Abstract
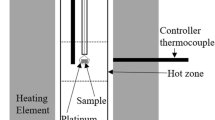
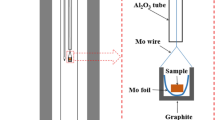
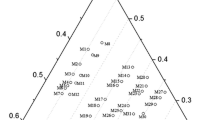
Phase relations in the system Ca-Ti-O have been established by equilibration of several samples at 1200 K for prolonged periods and identification of phases in quenched samples by optical and scanning electron microscopy, XRD and EDS. Samples representing 20 compositions in the ternary system were analyzed. There was negligible solid solubility of Ca in the phases along the binary Ti-O, and of Ti in CaO. Four ternary oxides were identified: CaTiO3, Ca4Ti3O10 and Ca3Ti2O7 containing tetravalent titanium, and CaTi2O4 containing trivalent titanium. Tie-lines link calcium titanite (CaTi2O4) with the three calcium titanates (CaTiO3, Ca4Ti3O10 and Ca3Ti2O7), CaO, oxygen excess TiO1+δ and stoichiometric TiO. Tie-lines connect CaTiO3 with TiO2−x , Magneli phases Ti n O2n−1 (28 ≥ n ≥ 4), Ti3O5, Ti2O3 and TiO1+δ . CaO was found to coexist with TiO, and Ti-O solid solutions α and β. The phase diagram is useful for understanding the mechanisms and kinetics of direct calciothermic reduction of TiO2 to metal and electrochemical reduction of TiO2 using graphite anode and molten CaCl2 electrolyte.
Similar content being viewed by others
References
Cancarevic M, Zinkevich M and Aldinger F 2007 Calphad 31 330
Chase Jr M W 1998 NIST-JANAF Thermochemical Tables Part-II, Monograph No. 9, Journal of Physical and Chemical Reference Data (New York: American Institute of Physics) 4th edn, p. 1734
Chen G Z, Fray D J and Farthing T W 2000 Nature 407 361
Deura T, Okabe T H, Oishi T and Ono K 1992 in Proceedings of the 24 th symposium on molten salt chemistry (ed.) Y Ito (Kyoto: Electrochemical Society of Japan) p. 3
Jacob K T and Mathews T 1991 J. Mater. Chem. 1 545
Jacob K T and Abraham K P 2009 J. Chem. Thermodyn. 41 816
Jacob K T, Hoque S M and Waseda Y 2000 Mater. Trans. JIM (Japan Institute of Metals) 41 681
Jongejan A and Wilkins A L 1970 J. Less-Common Metals 20 273
Kubaschewski O 1983 in Titanium: Physico-chemical properties of its compounds and alloys (ed.) K L Komarek Atomic Energy Review (Vienna: International Atomic Energy Agency) p. 22
Massalski T B, Okamoto H, Subramanian P R and Kacprzak L 1990 Binary alloy phase diagrams (Metals Park, Ohio: American Society for Metals) p. 2924
Okabe T H, Suzuki R O, Oishi T and Ono K 1991 Mater. Trans. JIM 32 485
Okamoto H 2001 J. Phase Equilibria 22 515
Oki T and Inoue H 1967 Mem. Fac. Eng. Nagoya Univ. 19 164
Rogge M P, Caldwell J H, Ingram D R, Green C E, Geselbracht M J and Siegrist T 1998 J. Solid State Chem. 141 338
Roth R S 1958 J. Res. Natl. Bur. Stand. 61 437
Smyth D M 1993 in Properties and applications of perovskitetype oxides (eds) L G Tejuca and J L G Fierro (New York: Marcel Dekker) p. 47
Ye G and Rosenqvist T 1991 Scand. J. Metall. 20 222
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jacob, K.T., Gupta, S. Phase diagram of the system Ca-Ti-O at 1200 K. Bull Mater Sci 32, 611–616 (2009). https://doi.org/10.1007/s12034-009-0094-9
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12034-009-0094-9