Abstract
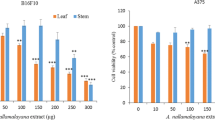
Cancer is considered most detrimental due to high mortality worldwide. Among them, skin cancers play a major part by affecting one in three cancer patients globally. About 2–3 million cancer cases were reported to be non-melanoma and melanoma skin cancers, respectively. Although chemotherapeutic drugs act on cancer cells but results in long-lasting morbidities which affects one’s quality of life and also works only in the initial stage of the cancer. Hence, an idea of traditional medicine to cure the disease efficiently with less side effects was pursued by the researchers. We have assessed the combination effect of p-coumaric acid and naringin in exerting anticancer activity using A431 (epidermoid carcinoma) cells. The MTT analysis of the combination on A431 cells showed the least IC50 concentration of 41 µg/ml which is effective than the standard drug imiquimod with IC50 concentration of 52 µg/ml. Further, flow cytometric analysis was carried out to identify the molecular mechanism behind the anticancer effects of the combination. The results revealed that the combination arrested the A431 cell cycle at S phase, induced apoptosis as indicated by more early and late apoptotic cells when compared with the control, and further altered reactive oxygen species (ROS) and mitochondrial membrane potential in A431 cells. Hence, the results suggest the potential anticancer effects of p-coumaric acid and naringin combination against the skin cancer (A431) cell line. The observed effects may be additive or synergistic effects in inducing ROS generation and apoptosis, and reducing the viability of A431 cells.






Similar content being viewed by others
Data availability
The data will be made available upon reasonable request from the corresponding author.
Abbreviations
- SSC:
-
Squamous Cell Carcinoma
- BCC:
-
Basal Cell Carcinoma
- ROS:
-
Reactive Oxygen Species
- MMP:
-
Mitochondrial Membrane Potential
- Drp1:
-
Dynamin-Related Protein
- ING1:
-
Inhibitor of Growth
- ICAD:
-
Inhibitor of Caspase-Activated DNAse
- CAD:
-
Caspase-Activated DNAse
References
Ossio R, Roldán-Marín R, Martínez-Said H, Adams DJ, Robles-Espinoza CD. Melanoma: a global perspective. Nat Rev Cancer. 2017;17(7):393–4. https://doi.org/10.1038/nrc.2017.43.
Vyas A, Das S, Singh D, Sonker AK, Gidwani B, Jain V, Singh M. Recent nanoparticulate approaches of drug delivery for skin cancer. Trends Appl Sci Res. 2012;7(8):620–35. https://doi.org/10.3923/tasr.2012.620.635.
McGuire Shelley. World cancer report Geneva 2014, Switzerland: World health organization, International agency for research on cancer WHO Press 2015. Adv Nutri. 2016;7(2):418–9. https://doi.org/10.3945/an.116.012211.
Parkin DM, Bray F, Ferlay J, Pisani P. Global cancer statistics. Cancer J Clin. 2005;55(2):74–108. https://doi.org/10.3322/canjclin.55.2.74.
Howe HL, Wingo PA, Thun MJ, et al. Annual report to the nation on the status of cancer (1973 through 1998), featuring cancers with recent increasing trends. J Natl Cancer Inst. 2001;93(11):824–42. https://doi.org/10.1093/jnci/93.11.824.
Deo SVS, Hazarika S, Shukla NK, Kumar S, Kar M, Samaiya A. Surgical management of skin cancers: experience from a regional cancer centre in North India. Indian J Cancer. 2005;42(3):145. https://doi.org/10.4103/0019-509x.17059.
Panda S. Nonmelanoma skin cancer in India: current scenario. Indian J Dermatol. 2010;55(4):373. https://doi.org/10.4103/0019-5154.74551.
Birt DF, Hendrich S, Wang W. Dietary agents in cancer prevention: flavonoids and isoflavonoids. Pharmacol Ther. 2001;90(2–3):157–77. https://doi.org/10.1016/s0163-7258(01)00137-1.
Park JH, Jin C, Lee BK, Kim G, Choi YH, Jeong YK. Naringenin induces apoptosis through downregulation of Akt and caspase-3 activation in human leukemia THP-1 cells. Food Chem Toxicol. 2008;46(12):3684–90. https://doi.org/10.1016/j.fct.2008.09.056.
Rice-Evans C, Miller NJ, Paganga G. Structure-antioxidant activity relationships of flavonoids and phenolic acids. Free Radical Biol Med. 1996;20(7):933–56. https://doi.org/10.1016/0891-5849(95)02227-9.
Croft KD. The chemistry and biological effects of flavonoids and phenolic acids a. Ann N Y Acad Sci. 1998;854(1):435–42. https://doi.org/10.1111/j.1749-6632.1998.tb09922.x.
Hertog MGL, Hollman PCH, Katan MB, Kromhout D. Intake of potentially anticarcinogenic flavonoids and their determinants in adults in the Netherlands. Nutr Cancer. 1993;20(1):21–9. https://doi.org/10.1080/01635589309514267.
