Abstract
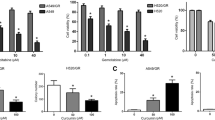
Non-small cell lung cancer (NSCLC) is a fatal malignancy all over the world. Emerging studies have shown that curcumin might repress NSCLC progression by regulating ferroptosis, but the underlying mechanism remains unclear. 16HBE, LK-2, and H1650 cell viability was detected using Cell Counting Kit-8 assay. LK-2 and H1650 cell proliferation, apoptosis, and angiopoiesis were measured using 5-ethynyl-2’-deoxyuridine, flow cytometry, and tube formation assay. Superoxide dismutase, Malondialdehyde, Glutathione, and lactate dehydrogenase levels in LK-2 and H1650 cells were examined using special assay kits. Fe+ level was assessed using an iron assay kit. Doublesex and Mab-3 related Transcription Factor 3 (DMRT3) and solute carrier family 7 member 11 (SLC7A11) protein levels were detected using western in NSCLC tissues, adjacent matched normal tissues, 16HBE cells, LK-2 cells, H1650 cells, and xenograft tumor tissues. Glutathione peroxidase 4, Acyl-CoA Synthetase Long Chain Family Member 4, and transferrin receptor 1 protein levels in LK-2 and H1650 cells were examined by western blot assay. DMRT3 and SLC7A11 levels were determined using real-time quantitative polymerase chain reaction. After JASPAR prediction, binding between DMRT3 and SLC7A11 promoter was verified using Chromatin immunoprecipitation and dual-luciferase reporter assays in LK-2 and H1650 cells. Role of curcumin on NSCLC tumor growth was assessed using the xenograft tumor model in vivo. Curcumin blocked NSCLC cell proliferation and angiopoiesis, and induced apoptosis and ferroptosis. DMRT3 or SLC7A11 upregulation partly abolished the suppressive role of curcumin on NSCLC development. In mechanism, DMRT3 was a transcription factor of SLC7A11 and increased the transcription of SLC7A11 via binding to its promoter region. Curcumin inhibited NSCLC growth in vivo by modulating DMRT3. Curcumin might constrain NSCLC cell malignant phenotypes partly through the DMRT3/SLC7A11 axis, providing a promising therapeutic strategy for NSCLC.







Similar content being viewed by others
References
Siegel, R. L., Miller, K. D., Wagle, N. S., & Jemal, A. (2023). Cancer statistics, 2023. CA: A Cancer Journal for Clinicians, 73(1), 17–48. https://doi.org/10.3322/caac.21763
Leiter, A., Veluswamy, R. R., & Wisnivesky, J. P. (2023). The global burden of lung cancer: Current status and future trends. Nature Reviews. Clinical Oncology, 20(9), 624–639. https://doi.org/10.1038/s41571-023-00798-3
Sung, H., Ferlay, J., Siegel, R. L., Laversanne, M., Soerjomataram, I., Jemal, A., & Bray, F. (2021). Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA: A Cancer Journal for Clinicians, 71(3), 209–249. https://doi.org/10.3322/caac.21660
Rodak, O., Peris-Díaz, M. D., Olbromski, M., Podhorska-Okołów, M., & Dzięgiel, P. (2021). Current landscape of non-small cell lung cancer: epidemiology, histological classification, targeted therapies, and immunotherapy. Cancers (Basel). https://doi.org/10.3390/cancers13184705
Duma, N., Santana-Davila, R., & Molina, J. R. (2019). Non-small cell lung cancer: epidemiology, screening, diagnosis, and treatment. Mayo Clinic Proceedings, 94(8), 1623–1640. https://doi.org/10.1016/j.mayocp.2019.01.013
