Abstract
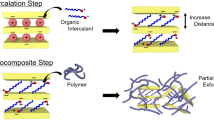
Muscovite clay is an ideal reinforcing filler due to its high-aspect ratio. However, it does not swell in water, making it hard to be treated and intercalated. In this study, ion exchange treatment is carried out on muscovite clay using cetyltrimethylammonium bromide (CTAB) cations via two-step intercalation method. The intercalation steps included: inorganic–inorganic ion exchange treatment and inorganic–organic ion exchange treatment under hydrothermal conditions. The intercalation of muscovite particles was examined with various techniques to analyse the physical and chemical changes. Furthermore, the hydrothermal conditions for effective \(\hbox {CTA}^{+}\) ion intercalation within muscovite interlayers prepared via the hydrothermal process at low temperature, \(180{^{\circ }}\hbox {C}\), under different hydrothermal reaction times and CTAB/Li-Mus mass ratio were investigated. Fourier transform infra-red (FTIR) analysis revealed that the \(\hbox {CTA}^{+}\) ions are diffused into the interlayers of aluminosilicate and formed a strong electrostatic bond with the clay surface. X-ray powder diffraction analysis showed that the interplanar spacing in the organo-muscovite samples is almost identical as the hydrothermal reaction time is prolonged beyond 12 h. An optimum limit of the CTAB to Li-Mus ratio is observed as the \(d_{002}\) plane spacing is increased with an increase of the mass ratio of CTAB to Li-Mus up to 1.0 C and decreased with a further increase in the mass ratio. In addition, the intercalated \(\hbox {CTA}^{+}\) chains are homogenously distributed and formed a paraffin-like arrangement in the muscovite clay. Besides, the structure of aluminosilicate layers is not affected or damaged after both treatments according to FTIR analysis.













Similar content being viewed by others
References
Bekri-Abbes I and Srasra E 2006 Bull. Mater. Sci. 29 251
Murray H H 1991 Appl. Clay Sci. 5 379
Sharma S, Kumar P M and Moholkar V S 2017 Ultrason. Sonochem. 36 212
Jahanmardi R, Kangarlou B and Dibazar A 2013 J. Nanostruct. Chem. 3 82
Strankowska J, Piszczyk Ł, Strankowski M, Danowska M, Szutkowski K, Jurga S et al. 2013 Eur. Phys. J. Spec. Top. 222 2179
Xi X, Zhen W, Bian S and Wang W 2015 Polymer (Korea) 39 601
Mousa M H, Dong Y and Davies I J 2016 Int. J. Polym. Mater. Polym. Biomater. 65 225
Yu X, Zhao L, Gao X, Zhang X and Wu N 2006 J. Solid State Chem. 179 1569
Yu X, Zhao L, Gao X, Zhang X and Wu N 2006 J. Solid State Chem. 179 1525
Omar M F, Akil H M, Ahmad Z A, Rasyid M F A and Noriman N Z 2016 J. Thermoplast. Compos. Mater. 29 867
Rashid E S A, Rasyid M F A, Akil H M, Ariffin K and Kooi C C 2011 Appl. Clay Sci. 52 295
Kudus M H A, Akil H M and Rasyid M F A 2012 Mater. Lett. 79 92
Aitta E, Leskelä M, Lajunen L H J, Jyrkäs K and Seppälä E 1986 J. Chem. Technol. Biotechnol. 36 169
Caseri W R, Shelden R A and Suter U W 1992 Colloid Polym. Sci. 270 392
Leiro J A, Torhola M and Laajalehto K 2017 J. Phys. Chem. Solids 100 40
Almeida L A, de Marques M F V and Dahmouche K 2018 J. Appl. Polym. Sci. 135 45587
Omar M F, Akil H M, Rasyid M F A and Sharif J M 2015 J. Compos. Mater. 49 1195
Yu X 2007 Micropor. Mesopor. Mat. 98 70
Bracke G, Satir M and Krauß P 1995 Clays Clay Miner. 43 732
Kodama T, Hasegawa K, Shimizu K and Komarneni S 2003 Sep. Sci. Technol. 38 679
Shimizu K, Hasegawa K, Nakamuro Y, Kodama T and Komarneni S 2004 J. Mater. Chem. 14 1031
Zhao L Y, Wang X K, Wu N Z and Xie Y C 2005 Colloid Polym. Sci. 283 699
Metz S, Anderson R L, Geatches D L, Suter J L, Lines R and Greenwell H C 2015 J. Phys. Chem. C 119 12625
Che Ismail N H, Ahmad Bakhtiar N S A and Md Akil H 2017 Mater. Chem. Phys. 196 324
Bae H J, Goh Y, Yim H, Yoo S Y, Choi J W and Kwon D K 2019 Mater. Chem. Phys. 221 168
Tominaga Y, Fukushima K, Takezawa Y, Shimamoto D, Imai Y and Hotta Y 2017 Adv. Powder Technol. 28 1911
Pozsgay A 2004 Eur. Polym. J. 40 27
Paul R, Datta S C, Manjaiah K M and Bhattacharyya R 2017 Appl. Clay Sci. 144 19
Ramadan A R, Esawi A M K and Gawad A A 2010 Appl. Clay Sci. 47 196
Sayin M and Von Reichenbach H G 1978 Clay Miner. 13 241
Stubičan V and Roy R 1961 Am. Mineral. 46 32
Farmer V C and Russell J D 1964 Spectrochim. Acta 20 1149
Olad A 2011 in Reddy B (ed) Advances in diverse industrial applications of nanocomposites (Croatia: InTech) p 122
Garcia A D and Catterton A J 2003 Microgram J. 1 44
Lee S Y and Kim S J 2002 Clays Clay Miner. 50 435
Saeid G, Mark T B and Andrew B 1997 J. Colloid Interface Sci. 196 191
Xi Y, Frost R L and He H 2007 J. Colloid Interface Sci. 305 150
Karp G 2013 in Witt K, Stauber L, Morris L (eds) Cell and molecular biology: concepts and experiments, 7th edn (Hoboken, NJ: John Wiley & Sons Ltd) p 120
Wang L, Chen Z, Wang X, Yan S, Wang J and Fan Y 2011 Appl. Clay Sci. 51 151
Liang Y, Ding H, Wang Y, Liang N and Wang G 2013 Appl. Clay Sci. 74 109
Acknowledgements
This study was financially supported by short term grant of Universiti Sains Malaysia (grant no. 304/PBAHAN/60313035).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Weng-Lip, L., Salleh, N.M., Rahman, N.‘.A. et al. Enhanced intercalation of organo-muscovite prepared via hydrothermal reaction at low temperature. Bull Mater Sci 42, 242 (2019). https://doi.org/10.1007/s12034-019-1931-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12034-019-1931-0