Abstract
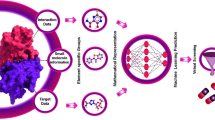
Varicella zoster virus (VZV) infection causes severe disease such as chickenpox, shingles, and postherpetic neuralgia, often leading to disability. Reactivation of latent VZV is associated with a decrease in specific cellular immunity in the elderly and in patients with immunodeficiency. However, due to the limited efficacy of existing therapy and the emergence of antiviral resistance, it has become necessary to develop new and effective antiviral drugs for the treatment of diseases caused by VZV, particularly in the setting of opportunistic infections. The goal of this work is to identify potent oxazole derivatives as anti-VZV agents by machine learning, followed by their synthesis and experimental validation. Predictive QSAR models were developed using the Online Chemical Modeling Environment (OCHEM). Data on compounds exhibiting antiviral activity were collected from the ChEMBL and uploaded in the OCHEM database. The predictive ability of the models was tested by cross-validation, giving coefficient of determination q2 = 0.87–0.9. The validation of the models using an external test set proves that the models can be used to predict the antiviral activity of newly designed and known compounds with reasonable accuracy within the applicability domain (q2 = 0.83–0.84). The models were applied to screen a virtual chemical library with expected activity of compounds against VZV. The 7 most promising oxazole derivatives were identified, synthesized, and tested. Two of them showed activity against the VZV Ellen strain upon primary in vitro antiviral screening. The synthesized compounds may represent an interesting starting point for further development of the oxazole derivatives against VZV. The developed models are available online at OCHEM http://ochem.eu/article/145978 and can be used to virtually screen for potential compounds with anti-VZV activity.





Similar content being viewed by others
Data Availability
The data used in this work and developed models are freely available online at OCHEM https://ochem.eu/article/145978
References
Johnson, R. W. (2010). Herpes zoster and postherpetic neuralgia. Expert Review of Vaccines, 9(3), 21–26. https://doi.org/10.1586/erv.10.30
Floß, N., & Dolff, S. (2019). Opportunistische Infektionen durch humane Herpesviren [Opportunistic infections by human herpes viruses]. Der Internist, 60(7), 678–683. https://doi.org/10.1007/s00108-019-0615-6
Andrei, G., & Snoeck, R. (2021). Advances and perspectives in the management of Varicella-Zoster Virus infections. Molecules, 26(4), 1132. https://doi.org/10.3390/molecules26041132
Kaplan, J. E., Benson, C., Holmes, K. K., Brooks, J. T., Pau, A., & Masur, H., Centers for Disease Control and Prevention (CDC), National Institutes of Health, & HIV Medicine Association of the Infectious Diseases Society of America. (2009). Guidelines for prevention and treatment of opportunistic infections in HIV-infected adults and adolescents: recommendations from CDC, the National Institutes of Health, and the HIV Medicine Association of the Infectious Diseases Society of America. MMWR. Recommendations and reports : Morbidity and mortality weekly report. Recommendations and reports, 58(RR-4), 1–207.
Panggabean, J. A., Adiguna, S. P., Rahmawati, S. I., Ahmadi, P., Zainuddin, E. N., Bayu, A., & Putra, M. Y. (2022). Antiviral activities of algal-based sulfated polysaccharides. Molecules, 27, 1178. https://doi.org/10.3390/molecules27041178
Lau, Z. L., Low, S. S., Ezeigwe, E. R., Chew, K. W., Chai, W. S., Bhatnagar, A., Yap, Y. J., & Show, P. L. (2022). A review on the diverse interactions between microalgae and nanomaterials: Growth variation, photosynthetic performance and toxicity. Bioresource technology, 351, 127048. https://doi.org/10.1016/j.biortech.2022.127048
Costa, J. A. V., Moreira, J. B., Fanka, L. S., da Kosinski, R. C., & de Morais, M. G. (2020). Microalgal biotechnology applied in biomedicine. In O. Konur (Ed.), Handbook of algal science technology and medicine (pp. 429–439). Elsevier.
