Abstract
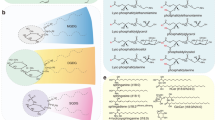
Salvia hispanica (chia) is the highest reported terrestrial plant source of alpha-linolenic acid (ALA, ~ 65%), an ω-3 polyunsaturated fatty acid with numerous health benefits. The molecular basis of high ALA accumulation in chia is yet to be understood. We have identified lysophosphatidylcholine acyltransferase (LPCAT) gene from the developing seed transcriptome data of chia and carried out its biochemical characterization through heterologous expression in Saccharomyces cerevisiae. Expression profiling showed that the enzyme was active throughout the seed development, indicating a pivotal role in oil biosynthesis. The enzyme could utilize both saturated and unsaturated lysophosphatidylcholine substrates at the same rate, to synthesize phosphatidylcholine (PC). The enzyme also exhibited lysophosphatidic acid acyltransferase (LPAAT) activity, by preferring lysophosphatidic acid substrate. Substrate specificity studies showed that the enzyme preferred both monounsaturated and polyunsaturated fatty acyl CoAs over saturated CoAs. This activity may play a key role in enriching the PC fraction with polyunsaturated fatty acids (PUFAs). PUFAs present on PC can be transferred to oil through the action of other acyltransferases. Our results describe a new LPCAT enzyme that can be used to biotechnologically alter oilseed crops to incorporate more PUFA in its seed oil.






Similar content being viewed by others
References
Exton, J. H. (1990). Signaling through phosphatidylcholine breakdown. Journal of Biological Chemistry, 265(1), 1–4.
Kent, C., & Carman, G. M. (1999). Interactions among pathways for phosphatidylcholine metabolism, CTP synthesis and secretion through the Golgi apparatus. Trends in biochemical sciences, 24(4), 146–150.
Bates, P. D., & Browse, J. (2011). The pathway of triacylglycerol synthesis through phosphatidylcholine in Arabidopsis produces a bottleneck for the accumulation of unusual fatty acids in transgenic seeds. The Plant Journal, 68(3), 387–399.
Myher, J. J., Kuksis, A., & Pind, S. (1989). Molecular species of glycerophospholipids and sphingomyelins of human erythrocytes: Improved method of analysis. Lipids, 24(5), 396–407.
Bao, X., Pollard, M., & Ohlrogge, J. (1998). The biosynthesis of erucic acid in developing embryos of Brassica rapa. Plant physiology, 118(1), 183–190.
Lands, W. E. (1965). Lipid metabolism. Annual review of biochemistry, 34(1), 313–346.
Stymne, S., & Stobart, A. K. (1984). Evidence for the reversibility of the acyl-CoA: Lysophosphatidylcholine acyltransferase in microsomal preparations from developing safflower (Carthamus tinctorius L.) cotyledons and rat liver. Biochemical Journal, 223(2), 305–314.
Chen, Q., Kazachkov, M., Zheng, Z., & Zou, J. (2007). The yeast acylglycerol acyltransferase LCA1 is a key component of Lands cycle for phosphatidylcholine turnover. FEBS letters, 581(28), 5511–5516.
Kazachkov, M., Chen, Q., Wang, L., & Zou, J. (2008). Substrate preferences of a lysophosphatidylcholine acyltransferase highlight its role in phospholipid remodeling. Lipids, 43(10), 895–902.
Zhao, Y., Chen, Y. Q., Bonacci, T. M., Bredt, D. S., Li, S., Bensch, W. R., Moller, D. E., Kowala, M., Konrad, R. J., & Cao, G. (2008). Identification and characterization of a major liver lysophosphatidylcholine acyltransferase. Journal of Biological Chemistry, 283(13), 8258–8265.
Harayama, T., Shindou, H., Ogasawara, R., Suwabe, A., & Shimizu, T. (2008). Identification of a novel noninflammatory biosynthetic pathway of platelet-activating factor. Journal of Biological Chemistry, 283(17), 11097–11106.
Wang, L., Shen, W., Kazachkov, M., Chen, G., Chen, Q., Carlsson, A. S., Stymne, S., Weselake, R. J., & Zou, J. (2012). Metabolic interactions between the lands cycle and the Kennedy pathway of glycerolipid synthesis in Arabidopsis developing seeds. The Plant Cell, 24(11), 4652–4669.
Zheng, Q., Li, J. Q., Kazachkov, M., Liu, K., & Zou, J. (2012). Identification of Brassica napus lysophosphatidylcholine acyltransferase genes through yeast functional screening. Phytochemistry, 75, 21–31.
Zhang, D., Jasieniecka-Gazarkiewicz, K., Wan, X., Luo, L., Zhang, Y., Banas, A., Jiang, M., & Gong, Y. (2015). Molecular characterization of two lysophospholipid: Acyl-CoA acyltransferases belonging to the MBOAT family in Nicotiana benthamiana. PLoS ONE. https://doi.org/10.1371/journal.pone.0144653
Arroyo-Caro, J. M., Chileh, T., Alonso, D. L., & García-Maroto, F. (2013). Molecular characterization of a lysophosphatidylcholine acyltransferase gene belonging to the MBOAT family in Ricinus communis L. Lipids, 48(7), 663–674.