Kanno S, Tomizawa A, Hiura T, et al. Inhibitory effects of naringenin on tumor growth in human cancer cell lines and sarcoma S-180-Implanted mice. Biol Pharm Bull. 2005;28(3):527–30. https://doi.org/10.1248/bpb.28.527.
An SM, Koh JH, Boo YC. p-coumaric acid not only inhibits human tyrosinase activity in vitro but also melanogenesis in cells exposed to UVB. Phytother Res. 2010;24(8):1175–80. https://doi.org/10.1002/ptr.3095.
Seo YK, Kim SJ, Boo YC, Baek JS, Lee SH, Koh JH. Effects of p-coumaric acid on erythema and pigmentation of human skin exposed to ultraviolet radiation. Clin Exp Dermatol. 2010;36(3):260–6. https://doi.org/10.1111/j.1365-2230.2010.03983.x.
Chang TS. Natural melanogenesis inhibitors acting through the down-regulation of tyrosinase activity. Materials (Basel). 2012;5(9):1661–85. https://doi.org/10.3390/ma5091661.
Boo YC. p-Coumaric acid as an active ingredient in cosmetics: a review focusing on its antimelanogenic effects. Antioxidants (Basel). 2019;8(8):275. https://doi.org/10.3390/antiox8080275.
Pop T, Diaconeasa Z. Recent advances in phenolic metabolites and skin cancer. Int J Mol Sci. 2021;22(18):9707. https://doi.org/10.3390/ijms22189707.
Dasari S, Yedjou CG, Brodell RT, Cruse A, Tchounwou PB. Therapeutic strategies and potential implications of silver nanoparticles in the management of skin cancer. Nanotechnol Rev. 2020;9(1):1500–21. https://doi.org/10.1515/ntrev-2020-0117.
Davis D, Hughes C. Dead cell discrimination In Living colour. Berlin: Springer; 2000.
Yang C, Pei W, Zhao J, Cheng Y, Zheng X, Rong J. Bornyl caffeate induces apoptosis in human breast cancer MCF-7 cells via the ROS- and JNK-mediated pathways. Acta Pharmacol Sin. 2013;35(1):113–23. https://doi.org/10.1038/aps.2013.162.
Ahamad S, Siddiqui S, Jafri A, Ahmad S, Afzal M, Ali A. Induction of apoptosis and antiproliferative activity of naringenin in human epidermoid carcinoma cell through ROS generation and cell cycle arrest. PLoS ONE. 2014;9(10): e110003. https://doi.org/10.1371/journal.pone.0110003.
Hu Q, Siu SO, Chen Y, et al. Senkyunolides reduce hydrogen peroxide-induced oxidative damage in human liver HepG2 cells via induction of heme oxygenase-1. Chem Biol Inter. 2010;183(3):380–9. https://doi.org/10.1016/j.cbi.2009.11.029.
Hu Q, Chen B, Le XC, Rong J. Concomitant induction of heme oxygenase-1 attenuates the cytotoxicity of arsenic species from lumbricus extract in human liver HepG2 cells. Chem Biodivers. 2012;9(4):739–54. https://doi.org/10.1002/cbdv.201100133.
Hegde M, Karki SS, Thomas E, et al. Novel levamisole derivative induces extrinsic pathway of apoptosis in cancer cells and inhibits tumor progression in mice. PLoS ONE. 2012;7(9): e43632. https://doi.org/10.1371/journal.pone.0043632.
Duncan BD. Multiple range tests for correlated and heteroscedastic means. Biometrics. 1957;13:359–64.
Strömberg U, Parkes BL, Holmén A, Peterson S, Holmberg E, Baigi A, Piel FB. Disease mapping of early- and late-stage cancer to monitor inequalities in early detection: a study of cutaneous malignant melanoma. Eur J Epidemiol. 2020;35(6):537–47. https://doi.org/10.1007/s10654-020-00637-0.
Sharma SH, Rajamanickam V, Sangeetha N. Antiproliferative effect of p-Coumaric acid targets UPR activation by downregulating Grp78 in colon cancer. Chem Biol Interact. 2018;291:16–28. https://doi.org/10.1016/j.cbi.2018.06.001.
Sharma SH, Rajamanickam V, Sangeetha N. Supplementation of p-coumaric acid exhibits chemopreventive effect via induction of Nrf2 in a short-term preclinical model of colon cancer. Eur J Cancer Prev. 2019;28(6):472–82. https://doi.org/10.1097/cej.0000000000000496.
Bubna AK. Imiquimod - Its role in the treatment of cutaneous malignancies. Indian J Pharmacol. 2015;47(4):354. https://doi.org/10.4103/0253-7613.161249.
Tillman DK Jr, Carroll MT. Topical imiquimod therapy for basal and squamous cell carcinomas: a clinical experience. Cutis. 2007;79(3):241–8.