Reck, M., Heigener, D. F., Mok, T., Soria, J. C., & Rabe, K. F. (2013). Management of non-small-cell lung cancer: Recent developments. Lancet, 382(9893), 709–719. https://doi.org/10.1016/s0140-6736(13)61502-0
Imran, M., Ullah, A., Saeed, F., Nadeem, M., Arshad, M. U., & Suleria, H. A. R. (2018). Cucurmin, anticancer, & antitumor perspectives: A comprehensive review. Critical Reviews in Food Science and Nutrition, 58(8), 1271–1293. https://doi.org/10.1080/10408398.2016.1252711
Kotha, R. R., & Luthria, D. L. (2019). Curcumin: Biological, Pharmaceutical, Nutraceutical, and Analytical Aspects. Molecules. https://doi.org/10.3390/molecules24162930
Kocaadam, B., & Şanlier, N. (2017). Curcumin, an active component of turmeric (Curcuma longa), and its effects on health. Critical Reviews in Food Science and Nutrition, 57(13), 2889–2895. https://doi.org/10.1080/10408398.2015.1077195
Górnicka, J., Mika, M., Wróblewska, O., Siudem, P., & Paradowska, K. (2023). Methods to improve the solubility of curcumin from turmeric. Life (Basel). https://doi.org/10.3390/life13010207
Luis, P. B., Kunihiro, A. G., Funk, J. L., & Schneider, C. (2020). Incomplete hydrolysis of curcumin conjugates by β-glucuronidase: Detection of complex conjugates in plasma. Molecular Nutrition & Food Research, 64(6), e1901037. https://doi.org/10.1002/mnfr.201901037
Wang, R., Han, J., Jiang, A., Huang, R., Fu, T., Wang, L., Zheng, Q., Li, W., & Li, J. (2019). Involvement of metabolism-permeability in enhancing the oral bioavailability of curcumin in excipient-free solid dispersions co-formed with piperine. International Journal of Pharmaceutics. https://doi.org/10.1016/j.ijpharm.2019.02.027
Soleimani, V., Sahebkar, A., & Hosseinzadeh, H. (2018). Turmeric (Curcuma longa) and its major constituent (curcumin) as nontoxic and safe substances: Review. Phytotherapy Research, 32(6), 985–995. https://doi.org/10.1002/ptr.6054
Ming, T., Tao, Q., Tang, S., Zhao, H., Yang, H., Liu, M., Ren, S., & Xu, H. (2022). Curcumin: An epigenetic regulator and its application in cancer. Biomedicine & Pharmacotherapy. https://doi.org/10.1016/j.biopha.2022.113956
Giordano, A., & Tommonaro, G. (2019). Curcumin and cancer. Nutrients. https://doi.org/10.3390/nu11102376
Mehta, H. J., Patel, V., & Sadikot, R. T. (2014). Curcumin and lung cancer—a review. Targeted Oncology, 9(4), 295–310. https://doi.org/10.1007/s11523-014-0321-1
Tang, X., Ding, H., Liang, M., Chen, X., Yan, Y., Wan, N., Chen, Q., Zhang, J., & Cao, J. (2021). Curcumin induces ferroptosis in non-small-cell lung cancer via activating autophagy. Thoracic Cancer, 12(8), 1219–1230. https://doi.org/10.1111/1759-7714.13904
Tang, C., Liu, J., Yang, C., Ma, J., Chen, X., Liu, D., Zhou, Y., Zhou, W., Lin, Y., & Yuan, X. (2022). Curcumin and its analogs in non-small cell lung cancer treatment: Challenges and expectations. Biomolecules. https://doi.org/10.3390/biom12111636
Zhang, T., & Zarkower, D. (2017). DMRT proteins and coordination of mammalian spermatogenesis. Stem Cell Research. https://doi.org/10.1016/j.scr.2017.07.026
Li, H. L., Zou, Z. C., Fang, C., Zheng, Y. P., Guo, X. M., & Yang, W. H. (2023). Mammalian DMRTs: Structure, function and relationship with cancer. Sheng Li Xue Bao, 75(2), 269–278.