Chan, S. S., Low, S. S., Chew, K. W., Ling, T. C., Rinklebe, J., Juan, J. C., Ng, E. P., & Show, P. L. (2022). Prospects and environmental sustainability of phyconanotechnology: A review on algae-mediated metal nanoparticles synthesis and mechanism. Environmental research, 212(pt A), 113140. https://doi.org/10.1016/j.envres.2022.113140
Huleihel, M., Ishanu, V., Tal, J., & Arad, S. (2001). Antiviral effect of red microalgal polysaccharides on herpes simplex and Varicella zoster viruses. Journal of applied phycology, 13, 127–134.
Lanh, P. T., Nguyen, H. M., Duong, B. T. T., Hoa, N. T., Thom, L. T., Tam, L. T., Thu, H. T., Nha, V. V., Hong, D. D., Mouradov, A., Koyande, A. K., Show, P.-L., & Van Quyen, D. (2021). Generation of microalga chlamydomonas reinhardtii expressing Vp28 protein as oral vaccine candidate for shrimps against White Spot Syndrome Virus (wssv) infection. Aquaculture, 540, 736737. https://doi.org/10.1016/j.aquaculture.2021.736737
Kozlovskaya, L. I., Andrei, G., Orlov, A. A., Khvatov, E. V., Koruchekov, A. A., Belyaev, E. S., Nikolaev, E. N., Korshun, V. A., Snoeck, R., Osolodkin, D. I., Matyugina, E. S., & Aralov, A. V. (2019). Antiviral activity spectrum of phenoxazine nucleoside derivatives. Antiviral research, 163, 117–124. https://doi.org/10.1016/j.antiviral.2019.01.010
Liu, C., Chen, Q., Yang, M., & Schneller, S. W. (2013). C-3 halo and 3-methyl substituted 5’-nor-3-deazaaristeromycins: Synthesis and antiviral properties. Bioorganic & medicinal chemistry, 21(1), 359–364. https://doi.org/10.1016/j.bmc.2012.09.051
Baszczyňski, O., Kaiser, M. M., Česnek, M., Břehová, P., Jansa, P., Procházková, E., Dračínský, M., Snoeck, R., Andrei, G., & Janeba, Z. (2018). Xanthine-based acyclic nucleoside phosphonates with potent antiviral activity against varicella-zoster virus and human cytomegalovirus. Antiviral chemistry & chemotherapy, 26, 2040206618813050. https://doi.org/10.1177/2040206618813050
Wang, M., Srivastava, P., Liu, C., Snoeck, R., Andrei, G., De Jonghe, S., & Herdewijn, P. (2018). Synthesis and antiviral evaluation of cyclopentyl nucleoside phosphonates. European journal of medicinal chemistry, 150, 616–625. https://doi.org/10.1016/j.ejmech.2018.03.008
Musella, S., di Sarno, V., Ciaglia, T., Sala, M., Spensiero, A., Scala, M. C., Ostacolo, C., Andrei, G., Balzarini, J., Snoeck, R., Novellino, E., Campiglia, P., Bertamino, A., & Gomez-Monterrey, I. M. (2016). Identification of an indol-based derivative as potent and selective Varicella Zoster Virus (VZV) inhibitor. European journal of medicinal chemistry, 124, 773–781. https://doi.org/10.1016/j.ejmech.2016.09.014
Metelytsia, L. O., Trush, M. M., Kovalishyn, V. V., Hodyna, D. M., Kachaeva, M. V., Brovarets, V. S., Pilyo, S. G., Sukhoveev, V. V., Tsyhankov, S. A., Blagodatnyi, V. M., & Semenyuta, I. V. (2021). 1,3-Oxazole derivatives of cytisine as potential inhibitors of glutathione reductase of Candida spp.: QSAR modeling, docking analysis and experimental study of new anti-Candida agents. Computational biology and chemistry, 90, 107407. https://doi.org/10.1016/j.compbiolchem.2020.107407
Semenyuta, I., Kovalishyn, V., Tanchuk, V., Pilyo, S., Zyabrev, V., Blagodatnyy, V., Trokhimenko, O., Brovarets, V., & Metelytsia, L. (2016). 1,3-Oxazole derivatives as potential anticancer agents: Computer modeling and experimental study. Computational biology and chemistry, 65, 8–15. https://doi.org/10.1016/j.compbiolchem.2016.09.012
Swellmeen, L. (2016). 1,3-oxazole derivatives: A review of biological activities as antipathogenic. Der Pharma Chemica, 8, 269–286.
