Abstract
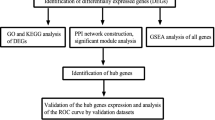
This study focused on identifying potential key lncRNAs associated with gout under the mechanisms of copper death and iron death through ceRNA network analysis and Random Forest (RF) algorithm, which aimed to provide new insights into the molecular mechanisms of gout, and potential molecular targets for future therapeutic strategies of gout. Initially, we conducted an in-depth bioinformatics analysis of gout microarray chips to screen the key cuproptosis-related genes (CRGs) and key ferroptosis-related genes (FRGs). Using these data, we constructed a key ceRNA network for gout. Finally, key lncRNAs associated with gout were identified through the RF algorithm combined with ROC curves, and validated using the Comparative Toxicogenomics Database (CTD). We successfully identified NLRP3, LIPT1, and DBT as key CRGs associated with gout, and G6PD, PRKAA1, LIG3, PHF21A, KLF2, PGRMC1, JUN, PANX2, and AR as key FRGs associated with gout. The key ceRNA network identified four downregulated key lncRNAs (SEPSECS-AS1, LINC01054, REV3L-IT1, and ZNF883) along with three downregulated mRNAs (DBT, AR, and PRKAA1) based on the ceRNA theory. According to CTD validation inference scores and biological functions of target mRNAs, we identified a potential gout-associated lncRNA ZNF883/hsa-miR-539-5p/PRKAA1 regulatory axis. This study identified the key lncRNA ZNF883 in the context of copper death and iron death mechanisms related to gout for the first time through the application of ceRNA network analysis and the RF algorithm, thereby filling a research gap in this field and providing new insights into the molecular mechanisms of gout. We further found that lncRNA ZNF883 might function in gout patients by regulating PRKAA1, the mechanism of which was potentially related to uric acid reabsorption in the proximal renal tubules and inflammation regulation. The proposed lncRNA ZNF883/hsa-miR-539-5p/PRKAA1 regulatory axis might represent a potential RNA regulatory pathway for controlling the progression of gout disease. This discovery offered new molecular targets for the treatment of gout, and had significant implications for future therapeutic strategies in managing the gout.















Similar content being viewed by others
Data Availability
The primary and processed data used in analysis can be downloaded from GEO public database (https://www.ncbi.nlm.nih.gov/geo/) (accessed on 20 June 2023).
References
Dalbeth, N., Gosling, A. L., Gaffo, A., & Abhishek, A. (2021). Gout. Lancet, 397, 1843–1855.
Latourte, A., Dumurgier, J., Paquet, C., & Richette, P. (2021). Hyperuricemia, gout, and the brain-an update. Current Rheumatology Reports, 23, 82.
Dehlin, M., Jacobsson, L., & Roddy, E. (2020). Global epidemiology of gout: Prevalence, incidence, treatment patterns and risk factors. Nature Reviews. Rheumatology, 16, 380–390.
Bridges, M. C., Daulagala, A. C., & Kourtidis, A. (2021). LNCcation: lncRNA localization and function. Journal of Cell Biology, 220, e202009045.
Bao, M. H., Luo, H. Q., Chen, L. H., Tang, L., Ma, K. F., Xiang, J., Dong, L. P., Zeng, J., Li, G. Y., & Li, J. M. (2016). Impact of high fat diet on long non-coding RNAs and messenger RNAs expression in the aortas of ApoE(-/-) mice. Scientific Reports, 6, 34161.
Zhou, X., Lu, J., Wu, B., & Guo, Z. (2022). HOXA11-AS facilitates the proliferation, cell cycle process and migration of keloid fibroblasts through sponging miR-188-5p to regulate VEGFA. Journal of Dermatological Science, 106, 111–118.
Zhou, L., Zhang, Q., Deng, H., Ou, S., Liang, T., & Zhou, J. (2022). The SNHG1-centered ceRNA network regulates cell cycle and Is a potential prognostic biomarker for hepatocellular carcinoma. Tohoku Journal of Experimental Medicine, 258, 265–276.
Shao, P., Liu, H., Xue, Y., Xiang, T., & Sun, Z. (2023). LncRNA HOTTIP promotes inflammatory response in acute gouty arthritis via miR-101-3p/BRD4 axis. International Journal of Rheumatic Diseases, 26, 305–315.