Bellefroid, E. J., Leclère, L., Saulnier, A., Keruzore, M., Sirakov, M., Vervoort, M., & De Clercq, S. (2013). Expanding roles for the evolutionarily conserved Dmrt sex transcriptional regulators during embryogenesis. Cellular and Molecular Life Sciences, 70(20), 3829–3845. https://doi.org/10.1007/s00018-013-1288-2
Kopp, A. (2012). Dmrt genes in the development and evolution of sexual dimorphism. Trends in Genetics, 28(4), 175–184. https://doi.org/10.1016/j.tig.2012.02.002
Yang, D., Liu, M., Jiang, J., Luo, Y., Wang, Y., Chen, H., Li, D., Wang, D., Yang, Z., & Chen, H. (2022). Comprehensive analysis of DMRT3 as a potential biomarker associated with the immune infiltration in a pan-cancer analysis and validation in lung adenocarcinoma. Cancers (Basel). https://doi.org/10.3390/cancers14246220
Zhang, S., Li, M., Ji, H., & Fang, Z. (2018). Landscape of transcriptional deregulation in lung cancer. BMC Genomics, 19(1), 435. https://doi.org/10.1186/s12864-018-4828-1
Ma, L., Chen, T., Zhang, X., Miao, Y., Tian, X., Yu, K., Xu, X., Niu, Y., Guo, S., Zhang, C., Qiu, S., Qiao, Y., Fang, W., Du, L., Yu, Y., & Wang, J. (2021). The m(6)A reader YTHDC2 inhibits lung adenocarcinoma tumorigenesis by suppressing SLC7A11-dependent antioxidant function. Redox Biology. https://doi.org/10.1016/j.redox.2020.101801
He, F., Zhang, P., Liu, J., Wang, R., Kaufman, R. J., Yaden, B. C., & Karin, M. (2023). ATF4 suppresses hepatocarcinogenesis by inducing SLC7A11 (xCT) to block stress-related ferroptosis. Journal of Hepatology, 79(2), 362–377. https://doi.org/10.1016/j.jhep.2023.03.016
Zhang, W., Sun, Y., Bai, L., Zhi, L., Yang, Y., Zhao, Q., Chen, C., Qi, Y., Gao, W., He, W., Wang, L., Chen, D., Fan, S., Chen, H., Piao, H. L., Qiao, Q., Xu, Z., Zhang, J., Zhao, J., … Wang, Y. (2021). RBMS1 regulates lung cancer ferroptosis through translational control of SLC7A11. Journal of Clinical Investigation. https://doi.org/10.1172/jci152067
Shah, M., Murad, W., Mubin, S., Ullah, O., Rehman, N. U., & Rahman, M. H. (2022). Multiple health benefits of curcumin and its therapeutic potential. Environmental Science and Pollution Research International, 29(29), 43732–43744. https://doi.org/10.1007/s11356-022-20137-w
Ashrafizadeh, M., Najafi, M., Makvandi, P., Zarrabi, A., Farkhondeh, T., & Samarghandian, S. (2020). Versatile role of curcumin and its derivatives in lung cancer therapy. Journal of Cellular Physiology, 235(12), 9241–9268. https://doi.org/10.1002/jcp.29819
Salehi, M., Movahedpour, A., Tayarani, A., Shabaninejad, Z., Pourhanifeh, M. H., Mortezapour, E., Nickdasti, A., Mottaghi, R., Davoodabadi, A., Khan, H., Savardashtaki, A., & Mirzaei, H. (2020). Therapeutic potentials of curcumin in the treatment of non-small-cell lung carcinoma. Phytotherapy Research, 34(10), 2557–2576. https://doi.org/10.1002/ptr.6704
Tong, X., Tang, R., Xiao, M., Xu, J., Wang, W., Zhang, B., Liu, J., Yu, X., & Shi, S. (2022). Targeting cell death pathways for cancer therapy: Recent developments in necroptosis, pyroptosis, ferroptosis, and cuproptosis research. Journal of Hematology & Oncology, 15(1), 174. https://doi.org/10.1186/s13045-022-01392-3
Strasser, A., & Vaux, D. L. (2020). Cell death in the origin and treatment of cancer. Molecular Cell, 78(6), 1045–1054. https://doi.org/10.1016/j.molcel.2020.05.014
Yan, H. F., Zou, T., Tuo, Q. Z., Xu, S., Li, H., Belaidi, A. A., & Lei, P. (2021). Ferroptosis: Mechanisms and links with diseases. Signal Transduction and Targeted Therapy, 6(1), 49. https://doi.org/10.1038/s41392-020-00428-9