Kovalishyn, V., Zyabrev, V., Kachaeva, M., Ziabrev, K., Keith, K., Harden, E., Hartline, C., James, S. H., & Brovarets, V. (2021). Design of new imidazole derivatives with anti-HCMV activity: QSAR modeling, synthesis and biological testing. Journal of computer-aided molecular design, 35(12), 1177–1187. https://doi.org/10.1007/s10822-021-00428-z
Kovalishyn, V., Grouleff, J., Semenyuta, I., Sinenko, V. O., Slivchuk, S. R., Hodyna, D., Brovarets, V., Blagodatny, V., Poda, G., Tetko, I. V., & Metelytsia, L. (2018). Rational design of isonicotinic acid hydrazide derivatives with antitubercular activity: Machine learning, molecular docking, synthesis and biological testing. Chemical biology & drug design, 92(1), 1272–1278. https://doi.org/10.1111/cbdd.13188
Kachaeva, M. V., Pilyo, S. G., Hartline, C. B., Harden, E. A., Prichard, M. N., Zhirnov, V. V., & Brovarets, V. S. (2019). In vitro activity of novel derivatives of 1,3-oxazole-4-carboxylate and 1,3-oxazole-4-carbonitrile against human cytomegalovirus. Medicinal chemistry research, 28, 1205–1211. https://doi.org/10.1007/s00044-019-02365-x
Abdurakhmanova, E. R., Brusnakov, M. Y., Golovchenko, O. V., Pilyo, S. G., Velychko, N. V., Harden, E. A., Prichard, M. N., James, S. H., Zhirnov, V. V., & Brovarets, V. S. (2020). Synthesis and in vitro anticytomegalovirus activity of 5-hydroxyalkylamino-1,3-oxazoles derivatives. Medicinal chemistry research., 29, 1669–1675. https://doi.org/10.1007/s00044-020-02593-6
Sushko, I., Novotarskyi, S., Körner, R., et al. (2011). Online chemical modeling environment (OCHEM): Web platform for data storage, model development and publishing of chemical information. Journal of computer-aided molecular design, 25(6), 533–554. https://doi.org/10.1007/s10822-011-9440-2
Karpov, P., Godin, G., & Tetko, I. V. (2020). Transformer-CNN: Swiss knife for QSAR modeling and interpretation. Journal of cheminformatics, 12(1), 17. https://doi.org/10.1186/s13321-020-00423-w
Tetko, I. V., Karpov, P., Bruno, E., Kimber, T. B., & Godin, G. (2019). Augmentation Is What You Need! In I. Tetko, V. Kůrková, P. Karpov, & F. Theis (Eds.), Lecture notes in computer science (pp. 831–835). Springer. https://doi.org/10.1007/978-3-030-30493-5_79
Tetko, I. V., Sushko, I., Pandey, A. K., Zhu, H., Tropsha, A., Papa, E., Oberg, T., Todeschini, R., Fourches, D., & Varnek, A. (2008). Critical assessment of QSAR models of environmental toxicity against Tetrahymena pyriformis: Focusing on applicability domain and overfitting by variable selection. Journal of chemical information and modeling, 48(9), 1733–1746. https://doi.org/10.1021/ci800151m
Jonhos, N. N., & Leone, F. G. (1977). Statistic and experimental design in engineering and the physical sciences. John Wiley & Sons.