Lee, C. P., Huang, Y. N., Nithiyanantham, S., Huang, C. M., & Ko, Y. C. (2019). LncRNA-Jak3:Jak3 coexpressed pattern regulates monosodium urate crystal-induced osteoclast differentiation through Nfatc1/Ctsk expression. Environmental Toxicology, 34, 179–187.
Kong, R., & Sun, G. (2023). Targeting copper metabolism: a promising strategy for cancer treatment. Frontiers in Pharmacology, 14, 1203447.
Jiang, X., Stockwell, B. R., & Conrad, M. (2021). Ferroptosis: mechanisms, biology and role in disease. Nature Reviews. Molecular Cell Biology, 22, 266–282.
Chang, S., Tang, M., Zhang, B., Xiang, D., & Li, F. (2022). Ferroptosis in inflammatory arthritis: A promising future. Frontiers in Immunology, 13, 955069.
Tsvetkov, P., Coy, S., Petrova, B., Dreishpoon, M., Verma, A., Abdusamad, M., Rossen, J., Joesch-Cohen, L., Humeidi, R., Spangler, R. D., Eaton, J. K., Frenkel, E., Kocak, M., Corsello, S. M., Lutsenko, S., Kanarek, N., Santagata, S., & Golub, T. R. (2022). Copper induces cell death by targeting lipoylated TCA cycle proteins. Science, 375, 1254–1261.
Chen, X., Kang, R., Kroemer, G., & Tang, D. (2021). Broadening horizons: the role of ferroptosis in cancer. Nature Reviews. Clinical Oncology, 18, 280–296.
Tay, Y., Rinn, J., & Pandolfi, P. P. (2014). The multilayered complexity of ceRNA crosstalk and competition. Nature, 505, 344–352.
Chen, H., Jiang, R., Huang, W., Chen, K., Zeng, R., Wu, H., Yang, Q., Guo, K., Li, J., Wei, R., Liao, S., Tse, H. F., Sha, W., & Zhuo, Z. (2022). Identification of energy metabolism-related biomarkers for risk prediction of heart failure patients using random forest algorithm. Frontiers in Cardiovascular Medicine, 9, 993142.
Xu, Y. T., Leng, Y. R., Liu, M. M., Dong, R. F., Bian, J., Yuan, L. L., Zhang, J. G., Xia, Y. Z., & Kong, L. Y. (2020). MicroRNA and long noncoding RNA involvement in gout and prospects for treatment. International Immunopharmacology, 87, 106842.
Wang, W., Chen, Z., & Hua, Y. (2023). Bioinformatics Prediction and Experimental Validation Identify a Novel Cuproptosis-Related Gene Signature in Human Synovial Inflammation during Osteoarthritis Progression. Biomolecules, 13, 127.
Agius, L., Chachra, S. S., & Ford, B. E. (2020). The protective role of the carbohydrate response element binding protein in the liver: The metabolite perspective. Frontiers in Endocrinology, 11, 594041.
Ben Salem, C., Slim, R., Fathallah, N., & Hmouda, H. (2017). Drug-induced hyperuricaemia and gout. Rheumatology (Oxford, England), 56, 679–688.
Yalimaimaiti, S., Liang, X., Zhao, H., Dou, H., Liu, W., Yang, Y., & Ning, L. (2023). Establishment of a prognostic signature for lung adenocarcinoma using cuproptosis-related lncRNAs. BMC Bioinformatics, 24, 81.
Cassandri, M., Smirnov, A., Novelli, F., Pitolli, C., Agostini, M., Malewicz, M., Melino, G., & Raschellà, G. (2017). Zinc-finger proteins in health and disease. Cell Death Discovery, 3, 17071.
Gong, L., Han, Y., Chen, R., Yang, P., & Zhang, C. (2022). LncRNA ZNF883-Mediated NLRP3 Inflammasome activation and epilepsy development involve USP47 Upregulation. Molecular Neurobiology, 59, 5207–5221.
Gong, L., Yang, P., Hu, L., & Zhang, C. (2020). MiR-181b suppresses the progression of epilepsy by regulation of lncRNA ZNF883. American Journal of Translational Research, 12, 2769–2780.
