Abstract
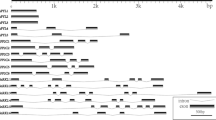
The last step of ethylene biosynthesis in apple is catalyzed by ACO (1-aminocyclopropane 1-carboxylic acid oxidase) encoded by MdACO1 and MdACO2. Both genes were expressed during early fruit development although at different level. Later on, the ACO transcript accumulation in persisting fruitlets decreased whereas in fruitlets undergoing abscission MdACO2 transcripts remained constant and those of MdACO1dramatically increased. The expression of MdACO was assessed by a particular type of semi-quantitative PCR involving 33P labeled primers and the following electrophoresis in polyacrylamide gel. The use of either the reverse primer or the anchored oligo dT13 produced clear and elegant results. Nevertheless, the initial quantity of labeled primer appeared to be a crucial factor for obtaining a wide range titration curve.




Similar content being viewed by others
References
Costa, F., Stella, S., Van de Weg, W. E., Guerra, W., Cecchinel, M., Dallavia, J., Bernie Koller, B., & Sansavini, S. (2005). Role of the genes Md-ACO1 and Md-ACS1 in ethylene production and shelf life of apple (Malus domestica Borkh). Euphytica, 141, 181–190.
Dal Cin, V., Danesin, M., Boschetti, A., Dorigoni, A., & Ramina, A. (2005). Ethylene biosynthesis and perception in apple fruitlet abscission (Malus domestica L. Borkh). Journal of Experimental Botany, 56, 2995–3005.
Dal Cin, V., Danesin, M., Rizzini, F. M., & Ramina, A. (2005). RNA extraction from plant tissues: The use of calcium to precipitate contaminating pectic sugars. Molecular Biotechnology, 31, 113–119.
Dean, T. R., Kohan, M., Betancourt, D., & Menetrez, M. Y. (2005). A simple polymerase chain reaction/restriction fragment length polymorphism assay capable of identifying medically relevant filamentous fungi. Molecular Biotechnology, 31, 21–28.
Hirano, M. (2004). RACE using only a gene-specific primer: Application of a temporal switching model. Molecular Biotechnology, 27, 179–186.
Nicot, N., Hausman, J.-F., Hoffmann, L., & Evers, D. (2005). Housekeeping gene selection for real-time RT-PCR normalization in potato during biotic and abiotic stress. Journal of Experimental Botany, 56, 2907–2914.
Sachse, K. (2004). Specificity and performance of PCR detection assays for microbial pathogens. Molecular Biotechnology, 26, 61–80.
Sambrook, J., Fritsch, E. F., & Maniatis, T. (1989). Molecular cloning: A laboratory manual. New York, NY: Cold Spring Harbor Laboratory.
Soares, S., Boxberg, Y. V., Ravaille-Veron, M., Vincent, J.-D., & Nothias, F. (2000). Morphofunctional plasticity in the adult hypothalamus induces regulation of polysialic acid-neural cell adhesion molecule through changing activity and expression levels of polysialyltransferases. The Journal of Neuroscience, 20, 2551–2557.
Stoughton, R. B. (2005). Applications of DNA microarrays in biology. Annu. Rev. Biochem., 74, 53–82.
Thrash-Bingham, C. A., & Tartof, K. D. (1999) aHIF: A natural antisense transcript overexpressed in human renal cancer and during hypoxia. Journal of the National Cancer Institute, 91, 143–151.
Volkov, R. A., Panchuk, I. I., & Scholl, F. (2003). Heat-stress-dependency and developmental modulation of gene expression: The potential of house-keeping genes as internal standards in mRNA expression profiling using real-time RT-PCR. Journal of Experimental Botany, 54, 2343–2349.
Wong, M. L., & Medrano, J. F. (2005). Real-time PCR for mRNA quantitation. Biotechniques, 39, 75–85.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dal Cin, V., Galla, G. & Ramina, A. MdACO expression during abscission. Mol Biotechnol 36, 9–13 (2007). https://doi.org/10.1007/s12033-007-0004-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12033-007-0004-6