Abstract
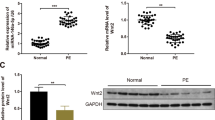
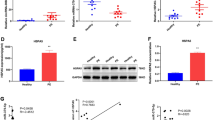
Preeclampsia (PE) is a pathologic condition in pregnant women which accounts for the inhibition of proliferation, migration and invasion of trophoblast cells. This study aimed to investigate the regulation of ubiquitin-specific peptidase 5 (USP5) on the trophoblast cells in PE. Expressions of USP5 in the placentas of PE patients and healthy donors were examined by qRT-PCR and Western blot. Hypoxia/reoxygenation (H/R) model in trophoblast cells was further established. Cell viability was examined using CCK-8 assay. Finally, the effect of overexpression and silence of USP5 using lentivirus transduction was studied. Our results showed that USP5 was lowly expressed in the placentas of PE patients as well as in H/R-induced trophoblast cells. In the experiments of overexpression, USP5 promoted the proliferation of trophoblast cells, and up-regulated the expressions of β-catenin and the downstream signals c-Myc and Cyclin D1 in trophoblast cells. On the other hand, silence of USP5 elicited the opposite results. The overexpression of USP5 in the H/R model greatly released the H/R-induced inhibition in the trophoblast cells, and moderated the down-regulation of β-catenin and c-Myc induced by H/R. We concluded that USP5 promoted the proliferation of trophoblast cells via the up-regulation of the Wnt/β-catenin signaling pathway.





Similar content being viewed by others
References
Filipek, A., & Jurewicz, E. (2018). Preeclampsia—A disease of pregnant women. Postepy Biochemii, 64, 232–229
Mol, B. W. J., Roberts, C. T., Thangaratinam, S., Magee, L. A., de Groot, C. J. M., & Hofmeyr, G. J. (2016). Pre-eclampsia. Lancet, 387, 999–1011
Poon, L. C., Shennan, A., Hyett, J. A., Kapur, A., Hadar, E., Divakar, H., McAuliffe, F., da Silva Costa, F., von Dadelszen, P., McIntyre, H. D., Kihara, A. B., Di Renzo, G. C., Romero, R., D’Alton, M., Berghella, V., Nicolaides, K. H., & Hod, M. (2019). The International Federation of Gynecology and Obstetrics (FIGO) initiative on pre-eclampsia: A pragmatic guide for first-trimester screening and prevention. International Journal of Gynaecology and Obstetrics, 145(Suppl 1), 1–33
Korkes, H. A., De Oliveira, L., Sass, N., Salahuddin, S., Karumanchi, S. A., & Rajakumar, A. (2017). Relationship between hypoxia and downstream pathogenic pathways in preeclampsia. Hypertension in Pregnancy, 36, 145–150
Chen, P. S., Chiu, W. T., Hsu, P. L., Lin, S. C., Peng, I. C., Wang, C. Y., & Tsai, S. J. (2020). Pathophysiological implications of hypoxia in human diseases. Journal of Biomedical Science, 27, 63
Tranquilli, A. L., Brown, M. A., Zeeman, G. G., Dekker, G., & Sibai, B. M. (2013). The definition of severe and early-onset preeclampsia. Statements from the International Society for the Study of Hypertension in Pregnancy (ISSHP). Pregnancy Hypertens, 3, 44–47
Myatt, L. (2002). Role of placenta in preeclampsia. Endocrine, 19, 103–111
Redman, C. W., & Sargent, I. L. (2005). Latest advances in understanding preeclampsia. Science, 308, 1592–1594
Zhang, Z., Wang, X., Zhang, L., Shi, Y., Wang, J., & Yan, H. (2017). Wnt/β-catenin signaling pathway in trophoblasts and abnormal activation in preeclampsia. Molecular Medicine Reports, 16, 1007–1013
Zhang, L., Song, Y., Ling, Z., Li, Y., Ren, X., Yang, J., Wang, Z., Xia, J., Zhang, W., & Cheng, B. (2019). R-spondin 2-LGR4 system regulates growth, migration and invasion, epithelial-mesenchymal transition and stem-like properties of tongue squamous cell carcinoma via Wnt/β-catenin signaling. eBioMedicine, 44, 275–288