Li, Y., Yao, M., Wu, T., Zhang, L., Wang, Y., Chen, L., Fu, G., Weng, X., & Wang, J. (2019). Loss of hypermethylated in cancer 1 (HIC1) promotes lung cancer progression. Cellular Signalling, 53, 162–169.
Wang, X., Han, L., Zhou, L., Wang, L., & Zhang, L. M. (2018). Prediction of candidate RNA signatures for recurrent ovarian cancer prognosis by the construction of an integrated competing endogenous RNA network. Oncology Reports, 40, 2659–2673.
Sun, W. F., Zhu, M. M., Li, J., Zhang, X. X., Liu, Y. W., Wu, X. R., & Liu, Z. G. (2015). Effects of Xie-Zhuo-Chu-Bi-Fang on miR-34a and URAT1 and their relationship in hyperuricemic mice. Journal of Ethnopharmacology, 161, 163–169.
Zhou, Z., Dong, Y., Zhou, H., Liu, J., & Zhao, W. (2019). MiR-143-3p directly targets GLUT9 to reduce uric acid reabsorption and inflammatory response of renal tubular epithelial cells. Biochemical and Biophysical Research Communications, 517, 413–420.
Hu, J., Wu, H., Wang, D., Yang, Z., & Dong, J. (2019). LncRNA ANRIL promotes NLRP3 inflammasome activation in uric acid nephropathy through miR-122-5p/BRCC3 axis. Biochimie, 157, 102–110.
Hainer, B. L., Matheson, E., & Wilkes, R. T. (2014). Diagnosis, treatment, and prevention of gout. American Family Physician, 90, 831–836.
Yang, L., Liu, Y., Zu, Y. G., Zhao, C. J., Zhang, L., Chen, X. Q., & Zhang, Z. H. J. C. E. J. (2011). Optimize the process of ionic liquid-based ultrasonic-assisted extraction of aesculin and aesculetin from Cortex fraxini by response surface methodology. Chemical Engineering Journal, 175, 539–547.
Preitner, F., Bonny, O., Laverrière, A., Rotman, S., Firsov, D., Da Costa, A., Metref, S., & Thorens, B. (2009). Glut9 is a major regulator of urate homeostasis and its genetic inactivation induces hyperuricosuria and urate nephropathy. Proceedings of the National Academy of Sciences of the United States of America, 106, 15501–15506.
Zhou, Y., Zhang, X., Li, C., Yuan, X., Han, L., Li, Z., Tan, X., Song, J., Wang, G., Jia, X., Feng, L., Qiao, X., & Liu, J. (2018). Research on the pharmacodynamics and mechanism of Fraxini Cortex on hyperuricemia based on the regulation of URAT1 and GLUT9. Biomedicine Pharmacotherapy = Biomedecine Pharmacotherapie, 106, 434–442.
Acknowledgements
Not applicable.
Funding
This work was supported by the National Natural Science Foundation of China (Grant No. 82060871); Jiangxi Provincial Natural Science Foundation (Grant No. 20232ACB206051); Key Research Laboratory and Clinical Research Base construction project of Jiangxi Provincial Administration of Traditional Chinese Medicine (Third Batch) [No. Gan Traditional Chinese Medicine Science and Education (2022) No. 8]; and Traditional Chinese Medicine Dominant Disease Specialty Project—Gout [No. Gan Traditional Chinese Medicine Comprehensive (2022) No. 4].
Author information
Authors and Affiliations
Contributions
Zichen Shao contributed toward conceptualization, methodology, formal analysis, writing—Original Draft, and visualization. Weikang Sun contributed toward validation. Qinqin Deng contributed toward data Curation. Ling Cheng contributed toward writing—Review & Editing. Xin Huang contributed toward writing— review & editing. Liekui Hu contributed toward writing—review & editing. Huanan Li contributed toward funding acquisition. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interests
All authors declare that they have no conflict of interests.
Ethics Approval
Not applicable.
Consent for Publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Shao, ZC., Sun, WK., Deng, QQ. et al. Identification of Key lncRNAs in Gout Under Copper Death and Iron Death Mechanisms: A Study Based on ceRNA Network Analysis and Random Forest Algorithm. Mol Biotechnol (2024). https://doi.org/10.1007/s12033-024-01099-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12033-024-01099-5