y 2014 Electrochim. Acta 142 43
Tsega M and Dejene F 2017 Heliyon 3 e00246
Ibrahim S A and Sreekantan S 2011 Adv. Mater. Res. 173 184
Ekemena O O, Patrick G N and Sreekanth B J 2015 J. Adv. Oxid. Technol. 18 253
Brinker C J and Scherer G W 2013 Sol-gel science: the physics and chemistry of sol-gel processing (Boston: Academic press)
Hanaor D A and Sorrell C C 2011 J. Mater. Sci. 46 855
Shen C M, Zhang X G, Zhou Y K and Li H L 2003 Mater. Chem. Phys. 78 437
Kumar S G and Rao K S R K 2014 Nanoscale 6 11574
Waseda Y, Matsubara E and Shinoda K 2011 X-ray diffraction crystallography: introduction, examples and solved problems (Heidelberg: Springer)
Grupp R, Nöthe M, Kieback B and Banhart J 2011 Nat. Commun. 2 298
Park J S, Baek S H, Jeong Y I, Noh B Y and Kim J H 2013 J. Power Sour. 244 527
Sudakar C, Subbanna G N and Kutty T R N 2004 J. Mater. Sci. 39 4271
Selvan R K and Gedanken A 2009 Nanotechnology 20 105602
Yuan T, Yu X, Cai R, Zhou Y and Shao Z 2010 J. Power Sour. 195 4997
Luo G, He J, Song X, Huang X, Yu X, Fang Y et al 2015 J. Alloys Compd. 621 268
Liu Y, Yan X, Xu B, Lan J, Yu Y, Yang X et al 2019 Chem. Eng. J. 361 1371
Wang J 2000 Analytical electrochemistry 2nd edn. (New York: Wiley-VCH)
Wolberg J 2006 Data analysis using the method of least squares: extracting the most information from experiments (Heidelberg: Springer)
Pohjalainen E, Rauhala T, Valkeapää M, Kallionen J and Kallio T 2015 J. Phys. Chem. C 119 2277
Deng D, Kim M G, Lee J Y and Cho J 2009 Energy Environ. Sci. 2 818
Bläubaum L, Röder F, Nowak C, Chan H S, Kwade A and Krewer U 2020 ChemElectroChem 7 4755
Erdas A, Ozcan S, Guler M O and Akbulut H 2015 Acta Phys. Pol. A 127 1026
Chen Z, Xu N, Li W, Zhao R, Dong Y, Liu J et al 2019 J. Mater. Chem. A 7 16347
Wang Y Q, Gu L, Guo Y G, Li H, He X Q, Tsukimoto S et al 2012 JACS 134 7874
Acknowledgements
We would like to appreciate the financial support from the Directorate for Research and Community Services of Universitas Indonesia through the Doctoral Research Grant for International Publication Year 2019, under contract no.: NKB-0153/UN2.R3.1/HKP.05.00/2019.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Priyono, S., Sofyan, N., Dhaneswara, D. et al. pH effect upon sol–gel processing of titanium butoxide and lithium acetate precursors on the characteristics of Li4Ti5O12 as an anode for lithium-ion batteries. Bull Mater Sci 45, 147 (2022). https://doi.org/10.1007/s12034-022-02736-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12034-022-02736-y