Abstract
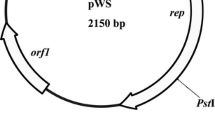
A shuttle vector pZL1 which can replicate both in Gluconobacter oxydans and Escherichia coli was constructed based on G. oxydans DSM2003 cryptic plasmid pGOX3, a homology of G. oxydans 621H pGOX3, and E. coli cloning vector pUC18. It was found to be stably maintained in G. oxydans during the serial subcultures in the absence of antibiotic pressure for 144 h. With pGOX3 as the reference sample, the relative copy number of pZL1 in G. oxydans is 13 determined by real-time fluorescence quantitative PCR (qPCR). The copy number of pZL1 is much higher than pBBR1MCS5 in E. coli. The vector pZL1 contains six commonly used restriction endonuclease sites, HindIII, SalI, XbaI, BamHI, SmaI, KpnI, and SacI, and is easy to manipulate in molecular biology experiments. The shuttle vector was used to express a reporter protein wasabi successfully in G. oxydans DSM2003 under the control of the tufB promoter.





Similar content being viewed by others
References
Gupta, A., Singh, V. K., Qazi, G. N., & Kumar, A. (2001). Gluconobacter oxydans: its biotechnological applications. Journal of Molecular Microbiology and Biotechnology, 3, 445–456.
Landis, B. H., McLaughlin, J. K., Heeren, R., Grabner, R. W., & Wang, P. T. (2002). Bioconversion of N-butylglucamine to 6-deoxy-6-butlylamino sorbose by Gluconobacter oxydans. Organic Process Research and Development, 6, 547–552.
Schedel, M. (2000). Regioselective oxidation of aminosorbitol with Gluconobacter oxydans, a key reaction in the industrial synthesis of 1-deoxynojirimycin. In D. R. Kelly (Ed.), Biotechnology, vol. 8b, biotransformations I (pp. 296–308). Weinheim, Germany: Wiley-VCH.
Xue-Peng, Y., Liu-Jing, W., Jin-Ping, L., Bo, Y., & Dong-Zhi, W. (2008). Membrane-bound pyrroloquinoline quinone-dependent dehydrogenase in Gluconobacter oxydans DSM2003, responsible for production of 6-(2-hydroxyethyl) amino-6-deoxy-L-sorbose. Applied and Environmental Microbiology, 74, 5250–5253.
Merfort, M., Herrmann, U., Ha, S.-W., Elfari, M., Bringer-Meyer, S., Görisch, H., et al. (2006). Modification of the membrane-bound glucose oxidation system in Gluconobacter oxydans significantly increases gluconate and 5-keto-D-gluconic acid accumulation. Biotechnology Journal, 1, 556–563.
Merfort, M., Herrmann, U., Bringer-Meyer, S., & Sahm, H. (2006). High-yield 5-keto-D-gluconic acid formation is mediated by soluble and membrane-bound gluconate-5-dyhydrogenases of Gluconobacter oxydans. Applied Microbiology and Biotechnology, 73, 443–451.
Fukaya, M., Okumura, H., Masai, H., Uozumi, T., & Beppu, T. (1989). Development of a host-vector system for Gluconobacter suboxydans. Agricultural and Biological Chemistry, 49, 2407–2411.
Condon, C., FitzGerald, R. J., & O’Gara, F. (1991). Conjugation and heterologous gene expression in Gluconobacter oxydans ssp. suboxydans. FEMS Microbiology Letters, 80, 173–178.
Schleyer, U., Bringer-Meyer, S., & Sakm, H. (2007). An easy cloning and expression vector system for Gluconobacter oxydans. International Journal of Food Microbiology, 65, 91–95.
Shinjoh, M., & Hoshino, T. (1995). Development of a stable shuttle vector and a conjugative transfer system for Gluconobacter oxydans. Journal of Fermentation and Bioengineering, 79, 95–99.
Fukaya, M., Okumura, H., Masai, H., Uozumi, T., & Beppu, T. (1985). Development of a host-vector system for Gluconobacter suboxydans. Agricultural and Biological Chemistry, 49, 2407–2411.
Sauto, Y., Ishii, Y., Hayashi, H., Imao, Y., Akashi, T., Yoshikawa, K., et al. (1997). Cloning of genes coding for L-sorbose and L-sorbosone dehydrogenases from Gluconobacter oxydans and microbial production of 2-keto-L-gluconate, a precursor of L-ascorbic acid in a recombinant G. oxydans strain. Applied and Environmental Microbiology, 63, 454–460.
Creavan, S., FitzGerald, R. J., & O’Gara, F. (1994). Transformation of Gluconobacter suboxydans by electroporation. Canadian Journal of Microbiology, 40, 491–494.
Naoto, T., Masakazu, S., & Kenzo, Y. (2003). Construction of a vector plasmid for use in Gluconobacter oxydans. Bioscience, Biotechnology, and Biochemistry, 67(1), 211–213.
Madako, S., & Tatsuo, H. (1995). Development of a stable shuttle vector and a conjugative transfer system for Gluconobacter oxydans. Journal of Fermentation and Bioengineering, 79(2), 95–99.
Prust, C., Hoffmeister, M., Liesegang, H., Wiezer, A., Fricke, W. F., Ehrenreich, A., et al. (2005). Comlete genome sequence of the acetic acid bacterium Gluconobacter oxydans. Nature Biotechnology, 23, 195–200.
Adele, L., Marston, & Jeffery, E. (1999). Dynamic movement of the ParA-like Soj protein of B. subtilis and its dual role in nucleoid organization and developmental regulation. Molecular Cell, 4, 673–682.
Yanisch-Perron, J. V., & Messing, J. (1985). Improved M13 phage cloning vectors and host strains: nucleotide sequences of the M13 mp18 and pUC19 vectors. Gene, 33, 103–119.
Sambrook, J., Fritsch, E. F., & Maniatis, T. (1989). Molecuar cloning: a laboratory manual (2nd ed.). Cold Spring Harbor, New York: Cold Spring Harbor Laboratory Press.
Acknowledgments
This work financially supported by the National Natural Science Foundation of China (Grant No. 20976053/B060804) and the National Special Fund for State Key Laboratory of Bioreactor Engineering (Grant No. 2060204).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, L., Lin, J., Ma, Y. et al. Construction of a Novel Shuttle Vector for Use in Gluconobacter oxydans . Mol Biotechnol 46, 227–233 (2010). https://doi.org/10.1007/s12033-010-9293-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12033-010-9293-2