Li, L., Peng, W., Zhou, Q., Wan, J. P., Wang, X. T., & Qi, H. B. (2020). LRP6 regulates Rab7-mediated autophagy through the Wnt/β-catenin pathway to modulate trophoblast cell migration and invasion. Journal of Cellular Biochemistry, 121, 1599–1609
Li, X. Y., Wu, H. Y., Mao, X. F., Jiang, L. X., & Wang, Y. X. (2017). USP5 promotes tumorigenesis and progression of pancreatic cancer by stabilizing FoxM1 protein. Biochemical and Biophysical Research Communications, 492, 48–54
Ma, X., Qi, W., Pan, H., Yang, F., & Deng, J. (2018). Overexpression of USP5 contributes to tumorigenesis in non-small cell lung cancer via the stabilization of β-catenin protein. American Journal of Cancer Research, 8, 2284–2295
Xu, X., Huang, A., Cui, X., Han, K., Hou, X., Wang, Q., Cui, L., & Yang, Y. (2019). Ubiquitin specific peptidase 5 regulates colorectal cancer cell growth by stabilizing Tu translation elongation factor. Theranostics, 9, 4208–4220
Du, Y., Lin, J., Zhang, R., Yang, W., Quan, H., Zang, L., Han, Y., Li, B., Sun, H., & Wu, J. (2019). Ubiquitin specific peptidase 5 promotes ovarian cancer cell proliferation through deubiquitinating HDAC2. Aging (Albany NY), 11, 9778–9793
Zhang, L., Li, H., Li, M., Zhang, W., Yang, Z., & Zhang, S. (2020). LRP6 is involved in the proliferation, migration and invasion of trophoblast cells via miR-346. International Journal of Molecular Medicine, 46, 211–223
Chen, L., Wang, J., Fan, X., Zhang, Y., Zhoua, M., Li, X., & Wang, L. (2021). LASP2 inhibits trophoblast cell migration and invasion in preeclampsia through inactivation of the Wnt/β-catenin signaling pathway. Journal of Receptor and Signal Transduction Research, 41, 67–73
Sheiner, E., Kapur, A., Retnakaran, R., Hadar, E., Poon, L. C., McIntyre, H. D., Divakar, H., Staff, A. C., Narula, J., Kihara, A. B., & Hod, M. (2019). FIGO (International Federation of Gynecology and Obstetrics) postpregnancy initiative: Long-term maternal implications of pregnancy complications-follow-up considerations. International Journal of Gynaecology and Obstetrics, 147(Suppl 1), 1–31
Boulanger, H., & Flamant, M. (2007). New insights in the pathophysiology of preeclampsia and potential therapeutic implications. Néphrologie & Thérapeutique, 3, 437–448
Wang, Z., Feng, W., & Liu, J. (2020) Current understanding of autoantibody against angiotensin II type 1 receptor in preeclampsia. J Matern Fetal Neonatal Med. https://doi.org/10.1080/14767058.2020.18467091-6
Phipps, E., Prasanna, D., Brima, W., & Jim, B. (2016). Preeclampsia: Updates in pathogenesis, definitions, and guidelines. Clinical Journal of the American Society of Nephrology, 11, 1102–1113
Kay, V. R., Wedel, N., & Smith, G. N. (2020). Family history of hypertension, cardiovascular disease, or diabetes and risk of developing preeclampsia: A systematic review. Journal of Obstetrics and Gynecology Canada., 43, 227–236
Finnegan, C., & Breathnach, F. M. (2020). The role of aspirin for preeclampsia prevention in women with diabetes. Current Diabetes Reports, 20, 76
Zhu, S., Li, Z., Cui, L., Ban, Y., Leung, P. C. K., Li, Y., & Ma, J. (2021) Activin A increases human trophoblast invasion by upregulating integrin beta1 through ALK4. FASEB Journal, 35, e21220
Illsley, N. P., DaSilva-Arnold, S. C., Zamudio, S., Alvarez, M., & Al-Khan, A. (2020). Trophoblast invasion: Lessons from abnormally invasive placenta (placenta accreta). Placenta, 102, 61–66
Qu, H., Yu, Q., Jia, B., Zhou, W., Zhang, Y., & Mu, L. (2021). HIF3 alpha affects preeclampsia development by regulating EVT growth via activation of the Flt1/JAK/STAT signaling pathway in hypoxia. Molecular Medicine Reports, 23, 68.