Xu, J., Carlsson, A. S., Francis, T., Zhang, M., Hoffman, T., Giblin, M. E., & Taylor, D. C. (2012). Triacylglycerol synthesis by PDAT1 in the absence of DGAT1 activity is dependent on re-acylation of LPC by LPCAT2. BMC plant biology, 12(1), 4.
Bates, P. D., Fatihi, A., Snapp, A. R., Carlsson, A. S., Browse, J., & Lu, C. (2012). Acyl editing and headgroup exchange are the major mechanisms that direct polyunsaturated fatty acid flux into triacylglycerols. Plant physiology, 160(3), 1530–1539.
Sreedhar, R. V., Kumari, P., Rupwate, S. D., Rajasekharan, R., & Srinivasan, M. (2015). Exploring triacylglycerol biosynthetic pathway in developing seeds of Chia (Salvia hispanica L.): A transcriptomic approach. PLoS ONE. https://doi.org/10.1371/journal.pone.0123580
Gopalam, R., Rupwate, S. D., & Tumaney, A. W. (2017). Selection and validation of appropriate reference genes for quantitative real-time PCR analysis in Salvia hispanica. PLoS ONE, 12(11), e0186978.
Livak, K. J., & Schmittgen, T. D. (2001). Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods, 25(4), 402–408.
Gietz, R. D., & Schiestl, R. H. (2007). High-efficiency yeast transformation using the LiAc/SS carrier DNA/PEG method. Nature protocols, 2(1), 31–34.
Dahlqvist, A., Ståhl, U., Lenman, M., Banas, A., Lee, M., Sandager, L., Ronne, H., & Stymne, S. (2000). Phospholipid: Diacylglycerol acyltransferase: An enzyme that catalyzes the acyl-CoA-independent formation of triacylglycerol in yeast and plants. Proceedings of the National Academy of Sciences USA, 97(12), 6487–6492.
Marchler-Bauer, A., Bo, Y., Han, L., He, J., Lanczycki, C. J., Lu, S., Chitsaz, F., Derbyshire, M. K., Geer, R. C., Gonzales, N. R., & Gwadz, M. (2017). CDD/SPARCLE: Functional classification of proteins via subfamily domain architectures. Nucleic acids research, 45(D1), D200–D203.
Mapelli-Brahm, A., Sánchez, R., Pan, X., Moreno-Pérez, A. J., Garcés, R., Martínez-Force, E., Weselake, R. J., Salas, J. J., & Venegas-Calerón, M. (2020). Functional characterization of lysophosphatidylcholine: Acyl-CoA acyltransferase genes from sunflower (Helianthus annuus L.). Frontiers in Plant Science, 11, 403.
Venkateshwari, V., Vijayakumar, A., Vijayakumar, A. K., Reddy, L. P. A., Srinivasan, M., & Rajasekharan, R. (2018). Leaf lipidome and transcriptome profiling of Portulaca oleracea: Characterization of lysophosphatidylcholine acyltransferase. Planta, 248(2), 347–367.
Gopalam, R., & Tumaney, A. W. (2021). Functional characterization of acyltransferases from Salvia hispanica that can selectively catalyze the formation of trilinolenin. Phytochemistry, 186, 112712.
Zaremberg, V., & McMaster, C. R. (2002). Differential partitioning of lipids metabolized by separate yeast glycerol-3-phosphate acyltransferases reveals that phospholipase D generation of phosphatidic acid mediates sensitivity to choline-containing lysolipids and drugs. Journal of Biological Chemistry, 277(41), 39035–39044.
Ståhl, U., Stålberg, K., Stymne, S., & Ronne, H. (2008). A family of eukaryotic lysophospholipid acyltransferases with broad specificity. FEBS letters, 582(2), 305–309.
Lager, I., Yilmaz, J. L., Zhou, X. R., Jasieniecka, K., Kazachkov, M., Wang, P., & P., Zou, J., Weselake, R., Smith, M.A., Bayon, S & Dyer, J. M. . (2013). Plant acyl-CoA: Lysophosphatidylcholine acyltransferases (LPCATs) have different specificities in their forward and reverse reactions. Journal of Biological Chemistry, 288(52), 36902–36914.
Tocher, D. R. (1998). Recent advances in the biochemistry and molecular biology of fatty acyl desaturases. Progress in Lipid Research, 37, 73–117.
Stymne, S. T. E. N., & STOBART, A. K. (1987). Triacylglycerol biosynthesis. Lipids: structure and function (pp. 175–214). Academic Press.
Acknowledgements
The authors thank the Director, CSIR-CFTRI, Mysuru for the facilities provided. The research work is financially supported by DST-SERB, under the project (EEQ/2016/000531). Rahul Gopalam is thankful to CSIR-UGC, New Delhi for providing fellowship.
Author information
Authors and Affiliations
Contributions
RG and AWT planned and designed experiments. RG, AD and SB performed the experiments. RG, DC AWT analyzed the data; RG prepared the original draft; All the authors reviewed, edited and finalized the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Gopalam, R., Datey, A., Bijoor, S. et al. Biochemical Characterization of Acyl-CoA: Lysophosphatidylcholine Acyltransferase (LPCAT) Enzyme from the Seeds of Salvia hispanica. Mol Biotechnol 63, 963–972 (2021). https://doi.org/10.1007/s12033-021-00354-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12033-021-00354-3