Abstract
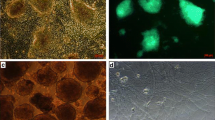
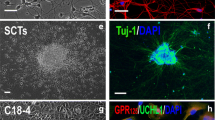
Embryonic stem (ES) like cells-derived testis represents a possible alternative to replace of neurons and glia. Here, we differentiated spermatogonia cells to oligoprogenitor (OP) like cells and transplanted them to demyelination model and assess their recovery potential in a demyelinated corpus callosum model in rats. ES like cells were differentiated to OP like cells using appropriate inducers and were transplanted in situ to demyelinated corpus callosum. Cell integration as well as demyelination extension and myelination intensity changes were evaluated using histologic studies and immunocytochemistry after 2 and 4 weeks post transplantation. Investigation of Nestin, NF68, Olig2, and NG2 by immunocytochemical technique indicated the differentiation of ES like cells to neuroprogenitor and oligodendrocyte like cells in each induction stage. Histologic findings showed a significant decrease in demyelination extension and a significant increase in remyelination intensity in cell transplanted groups. Also on the base of PLP expression, differentiation of transplanted cells was confirmed to myelinogenic cells using immunocytochemistry technique. We conclude that ES like cells derived from spermatogonia cells can be differentiated to OP like cells that can form myelin after transplantation into the demyelination model in rat, this represents recovery potential of spermatogonia cells which opens new window for cell therapeutic approaches using spermatogonial stem cells.




Similar content being viewed by others
References
Noseworthy, J. H., Lucchinetti, C., Rodriguez, M., & Weinshenker, B. G. (2000). Multiple sclerosis. The New England Journal of Medicine, 343, 938–952.
Manganas, L. N., & Maletic Savatic, M. (2005). Stem cell therapy for central nervous system demyelinating disease. Current Neurology and Neuroscience Reports, 5(3), 225–231.
Siddharthan, C., David, H., Alexis, J., Chao, Z., Alastair, C., & Robin, J. M. F. (2008). Myelin repair: the role of stem and precursor cells in multiple sclerosis. Philosophical Transactions of the Royal Society B, 363, 171–183.
Brustle, O., Jones, K. N., Learish, R. D., Karram, K., Choudhary, K., Wiestler, O. D., et al. (1999). Embryonic stem cell-derived glial precursors: a source of myelinating transplants. Science, 285, 754–756.
Martino, G., Franklin, R. J., Van Evercooren, A. B., & Kerr, D. A. (2010). Stem cell transplantation in multiple sclerosis: current status and future prospects. Nature Reviews Neurology, 6, 247–255.
Mizrak, S. C., Chikhovskaya, J. V., Sadri-Ardekani, H., van Daalen, S., Korver, C. M., Hovingh, S. E., et al. (2010). Embryonic stem cell-like cells derived from adult human testis. Human Reproduction, 25(1), 158–167.
Nayernia, K. (2007). Stem cells derived from testis show promise for treating a wide variety of medical conditions. Cell Research, 17, 895–897.
Kanatsu-Shinohara, M., Ogonuki, N., Inoue, K., Miki, H., Ogura, A., Toyokuni, S., et al. (2003). Long-termproliferation in culture and germline transmission of mouse male germline stem cells. Biology of Reproduction, 69, 612–616.
Streckfuss-Bömeke, K., Vlasov, A., Hülsmann, S., Yin, D., Nayernia, K., Engel, E., et al. (2009). Generation of functional neurons and glia from multipotent adult mouse germ-line stem cells. Stem Cell Research, 2, 139–154.
Aditya, G. S., & Chakrabarty, A. (2007). Multiple sclerosis and demyelination. Current Diagnostic Pathology, 13, 193–202.
Kotter, M. R., Setzu, A., Sim, F. J., Van Rooijen, N., & Franklin, R. J. (2001). Macrophage depletion impairs oligodendrocyte remyelination following lysolecithin-induced demyelination. Glia, 35(3), 204–212.
Woodruff, R., & Franklin, R. (1999). Demyelination and remyelination of the caudal cerebellar peduncle of adult rats following stereotaxic injections of lysolecithin, ethidium bromide, and complement/anti-galactocerebroside: a comparative study. Glia, 25, 216–228.