Sushko, I., Novotarskyi, S., Körner, R., Pandey, A. K., Kovalishyn, V. V., Prokopenko, V. V., & Tetko, I. V. (2010). Applicability domain for in silico models to achieve accuracy of experimental measurements. Journal of chemometrics, 24, 202–208. https://doi.org/10.1002/cem.1296
Hartline, C. B., Keith, K. A., Eagar, J., Harden, E. A., Bowlin, T. L., & Prichard, M. N. (2018). A standardized approach to the evaluation of antivirals against DNA viruses: orthopox-, adeno-, and herpesviruses. Antiviral research, 159, 104–112. https://doi.org/10.1016/j.antiviral.2018.09.015
Hochscherf, J., Lindenblatt, D., Witulski, B., Birus, R., Aichele, D., Marminon, C., Bouaziz, Z., Le Borgne, M., Jose, J., & Niefind, K. (2017). Unexpected binding mode of a potent indeno[1,2-b]indole-type inhibitor of protein kinase CK2 revealed by complex structures with the catalytic subunit CK2α and Its paralog CK2α’. Pharmaceuticals (Basel, Switzerland), 10(4), 98. https://doi.org/10.3390/ph10040098
Brown, N. R., Korolchuk, S., Martin, M. P., Stanley, W. A., Moukhametzianov, R., Noble, M., & Endicott, J. A. (2015). CDK1 structures reveal conserved and unique features of the essential cell cycle CDK. Nature communications, 6, 6769. https://doi.org/10.1038/ncomms7769
Mapelli, M., Massimiliano, L., Crovace, C., Seeliger, M. A., Tsai, L. H., Meijer, L., & Musacchio, A. (2005). Mechanism of CDK5/p25 binding by CDK inhibitors. Journal of medicinal chemistry, 48(3), 671–679. https://doi.org/10.1021/jm049323m
Berman, H. M., Westbrook, J., Feng, Z., Gilliland, G., Bhat, T. N., Weissig, H., Shindyalov, I. N., & Bourne, P. E. (2000). The protein data bank. Nucleic acids research, 28(1), 235–242. https://doi.org/10.1093/nar/28.1.235
Trott, O., & Olson, A. J. (2010). AutoDock Vina: Improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading. Journal of computational chemistry, 31(2), 455–461. https://doi.org/10.1002/jcc.21334
Sanner, M. F. (1999). Python: A programming language for software integration and development. Journal of molecular graphics & modelling, 17(1), 57–61.
Sushko, I., Salmina, E., Potemkin, V. A., Poda, G., & Tetko, I. V. (2012). ToxAlerts: A web server of structural alerts for toxic chemicals and compounds with potential adverse reactions. Journal of chemical information and modeling, 52(8), 2310–2316. https://doi.org/10.1021/ci300245q
Babii, S. B., Zyabrev, V. S., & Drach, B. S. (2001). Conversion of N-(1-Arylsulfonyl-2,2-dichloroethenyl)carboxamides into derivatives of 4,5-Dimercaptooxazole. Russian journal of organic chemistry, 37, 1149–1152. https://doi.org/10.1023/A:1013196415680
Kachaeva, M. V., Hodyna, D. M., Obernikhina, N. V., Pilyo, S. G., Kovalenko, Y. S., Prokopenko, V. M., Kachkovsky, O. D., & Brovarets, V. S. (2019). Design, synthesis and evaluation of novel sulfonamides as potential anticancer agents. Journal of heterocyclic chemistry, 56, 3122–3134. https://doi.org/10.1016/j.compbiolchem.2018.04.006
Lipinski, C. A., Lombardo, F., Dominy, B. W., & Feeney, P. J. (2001). Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Advanced drug delivery reviews, 46(1–3), 3–26. https://doi.org/10.1016/s0169-409x(00)00129-0
Pires, D. E., Blundell, T. L., & Ascher, D. B. (2015). pkCSM: Predicting small-molecule pharmacokinetic and toxicity properties using graph-based signatures. Journal of medicinal chemistry, 58(9), 4066–4072. https://doi.org/10.1021/acs.jmedchem.5b00104
Andrei, G., & Snoeck, R. (2013). Advances in the treatment of varicella-zoster virus infections. Advances in pharmacology, 67, 107–168. https://doi.org/10.1016/B978-0-12-405880-4.00004-4