Rochette, L., Dogon, G., Rigal, E., Zeller, M., Cottin, Y., & Vergely, C. (2022). Lipid peroxidation and iron metabolism: Two corner stones in the homeostasis control of ferroptosis. International Journal of Molecular Sciences. https://doi.org/10.3390/ijms24010449
Zou, J., Wang, L., Tang, H., Liu, X., Peng, F., & Peng, C. (2021). Ferroptosis in non-small cell lung cancer: Progression and therapeutic potential on it. International Journal of Molecular Sciences. https://doi.org/10.3390/ijms222413335
Xu, X., Zhang, X., Zhang, Y., & Wang, Z. (2021). Curcumin suppresses the malignancy of non-small cell lung cancer by modulating the circ-PRKCA/miR-384/ITGB1 pathway. Biomedicine & Pharmacotherapy. https://doi.org/10.1016/j.biopha.2021.111439
Wang, J. Y., Wang, X., Wang, X. J., Zheng, B. Z., Wang, Y., Wang, X., & Liang, B. (2018). Curcumin inhibits the growth via Wnt/β-catenin pathway in non-small-cell lung cancer cells. European Review for Medical and Pharmacological Sciences, 22(21), 7492–7499. https://doi.org/10.26355/eurrev_201811_16290
Xu, X., & Zhu, Y. (2017). Curcumin inhibits human non-small cell lung cancer xenografts by targeting STAT3 pathway. American Journal of Translational Research, 9(8), 3633–3641.
Katsuta, E., Huyser, M., Yan, L., & Takabe, K. (2021). A prognostic score based on long-term survivor unique transcriptomic signatures predicts patient survival in pancreatic ductal adenocarcinoma. American Journal of Cancer Research, 11(9), 4294–4307.
Meng, Y., Li, S., Gu, D., Xu, K., Du, M., Zhu, L., Chu, H., Zhang, Z., Wu, Y., Fu, Z., & Wang, M. (2020). Genetic variants in m6A modification genes are associated with colorectal cancer risk. Carcinogenesis, 41(1), 8–17. https://doi.org/10.1093/carcin/bgz165
Lambert, S. A., Jolma, A., Campitelli, L. F., Das, P. K., Yin, Y., Albu, M., Chen, X., Taipale, J., Hughes, T. R., & Weirauch, M. T. (2018). The Human Transcription Factors. Cell, 172(4), 650–665. https://doi.org/10.1016/j.cell.2018.01.029
Li, S., Lu, Z., Sun, R., Guo, S., Gao, F., Cao, B., & Aa, J. (2022). The role of SLC7A11 in Cancer: Friend or Foe? Cancers (Basel). https://doi.org/10.3390/cancers14133059
Lu, X., Kang, N., Ling, X., Pan, M., Du, W., & Gao, S. (2021). MiR-27a-3p promotes non-small cell lung cancer through SLC7A11-mediated-ferroptosis. Frontiers in Oncology. https://doi.org/10.3389/fonc.2021.759346
Xu, Y., Lv, D., Yan, C., Su, H., Zhang, X., Shi, Y., & Ying, K. (2022). METTL3 promotes lung adenocarcinoma tumor growth and inhibits ferroptosis by stabilizing SLC7A11 m(6)A modification. Cancer Cell International, 22(1), 11. https://doi.org/10.1186/s12935-021-02433-6
Acknowledgements
None.
Funding
None.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
12033_2024_1166_MOESM1_ESM.tif
Supplementary file1 (TIF 220 KB)—Figure S1 DMRT3 and SLC7A11 expression were detected in NSCLC cells. (A) RT-qPCR assay was used to measure DMRT3 mRNA level in LK-2 and H1650 cells treated with pcDNA, DMRT3, Curcumin + pcDNA, or Curcumin + DMRT3. (B) SLC7A11 mRNA level was assessed in LK-2 and H1650 cells treated with pcDNA, SLC7A11, Curcumin + pcDNA, or Curcumin + SLC7A11 using RT-qPCR assay. *P < 0.05
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Xu, B., Zhou, L. & Zhang, Q. Curcumin Inhibits the Progression of Non-small Cell Lung Cancer by Regulating DMRT3/SLC7A11 Axis. Mol Biotechnol (2024). https://doi.org/10.1007/s12033-024-01166-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12033-024-01166-x