Abstract
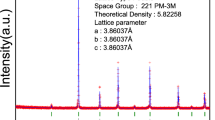
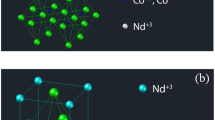
In this study, oxides Ln0.6Sr0.4Co0.2M0.8O3−δ (Ln = La,Nd and M = Mn,Fe) have been prepared and characterized to study the influence of the different cations on thermal expansion coefficient (TEC). TEC decreases favourably with replacement of Nd3+ and Mn3+ ions in the lattice. Nd3+ leads to decreasing ionicity of the Ln–O bond and phase transition from rhombohedral to orthorhombic. Structural parameters of compounds have been determined by X-ray diffraction (XRD). FT-IR analysis has been used to compare the bond strength of different cations in the lattice. Variations of lattice parameters of compounds have been monitored by in-situ XRD at different temperatures in order to study the thermo-chemical expansivity. Concentration of oxide ion vacancy has been determined by high temperature gravimetry. The results show more stability of Mn4+ ions in high temperature in comparison with Fe4+ ions, which suppreses the tendency to lose oxygen from the lattice with the increase in temperature.








Similar content being viewed by others
References
Sun C, Hui R and Roller J 2010 J. Solid State Electrochem. 14 1125
Radhika D and Nesaraj A S 2013 J. Renew. Energy Dev. 2 87
Fergus J W 2007 J. Hydrogen Econ. 12 56
Chang Ch, Hsu Ch, Huang J and Hwang B 2015 J. Alloys Compd. 620 233
Lenka R K, Mahata T, Patro P K, Tyagi A K and Sinha P K 2012 J. Alloys Compd. 537 100
Lee K T and Manthiram A 2006 J. Electrochem. Soc. 153 794
Skinner S J 2001 Int. J. Inorg. Mater. 3 113
Wang J H, Liu M L and Lin M C 2006 J. Solid State Ion. 177 939
Mizusaki J 1992 J. Solid State Ion. 52 79
Hashimoto S, Fukuda Y, Kuhn M, Sato K, Yashiro K and Mizusaki J 2011 J. Solid State Ion. 186 37
Kuhn M, Hashimoto S, Sato K, Yashiro K and Mizusaki J 2011 J. Solid State Ion. 195 7
Bishop S R, Duncan K L and Wachsman E D 2010 J. Am. Ceram. Soc. 93 4115
Miyoshi S, Hong J O, Yashiro K, Kaimai A, Nigara Y, Kawamura K, Kawada T and Mizusaki J 2003 J. Solid State Ion. 161 209
Hilpert K, Steinbrech R W, Boroomand F, Wessel E, Meschke F, Zuev A, Teller O, Nickel H and Singheiser L 2003 J. Eur. Ceram. Soc. 23 3009
Bishop S R, Tuller H L, Kuru Y and Yildiz B 2011 J. Eur. Ceram. Soc. 31 2351
Kuhn M, Hashimoto S, Sato K, Yashiro K and Mizusaki J 2013 J. Solid State Ion. 241 12
Hashimoto Sh, Fukuda Y, Kuhn M, Sato K, Yashiro K and Mizusaki J 2011 J. Solid State Ion. 186 37
Riza F, Ftikos Ch, Tietz F and Fischer W 2001 J. Eur. Ceram. Soc. 21 1769
Kuhn M, Hashimoto S, Sato K, Yashiro K and Mizusaki J 2011 J. Solid State Ion. 195 7
Tai L-W, Nasrallah M M, Anderson H U, Sparlin D M and Sehlin S R 1995 J. Solid State Ion. 76 259
Radaelli P G, Iannone G, Marezio M, Hwang H Y, Cheong S-W, Jorgensen J D and Argyriou N D 1997 J. Phys. Rev. B 56 8265
Mori M, Hiei Y, Sammes N M and Tompsett G A 2000 J. Electrochem. Soc. 147 1295
Callister W D 2000 Journal of materials science and engineering: an introduction (New York: John Wiley & Sons Inc.) 5th ed., p 23
Barsoum M 1997 Fundamentals of ceramics (New York: McGraw-Hill Companies Inc.) p 26
De Marzi G, Popovic Z V, Cantarero A, Mitrovic Z D and Paunovic N 2003 J. Phys. Rev. B 68 643022
Mogensen M, Lybye D, Bonanos N, Hendriksen P V and Poulsen F W 2004 J. Solid State Ion. 174 279
Kilner J A and Brook R J 1982 Solid State Ionics 6 237
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
HEYDARI, F., MAGHSOUDIPOUR, A., ALIZADEH, M. et al. Synthesis and evaluation of effective parameters in thermal expansion coefficient of Ln0.6Sr0.4Co0.2M0.8O3−δ (Ln = La,Nd and M = Mn,Fe) perovskite oxide. Bull Mater Sci 38, 1009–1014 (2015). https://doi.org/10.1007/s12034-015-0942-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12034-015-0942-8