De, S. K., Hart, J. C., & Breuer, J. (2015). Herpes simplex virus and varicella zoster virus: Recent advances in therapy. Current opinion in infectious diseases, 28(6), 589–595. https://doi.org/10.1097/QCO.0000000000000211
Qiu, X., Janson, C. A., Culp, J. S., Richardson, S. B., Debouck, C., Smith, W. W., & Abdel-Meguid, S. S. (1997). Crystal structure of varicella-zoster virus protease. Proceedings of the National Academy of Sciences of the United States of America, 94(7), 2874–2879. https://doi.org/10.1073/pnas.94.7.2874
Leisenfelder, S. A., & Moffat, J. F. (2006). Varicella-zoster virus infection of human foreskin fibroblast cells results in atypical cyclin expression and cyclin-dependent kinase activity. Journal of virology, 80(11), 5577–5587. https://doi.org/10.1128/JVI.00163-06
Habran, L., Bontems, S., Di Valentin, E., Sadzot-Delvaux, C., & Piette, J. (2005). Varicella-zoster virus IE63 protein phosphorylation by roscovitine-sensitive cyclin-dependent kinases modulates its cellular localization and activity. The Journal of biological chemistry, 280(32), 29135–29143. https://doi.org/10.1074/jbc.M503312200
Bontems, S., Di Valentin, E., Baudoux, L., Rentier, B., Sadzot-Delvaux, C., & Piette, J. (2002). Phosphorylation of varicella-zoster virus IE63 protein by casein kinases influences its cellular localization and gene regulation activity. The Journal of biological chemistry, 277(23), 21050–21060. https://doi.org/10.1074/jbc.M111872200
Leisenfelder, S. A., Kinchington, P. R., & Moffat, J. F. (2008). Cyclin-dependent kinase 1/cyclin B1 phosphorylates varicella-zoster virus IE62 and is incorporated into virions. Journal of virology, 82(24), 12116–12125. https://doi.org/10.1128/JVI.00153-08
Grose, C., Jackson, W., & Traugh, J. A. (1989). Phosphorylation of varicella-zoster virus glycoprotein gpI by mammalian casein kinase II and casein kinase I. Journal of virology, 63(9), 3912–3918. https://doi.org/10.1128/JVI.63.9.3912-3918.1989
Ye, M., Duus, K. M., Peng, J., Price, D. H., & Grose, C. (1999). Varicella-zoster virus Fc receptor component gI is phosphorylated on its endodomain by a cyclin-dependent kinase. Journal of virology, 73(2), 1320–1330. https://doi.org/10.1128/JVI.73.2.1320-1330.1999
Olson, J. K., Bishop, G. A., & Grose, C. (1997). Varicella-zoster virus Fc receptor gE glycoprotein: Serine/threonine and tyrosine phosphorylation of monomeric and dimeric forms. Journal of virology, 71(1), 110–119. https://doi.org/10.1128/JVI.71.1.110-119.1997
Taylor, S. L., Kinchington, P. R., Brooks, A., & Moffat, J. F. (2004). Roscovitine, a cyclin-dependent kinase inhibitor, prevents replication of varicella-zoster virus. Journal of virology, 78(6), 2853–2862. https://doi.org/10.1128/jvi.78.6.2853-2862.2004
Acknowledgements
These studies were funded in part with Federal funds from the National Institute of Allergy and Infectious Diseases, National Institutes of Health, Department of Health and Human Services, under Contract No. HHSN75N93019D00016 (SHJ). This work was also supported by the National Research Foundation of Ukraine (Grant number 2020.01/0075) and by the NAS of Ukraine.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare that are relevant to the content of this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kovalishyn, V., Severin, O., Kachaeva, M. et al. In Silico Design and Experimental Validation of Novel Oxazole Derivatives Against Varicella zoster virus. Mol Biotechnol 66, 707–717 (2024). https://doi.org/10.1007/s12033-023-00670-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12033-023-00670-w