Abstract
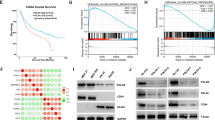
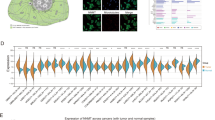
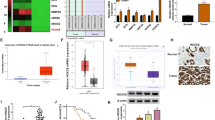
Neuroblastoma (NB) is the deadliest pediatric solid tumor due to its rapid proliferation. Aberrant expression of MYCN is deemed as the most remarkable feature for the predictive hallmark of NB progression and recurrence. However, the phenomenon that only detection of MYCN in the nearly 20% of NB patients hints that there should be other vital oncogenes in the progression of NB. Here, we firstly show that MSI2 mRNA is augmented by analyzing public GEO datasets in the malignant stage according to International Neuroblastoma Staging System (INSS) stages. Although accumulating evidences uncover the emerging roles of MSI2 in several cancers, the regulatory functions and underlying mechanisms of MSI2 in NB remain under-investigated. Herein, we identified that high-expressed MSI2 and low-expressed n-Myc group account for 43.1% of total NB clinical samples (n = 65). Meanwhile, MSI2 expression is profoundly upregulated along with NB malignancy and negatively associated with the survival outcome of NB patients in the NB tissue microarray (NB: n = 65; Ganglioneuroblastoma: n = 31; Ganglioneuroma: n = 27). In vitro, our results revealed that MSI2 promoted migration, invasion, and proliferation of NB cells via enhancing pentose phosphate pathway. Mechanistically, MSI2 upregulated the key enzyme glucose-6-phosphate dehydrogenase (G6PD) via directly binding to 3'-untranslated regions of c-Myc mRNA to facilitate its stability, resulting in enhancing pentose phosphate pathway. Our findings reveal that MSI2 promotes pentose phosphate pathway via activating c-Myc-G6PD signaling, suggesting that MSI2 exhibits a novel and powerful target for the diagnosis and treatment of NB.







Similar content being viewed by others
Data and materials availability
All data supporting the conclusions of this study are included in this published article. All data are included in the manuscript.
Abbreviations
- MSI2:
-
Musashi-2
- G6PD:
-
Glucose-6-phosphate dehydrogenase
- NB:
-
Neuroblastoma
- TMA:
-
Tissue microarray
- IHC:
-
Immunohistochemistry
- INRG:
-
The International Neuroblastoma Risk Group
- RIP:
-
RNA-binding protein immunoprecipitation
References
Pinto NR, Applebaum MA, Volchenboum SL, Matthay KK, London WB, Ambros PF, et al. Advances in risk classification and treatment strategies for neuroblastoma. J Clin Oncol. 2015;33(27):3008–17.
Shimada H, Umehara S, Monobe Y, Hachitanda Y, Nakagawa A, Goto S, et al. International neuroblastoma pathology classification for prognostic evaluation of patients with peripheral neuroblastic tumors: a report from the Children’s Cancer Group. Cancer. 2001;92(9):2451–61.
DuBois SG, Kalika Y, Lukens JN, Brodeur GM, Seeger RC, Atkinson JB, et al. Metastatic sites in stage IV and IVS neuroblastoma correlate with age, tumor biology, and survival. J Pediatr Hematol Oncol. 1999;21(3):181–9.
Maris JM. Recent advances in neuroblastoma. N Engl J Med. 2010;362(23):2202–11.
Mora J, Cheung NKV, Gerald WL. Genetic heterogeneity and clonal evolution in neuroblastoma. Br J Cancer. 2001;85(2):182–9.
Nakamura M, Okano H, Blendy JA, Montell C. Musashi, a neural RNA-binding protein required for Drosophila adult external sensory organ development. Neuron. 1994;13(1):67–81.
Kudinov AE, Karanicolas J, Golemis EA, Boumber Y. Musashi RNA-binding proteins as cancer drivers and novel therapeutic targets. Clin Cancer Res. 2017;23(9):2143–53.
Park SM, Deering RP, Lu Y, Tivnan P, Lianoglou S, Al-Shahrour F, et al. Musashi-2 controls cell fate, lineage bias, and TGF-beta signaling in HSCs. J Exp Med. 2014;211(1):71–87.
Sakakibara S, Nakamura Y, Yoshida T, Shibata S, Koike M, Takano H, et al. RNA-binding protein Musashi family: roles for CNS stem cells and a subpopulation of ependymal cells revealed by targeted disruption and antisense ablation. Proc Natl Acad Sci USA. 2002;99(23):15194–9.