Ning, F., Xin, H., Liu, J., Lv, C., Xu, X., Wang, M., Wang, Y., Zhang, W., & Zhang, X. (2020). Structure and function of USP5: Insight into physiological and pathophysiological roles. Pharmacological Research, 157, 104557
Nakajima, S., Lan, L., Wei, L., Hsieh, C. L., Rapic-Otrin, V., Yasui, A., & Levine, A. S. (2014). Ubiquitin-specific protease 5 is required for the efficient repair of DNA double-strand breaks. PLoS ONE, 9, e84899
Nostramo, R., Varia, S. N., Zhang, B., Emerson, M. M., & Herman, P. K. (2016). Deubiquitination and the regulation of stress granule assembly. Molecular and Cellular Biology, 36, 173–183
Liu, Q., Wu, Y., Qin, Y., Hu, J., Xie, W., Qin, F. X., & Cui, J. (2018). Broad and diverse mechanisms used by deubiquitinase family members in regulating the type I interferon signaling pathway during antiviral responses. Science Advances, 4, eaar2824
Lian, J., Liu, C., Guan, X., Wang, B., Yao, Y., Su, D., Ma, Y., Fang, L., & Zhang, Y. (2020). Ubiquitin specific peptidase 5 enhances STAT3 signaling and promotes migration and invasion in pancreatic cancer. Journal of Cancer, 11, 6802–6811
Wu, L., Zhang, C., Chu, M., Fan, Y., Wei, L., Li, Z., Yao, Y., & Zhuang, W. (2020). miR-125a suppresses malignancy of multiple myeloma by reducing the deubiquitinase USP5. Journal of Cellular Biochemistry, 121, 642–650
Xue, S., Wu, W., Wang, Z., Lu, G., Sun, J., Jin, X., Xie, L., Wang, X., Tan, C., Wang, Z., Wang, W., & Ding, X. (2020). Corrigendum: USP5 promotes metastasis in non-small cell lung cancer by inducing epithelial-mesenchymal transition via Wnt/β-catenin pathway. Frontiers in Pharmacology, 11, 948
Zhang, Z., Gao, W., Zhou, L., Chen, Y., Qin, S., Zhang, L., Liu, J., He, Y., Lei, Y., Chen, H. N., Han, J., Zhou, Z. G., Nice, E. C., Li, C., Huang, C., & Wei, X. (2019). Repurposing Brigatinib for the treatment of colorectal cancer based on inhibition of ER-phagy. Theranostics, 9, 4878–4892
Meng, J., Ai, X., Lei, Y., Zhong, W., Qian, B., Qiao, K., Wang, X., Zhou, B., Wang, H., Huai, L., Zhang, X., Han, J., Xue, Y., Liang, Y., Zhou, H., Chen, S., Sun, T., & Yang, C. (2019). USP5 promotes epithelial-mesenchymal transition by stabilizing SLUG in hepatocellular carcinoma. Theranostics, 9, 573–587
Yu, X., Zhang, Y., Yang, P., Gao, X., & Wang, Y. (2019). Downregulated low-density lipoprotein receptor-related protein 6 induces the maldevelopment of extravillous trophoblast via Wnt/β-catenin signaling pathway. Molecular and Cellular Probes, 44, 21–28
Funding
The study was supported by the Scientific Research Fund Project of Hebei Health and Family Planning Commission (20190471).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
None to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Li, L., Wang, S., Wang, M. et al. Ubiquitin-Specific Peptidase 5 is Involved in the Proliferation of Trophoblast Cells by Regulating Wnt/β-Catenin Signaling. Mol Biotechnol 63, 686–693 (2021). https://doi.org/10.1007/s12033-021-00330-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12033-021-00330-x