Abstract
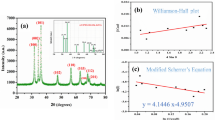
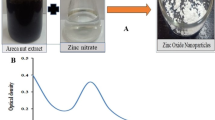
Chemical co-precipitation route was successfully employed to synthesize polyethylene glycol-coated pure and doped \(\hbox {Zn}_{1-x}\hbox {Mn}_{x}\hbox {S}\,(0\le x \le 0.1)\) nanoparticles. The crystallographic and morphological analyses have been done by X-ray diffraction (XRD) and transmission electron microscopy (TEM), respectively. The formation of cubic crystal structure and quasi-spherical morphology has been revealed by XRD and TEM, respectively. The optical analyses have been done by UV–Vis absorption spectroscopy and energy resolved photoluminescence spectroscopy. Energy dispersive X-ray spectroscopy study has been carried to analyse the elemental composition. The doping concentration dependent photo-catalytic activity was checked to analyse the photo-catalytic potential of \(\hbox {Zn}_{1-x}\hbox {Mn}_{x}\hbox {S}\) nanoparticles under UV irradiation.









Similar content being viewed by others
References
Biswas S and Kar S 2008 Nanotechnology 19 045710
Matsui I 2005 J. Chem. Eng. Jpn. 38 535
Huang C Y, Wang D Y, Wang C H, Chen Y T et al 2010 ACS Nano 10 5849
Ayodhya D and Veerabhadram G 2016 J. Fluoresc. 26 2165
He Y, Wang H F and Yan X P 2008 Anal. Chem. 80 3832
Sung T W and Lo Y L 2012 Sens. Actuators B: Chem. 165 119
Koneswaran M and Narayanaswamy R 2009 Sens. Actuators B: Chem. 139 104
Hu H and Zhang W 2006 Opt. Mater. 28 536
Hu Z, Li L, Zhou X, Fu X and Gu G 2006 J. Colloid Interface Sci. 294 328
Ahmad T, Khatoon S, Coolahan K and Lofland S E 2013 J. Mater. Res. 28 1245
Planelles-Arago J, Julian-Lopez B, Cordoncillo E, Escribano P, Pellé F, Viana B et al 2008 J. Mater. Chem. 185 193
Lenggoro I W, Okuyama K, de la Mora J F and Tohge N 2000 J. Aerosol Sci. 31 121
Al-Hartomy O A, Ubaidullah M, Kumar D, Madani J H and Ahmad T 2013 J. Mater. Res. 28 1070
Ahmad T, Ramanujachary K V, Lofland S E and Ganguli A K 2006 J. Chem. Sci. 118 513
Cholan S, Shanmugam N, Kannadasan N, Sathish Kumar K and Deivam K 2014 J. Mater. Res. Technol. 3 222
Viswanath R, Naik H B, Kumar G Y, Kumar P P, Harish K N, Prabhakara M C et al 2014 Appl. Surf. Sci. 301 126
Kaur B, Singh K and Malik A K 2017 Dyes Pigments 142 153
Bhargava R N, Gallagher D, Hong X and Nurmikko A 1994 Phys. Rev. Lett. 72 416
Goede O, Heimbrodt W and Weinhold V 1986 Phys. Status Solidi b 136 K49
Viswanath R, Naik H B, Kumar G Y, Kumar P P, Kumar G A and Praveen R 2014 J. Lumin. 153 446
Houas A, Lachheb H, Ksibi M, Elaloui E, Guillard C and Herrmann J M 2001 Appl. Catal. B 31 145
Ahmad T, Phul R, Alam P, Lone I H, Shahazad M, Ahmed J et al 2017 RSC Adv. 7 27549
Acknowledgements
One of the authors, Mrs Balwinder Kaur is thankful to University Grants Commission (UGC), New Delhi, India for providing financial support under scheme Maulana Azad National Fellowships (MANF). The author is obliged to Sophisticated Instrument Centre, Punjabi University, Patiala for providing necessary facilities. The author is grateful to EMN laboratory Punjab Agriculture University, Ludhiana for TEM analysis.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kaur, B., Chand, S., Singh, K. et al. Detoxification of dye contaminated water by Mn\(^{2+}\)-doped ZnS nanostructures. Bull Mater Sci 42, 61 (2019). https://doi.org/10.1007/s12034-019-1738-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12034-019-1738-z