Wang S, Li N, Yousefi M, Nakauka-Ddamba A, Li F, Parada K, et al. Transformation of the intestinal epithelium by the MSI2 RNA-binding protein. Nat Commun. 2015;6:6517.
Zhu Y, Zhou B, Hu X, Ying S, Zhou Q, Xu W, et al. LncRNA LINC00942 promotes chemoresistance in gastric cancer by suppressing MSI2 degradation to enhance c-Myc mRNA stability. Clin Transl Med. 2022;12(1):e703.
Qu C, He L, Yao N, Li JY, Jiang YCA, Li BK, et al. Myofibroblast-specific Msi2 knockout inhibits HCC progression in a mouse model. Hepatology. 2021;74(1):458–73.
Dong WW, Liu XB, Yang CQ, Wang D, Xue YX, Ruan XL, et al. Glioma glycolipid metabolism: MSI2-SNORD12B-FIP1L1-ZBTB4 feedback loop as a potential treatment target. Clin Transl Med. 2021;11(5):e411.
Hattori A, McSkimming D, Kannan N, Ito T. RNA binding protein MSI2 positively regulates FLT3 expression in myeloid leukemia. Leukemia Res. 2017;54:47–54.
Hattori A, Tsunoda M, Konuma T, Kobayashi M, Nagy T, Glushka J, et al. Cancer progression by reprogrammed BCAA metabolism in myeloid leukaemia. Nature. 2017;545(7655):500–4.
Makhov P, Bychkov I, Faezov B, Deneka A, Kudinov A, Nicolas E, et al. Musashi-2 (MSI2) regulates epidermal growth factor receptor (EGFR) expression and response to EGFR inhibitors in EGFR-mutated non-small cell lung cancer (NSCLC). Oncogenesis. 2021;10(3):29.
Heiden MGV, Cantley LC, Thompson CB. Understanding the Warburg effect: the metabolic requirements of cell proliferation. Science. 2009;324(5930):1029–33.
Gatenby RA, Gillies RJ. Why do cancers have high aerobic glycolysis? Nat Rev Cancer. 2004;4(11):891–9.
Wamelink M, Struys E, Jakobs C. The biochemistry, metabolism and inherited defects of the pentose phosphate pathway: a review. J Inherit Metab Dis. 2008;31(6):703–17.
Jiang P, Du WJ, Wu MA. Regulation of the pentose phosphate pathway in cancer. Protein Cell. 2014;5(8):592–602.
Bose S, Huang Q, Ma Y, Wang L, Rivera GO, Ouyang Y, et al. G6PD inhibition sensitizes ovarian cancer cells to oxidative stress in the metastatic omental microenvironment. Cell Rep. 2022;39(13):111012.
Aurora AB, Khivansara V, Leach A, Gill JG, Martin-Sandoval M, Yang CD, et al. Loss of glucose 6-phosphate dehydrogenase function increases oxidative stress and glutaminolysis in metastasizing melanoma cells. Proc Natl Acad Sci USA. 2022;119(6):e2120617119.
Jiang P, Huang M, Qi WW, Wang FH, Yang TY, Gao TX, et al. FUBP1 promotes neuroblastoma proliferation via enhancing glycolysis-a new possible marker of malignancy for neuroblastoma. J Exp Clin Canc Res. 2019;38(1):400.
Qiu B, Matthay KK. Advancing therapy for neuroblastoma. Nat Rev Clin Oncol. 2022;19(8):515–33.
Chen YY, Takita J, Choi YL, Kato M, Ohira M, Sanada M, et al. Oncogenic mutations of ALK kinase in neuroblastoma. Nature. 2008;455(7215):971–4.
Pugh TJ, Morozova O, Attiyeh EF, Asgharzadeh S, Wei JS, Auclair D, et al. The genetic landscape of high-risk neuroblastoma. Nat Genet. 2013;45(3):279–84.
Ponzoni M, Bachetti T, Corrias MV, Brignole C, Pastorino F, Calarco E, et al. Recent advances in the developmental origin of neuroblastoma: an overview. J Exp Clin Cancer Res. 2022;41(1):92.
Wang PL, Teng L, Feng YC, Yue YM, Han MM, Yan Q, et al. The N-Myc-responsive lncRNA MILIP promotes DNA double-strand break repair through non-homologous end joining. Proc Natl Acad Sci USA. 2022;119(49):e2208904119.
Grunblatt E, Wu N, Zhang H, Liu X, Norton JP, Ohol Y, et al. MYCN drives chemoresistance in small cell lung cancer while USP7 inhibition can restore chemosensitivity. Genes Dev. 2020;34(17–18):1210–26.