Bharti S, Rani N, Krishnamurthy B, Arya DS. Preclinical evidence for the pharmacological actions of Naringin: a review. Planta Med. 2014;80(06):437–51. https://doi.org/10.1055/s-0034-1368351.
Pfeffer CM, Singh ATK. Apoptosis: a target for anticancer therapy. Int J Mol Sci. 2018;19(2):448. https://doi.org/10.3390/ijms19020448.
Picard M, McEwen BS, Epel ES, Sandi C. An energetic view of stress: Focus on mitochondria. Front Neuroendocrinol. 2018;49:72–85. https://doi.org/10.1016/j.yfrne.2018.01.001.
Burke PJ. Mitochondria, bioenergetics and apoptosis in cancer. Trends in Cancer. 2017;3(12):857–70. https://doi.org/10.1016/j.trecan.2017.10.006.
Pancrazi L, Di Benedetto G, Colombaioni L, et al. Foxg1 localizes to mitochondria and coordinates cell differentiation and bioenergetics. Proc Natl Acad Sci USA. 2015;112(45):13910–5. https://doi.org/10.1073/pnas.1515190112.
Cohen GM. Caspases: the executioners of apoptosis. Biochem J. 1997;326(1):1–16. https://doi.org/10.1042/bj3260001.
Thornberry NA, Rano TA, Peterson EP, et al. A combinatorial approach defines specificities of members of the Caspase family and Granzyme B. J Biol Chem. 1997;272(29):17907–11. https://doi.org/10.1074/jbc.272.29.17907.
Enari M, Sakahira H, Yokoyama H, Okawa K, Iwamatsu A, Nagata S. A caspase-activated DNase that degrades DNA during apoptosis, and its inhibitor ICAD. Nature. 1998;391(6662):43–50. https://doi.org/10.1038/34112.
Nagata S. Apoptotic DNA fragmentation. Exp Cell Res. 2000;256(1):12–8. https://doi.org/10.1006/excr.2000.4834.
Arunasree KM. Anti-proliferative effects of carvacrol on a human metastatic breast cancer cell line, MDA-MB 231. Phytomedicine. 2010;17(8–9):581–8. https://doi.org/10.1016/j.phymed.2009.12.008.
Mehdi SJ, Ahmad A, Irshad M, Manzoor N, Rizvi MMA. Cytotoxic effect of carvacrol on human cervical cancer cells. Biol Med. 2011;3(2):307–12. https://doi.org/10.4172/0974-8369.10000119.
Yin Q, Yan F, Zu X, et al. Anti-proliferative and pro-apoptotic effect of carvacrol on human hepatocellular carcinoma cell line HepG-2. Cytotechnology. 2011;64(1):43–51. https://doi.org/10.1007/s10616-011-9389-y.
Heidarian E, Keloushadi M. Antiproliferative and anti-invasion effects of carvacrol on PC3 human prostate cancer cells through reducing pSTAT3, pAKT, and pERK1/2 signaling proteins. Int J Prev Med. 2019;10(1):156. https://doi.org/10.4103/ijpvm.ijpvm_292_17.
Ahmed H, Shousha W, El-mezayen H, Ismaiel N, Mahmoud N. In vivo antitumor potential of carvacrol against hepatocellular carcinoma in rat model. World J Pharm Pharmaceut Sci. 2013;2:2367–96.
Martin-Cordero C, Leon-Gonzalez AJ, Calderon-Montano JM, Burgos-Moron E, Lopez-Lazaro M. Pro-Oxidant natural products as anticancer agents. Current Drug Targets. 2012;13(8):1006–28. https://doi.org/10.2174/138945012802009044.
Fruehauf JP, Meyskens FL. Reactive oxygen species: a breath of life or death? Clin Cancer Res. 2007;13(3):789–94. https://doi.org/10.1158/1078-0432.ccr-06-2082.
Trachootham D, Alexandre J, Huang P. Targeting cancer cells by ROS-mediated mechanisms: a radical therapeutic approach? Nat Rev Drug Discovery. 2009;8(7):579–91. https://doi.org/10.1038/nrd2803.
Mokhtari R, Homayouni TS, Baluch N, et al. Combination therapy in combating cancer. Oncotarget. 2017;8(23):38022–43. https://doi.org/10.18632/oncotarget.16723.
Acknowledgements
The authors thank the President, CEO, Vice-Chancellor, Registrar KAHE for providing b infrastructure and encouragement exercise this study.
Funding
The authors have not disclosed any funding.
Author information
Authors and Affiliations
Contributions
All Authors have been intellectually committed to the study design and all processes.
Corresponding author
Ethics declarations
Competing interest
The Authors have no financial conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Velusamy, P., Muthusami, S. & Arumugam, R. In vitro evaluation of p-coumaric acid and naringin combination in human epidermoid carcinoma cell line (A431). Med Oncol 41, 4 (2024). https://doi.org/10.1007/s12032-023-02230-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12032-023-02230-3