Yaldizli, O., Atefy, R., Gass, A., Sturm, D., Glassl, S., Tettenborn, B., et al. (2010). Corpus callosum index and long-term disability in multiple sclerosis patients. Journal of Neurology, 257, 1256–1264.
Nazm Bojnordi, M., Movahedin, M., Tiraihi, T., & Javan, M. (2013). Alteration in genes expression patterns during in vitro differentiation of mouse spermatogonial cells into neuroepithelial like cells. Cytotechnology, 65(1), 97–104.
Sherafat, M. A., Heibatollahi, M., Mongabadi, S., Moradi, F., Javan, M., & Ahmadiani, (2012). A electromagnetic field stimulation potentiates endogenous myelin repair by recruiting subventricular neural stem cells in an experimental model of white matter demyelination. Journal of Molecular Neuroscience, 48, 144–153.
Mozafari, S., Sherafat, M. A., Javan, M., Mirnajafi-Zadeh, J., & Tiraihi, T. (2010). Visual evoked potentials and MBP gene expression implyendogenous myelin repair in adult rat optic nerve and chiasma following local lysolecithin induced demyelination. Brain Research, 1351, 50–56.
Guan, K., Nayernia, K., Maier, L. S., Wagner, S., Dressel, R., Lee, J. H., et al. (2006). Pluripotency of spermatogonial stem cells from adult mouse testis. Nature, 440(7088), 1199–1203.
Hasenfuss, G. (2007). Generation of functional cardiomyocytes from adult mouse spermatogonial stem cells. Circulation Research, 100, 1615–1625.
De Rooij, D. J., & Mizrak, C. (2008). Deriving multipotent stem cells from mouse spermatogonial stem cells: A new tool for developmental and clinical research. Development, 135(430), 2207–2213.
Annapurna, A., Krishna, K. V., Murali Mohan Rao, P., Someswara Rao, K., & Rajasekhar, J. (2002). Multiple sclerosis: The disease and its treatment. Indian Journal of Pharmacology, 34, 3–15.
Caon, C., Saunders, C., Smrtka, J., Baxter, N., & Shoemaker, J. (2010). Injectable disease-modifying therapy for relapsing-remitting multiple sclerosis: A review of adherence data. Journal of Neuroscience Nursing, 42(5), S5–S9.
Chandran, S., Hunt, D., Joannides, A., Zhao, C., Compston, A., & Franklin, R. J. (2008). Myelin repair: The role of stem and precursor cells in multiple sclerosis. Philosophical transactions of the Royal Society of London Series B Biological Sciences, 363(1489), 171–183.
Stangel, M., & Hartung, H. P. (2002). Remyelinating strategies for the treatment of multiple sclerosis. Progress in Neurobiology, 68(5), 361–376.
Maden, M., Sonneveld, E., van der Saag, P. T., & Gale, E. (1998). The distribution of endogenous retinoic acid in the chick embryo: Implications for developmental mechanisms. Development, 125, 4133–4144.
Tirotta, E., Carbajal, K. S., Schaumburg, C. S., Whitman, L., & Lane, T. E. (2010). Cell replacement therapies to promote remyelination in a viral model of demyelination. Journal of Neuroimmunology, 224(1–2), 101–107.
Blakemore, W. F., Chari, D. M., Gilson, J. M., & Crang, A. J. (2002). Modelling large areas of demyelination in the rat reveals the potential and possible limitations of transplanted glial cells for remyelination in the CNS. Glia, 38(2), 155–168.
Chari, D. M., & Blakemore, W. F. (2002). New insights into remyelination failure in multiple sclerosis: Implications for glial cell transplantation. Multiple Sclerosis, 8(4), 271–277.
Mardanpour, P., Guan, K., Nolte, J., Lee, J., Hasenfuss, G., Engel, W., et al. (2008). Potency of germ cells and its relevance for regenerative medicine. Journal of Anatomy, 213(26–29), 457.
Acknowledgments
This research is derived from PhD thesis of Anatomical science with funding support from Medical Faculty of Tarbiat Modares University and Iranian council of stem cell technology.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Nazm Bojnordi, M., Movahedin, M., Tiraihi, T. et al. Oligoprogenitor Cells Derived from Spermatogonia Stem Cells Improve Remyelination in Demyelination Model. Mol Biotechnol 56, 387–393 (2014). https://doi.org/10.1007/s12033-013-9722-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12033-013-9722-0