Kharas MG, Lengner CJ. Stem cells, cancer, and MUSASHI in blood and guts. Trends Cancer. 2017;3(5):347–56.
Stine ZE, Schug ZT, Salvino JM, Dang CV. Targeting cancer metabolism in the era of precision oncology. Nat Rev Drug Discov. 2022;21(2):141–62.
Wu S, Wang H, Li Y, Xie Y, Huang C, Zhao H, et al. Transcription factor YY1 promotes cell proliferation by directly activating the pentose phosphate pathway. Cancer Res. 2018;78(16):4549–62.
Ding H, Chen Z, Wu K, Huang SM, Wu WL, LeBoeuf SE, et al. Activation of the NRF2 antioxidant program sensitizes tumors to G6PD inhibition. Sci Adv. 2021;7(47):eabk1023.
Zhang R, Tao F, Ruan S, Hu M, Hu Y, Fang Z, et al. The TGFbeta1-FOXM1-HMGA1-TGFbeta1 positive feedback loop increases the cisplatin resistance of non-small cell lung cancer by inducing G6PD expression. Am J Transl Res. 2019;11(11):6860–76.
Yin X, Tang B, Li JH, Wang Y, Zhang L, Xie XY, et al. ID1 promotes hepatocellular carcinoma proliferation and confers chemoresistance to oxaliplatin by activating pentose phosphate pathway. J Exp Clin Cancer Res. 2017;36(1):166.
Fang Y, Shen ZY, Zhan YZ, Feng XC, Chen KL, Li YS, et al. CD36 inhibits beta-catenin/c-myc-mediated glycolysis through ubiquitination of GPC4 to repress colorectal tumorigenesis. Nat Commun. 2019;10(1):3981.
Tang YC, Hsiao JR, Jiang SS, Chang JY, Chu PY, Liu KJ, et al. c-MYC-directed NRF2 drives malignant progression of head and neck cancer via glucose-6-phosphate dehydrogenase and transketolase activation. Theranostics. 2021;11(11):5232–47.
Cao L, Zhang P, Li J, Wu M. LAST, a c-Myc-inducible long noncoding RNA, cooperates with CNBP to promote CCND1 mRNA stability in human cells. eLife. 2017;6:e30433.
He L, Liu J, Collins I, Sanford S, O’Connell B, Benham CJ, et al. Loss of FBP function arrests cellular proliferation and extinguishes c-myc expression. EMBO J. 2000;19(5):1034–44.
Arnovitz S, Mathur P, Tracy M, Mohsin A, Mondal S, Quandt J, et al. Tcf-1 promotes genomic instability and T cell transformation in response to aberrant beta-catenin activation. Proc Natl Acad Sci U S A. 2022;119(32):e2201493119.
Umek T, Sollander K, Bergquist H, Wengel J, Lundin KE, Smith CIE, et al. Oligonucleotide binding to non-B-DNA in MYC. Molecules. 2019;24(5):1000.
Duggimpudi S, Kloetgen A, Maney SK, Munch PC, Hezaveh K, Shaykhalishahi H, et al. Transcriptome-wide analysis uncovers the targets of the RNA-binding protein MSI2 and effects of MSI2’s RNA-binding activity on IL-6 signaling. J Biol Chem. 2018;293(40):15359–69.
Acknowledgements
The authors thank for giving NB cell lines for a gift by Sun Yat-sen University School of Medicine.
Funding
This project was funded by the National Nature Science Foundation of China (NO: 82202829); the Guangdong Basic and Applied Basic research Foundation (NO: 2021A1515110094); the Sci-tech Research Project of Guangzhou Municipality (NO: 202201010180; NO: 202102021165; NO: 202102020776); and the Science Foundation of Guangzhou People’s First Hospital (KY09040041).
Author information
Authors and Affiliations
Contributions
BX designed the whole experiments and funded this study; PJ performed the experiments and data analysis; TZ collected clinical samples and analyzed the data; BW, XL, and MF carried out the experiments in vitro. All authors reviewed and agreed to submit the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical approval and consent to the participate
The authors declare that the manuscript was approved by the Medical Ethics Committee of Southern China University of Technology.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jiang, P., Zhang, T., Wu, B. et al. Musashi-2 (MSI2) promotes neuroblastoma tumorigenesis through targeting MYC-mediated glucose-6-phosphate dehydrogenase (G6PD) transcriptional activation. Med Oncol 40, 332 (2023). https://doi.org/10.1007/s12032-023-02199-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12032-023-02199-z