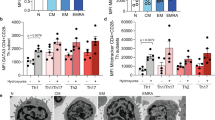
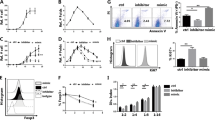
Abstract
The damaged DNA-binding protein 1 (DDB1) enhances the survival and maintenance of multipotent cells through promoting the Cullin 4 E3 ligase complex-dependent ubiquitination and subsequent degradation of downstream substrates. Naive T cells could be activated and differentiated into effector and memory T cells by exogenous stimulatory molecules, which are essential in immune response and inflammation. However, possible regulation and molecular mechanisms of DDB1 in T-cell activation-induced apoptosis were largely unknown. Here, in this study, we uncovered that DDB1 could downregulate the expression of histone methyltransferase SETD7 through decreasing its mRNA level and then regulated activation-induced apoptosis of T-cell line Jurkat cells. Furthermore, RNA-sequencing assay on activated Jurkat cells confirmed that the SETD7 attenuated the activation of Jurkat cells. Our study revealed the non-enzymatic functions of DDB1 on the activation-induced apoptosis of T cells.




Similar content being viewed by others
References
Iovine B, Iannella ML, Bevilacqua MA. Damage-specific DNA binding protein 1 (DDB1): a protein with a wide range of functions. Int J Biochem Cell Biol. 2011;43(12):1664–7. https://doi.org/10.1016/j.biocel.2011.09.001.
Sarikas A, Hartmann T, Pan ZQ. The cullin protein family. Genome Biol. 2011;12(4):220. https://doi.org/10.1186/gb-2011-12-4-220.
Kapetanaki MG, Guerrero-Santoro J, Bisi DC, Hsieh CL, Rapic-Otrin V, Levine AS. The DDB1-CUL4ADDB2 ubiquitin ligase is deficient in xeroderma pigmentosum group E and targets histone H2A at UV-damaged DNA sites. Proc Natl Acad Sci USA. 2006;103(8):2588–93. https://doi.org/10.1073/pnas.0511160103.
Bevilacqua MA, Iovine B, Zambrano N, D’Ambrosio C, Scaloni A, Russo T, et al. Fibromodulin gene transcription is induced by ultraviolet irradiation, and its regulation is impaired in senescent human fibroblasts. J Biol Chem. 2005;280(36):31809–17. https://doi.org/10.1074/jbc.M414677200.
Wang X, Wang HY, Hu GS, Tang WS, Weng L, Zhang Y, et al. DDB1 binds histone reader BRWD3 to activate the transcriptional cascade in adipogenesis and promote onset of obesity. Cell Rep. 2021;35(12):109281. https://doi.org/10.1016/j.celrep.2021.109281.
Wittschieben BO, Wood RD. DDB complexities. DNA Repair (Amst). 2003;2(9):1065–9. https://doi.org/10.1016/s1568-7864(03)00113-7.
Ding WY, Huang J, Wang H. Waking up quiescent neural stem cells: molecular mechanisms and implications in neurodevelopmental disorders. PLoS Genet. 2020;16(4):e1008653. https://doi.org/10.1371/journal.pgen.1008653.
Fu L, Cui CP, Zhang L. Regulation of stem cells by cullin-RING ligase. In: Sun Y, Wei W, Jin J, editors. Cullin-RING ligases and protein neddylation: biology and therapeutics. Singapore: Springer; 2020. p. 79–98.
Cang Y, Zhang J, Nicholas SA, Bastien J, Li B, Zhou P, et al. Deletion of DDB1 in mouse brain and lens leads to p53-dependent elimination of proliferating cells. Cell. 2006;127(5):929–40. https://doi.org/10.1016/j.cell.2006.09.045.
Gao J, Buckley SM, Cimmino L, Guillamot M, Strikoudis A, Cang Y, et al. The CUL4-DDB1 ubiquitin ligase complex controls adult and embryonic stem cell differentiation and homeostasis. elife. 2015;4:e07539. https://doi.org/10.7554/eLife.07539.
Zhao L, Liao H, Wang X, Chen YG. DDB1 maintains intestinal homeostasis by preventing cell cycle arrest. Cell Regen. 2022;11(1):18. https://doi.org/10.1186/s13619-022-00119-6.
Pepper M, Jenkins MK. Origins of CD4(+) effector and central memory T cells. Nat Immunol. 2011;12(6):467–71. https://doi.org/10.1038/ni.2038.
Zhou L, Duan J. The NMDAR GluN1-1a C-terminus binds to CaM and regulates synaptic function. Biochem Biophys Res Commun. 2021;534:323–9. https://doi.org/10.1016/j.bbrc.2020.11.085.
Hao Z, Liu L, Tao Z, Wang R, Ren H, Sun H, et al. Motor dysfunction and neurodegeneration in a C9orf72 mouse line expressing poly-PR. Nat Commun. 2019;10(1):2906. https://doi.org/10.1038/s41467-019-10956-w.
Martin C, Zhang Y. The diverse functions of histone lysine methylation. Nat Rev Mol Cell Biol. 2005;6(11):838–49. https://doi.org/10.1038/nrm1761.
Bannister AJ, Kouzarides T. Reversing histone methylation. Nature. 2005;436(7054):1103–6. https://doi.org/10.1038/nature04048.
Kaur M, Khan MM, Kar A, Sharma A, Saxena S. CRL4-DDB1-VPRBP ubiquitin ligase mediates the stress triggered proteolysis of Mcm10. Nucleic Acids Res. 2012;40(15):7332–46. https://doi.org/10.1093/nar/gks366.
Pan Z-Q, Kentsis A, Dias DC, Yamoah K, Wu K. Nedd8 on cullin: building an expressway to protein destruction. Oncogene. 2004;23(11):1985–97. https://doi.org/10.1038/sj.onc.1207414.
Nawrocki ST, Griffin P, Kelly KR, Carew JS. MLN4924: a novel first-in-class inhibitor of NEDD8-activating enzyme for cancer therapy. Expert Opin Investig Drugs. 2012;21(10):1563–73. https://doi.org/10.1517/13543784.2012.707192.
Yang L, Chen W, Li L, Xiao Y, Fan S, Zhang Q, et al. Ddb1 Is essential for the expansion of CD4(+) helper t cells by regulating cell cycle progression and cell death. Front Immunol. 2021;12:722273. https://doi.org/10.3389/fimmu.2021.722273.
Ceuppens JL, Baroja ML, Lorre K, Van Damme J, Billiau A. Human T cell activation with phytohemagglutinin. The function of IL-6 as an accessory signal. J Immunol. 1988;141(11):3868–74.
Tran HTT, Herz C, Ruf P, Stetter R, Lamy E. Human T2R38 bitter taste receptor expression in resting and activated lymphocytes. Front Immunol. 2018;9:2949. https://doi.org/10.3389/fimmu.2018.02949.
Hildeman DA, Mitchell T, Teague TK, Henson P, Day BJ, Kappler J, et al. Reactive oxygen species regulate activation-induced T cell apoptosis. Immunity. 1999;10(6):735–44. https://doi.org/10.1016/S1074-7613(00)80072-2.
Mehta AK, Gracias DT, Croft M. TNF activity and T cells. Cytokine. 2018;101:14–8. https://doi.org/10.1016/j.cyto.2016.08.003.
Zhu J, Yamane H, Paul WE. Differentiation of effector CD4 T cell populations (*). Annu Rev Immunol. 2010;28:445–89. https://doi.org/10.1146/annurev-immunol-030409-101212.
Vallianatos CN, Iwase S. Disrupted intricacy of histone H3K4 methylation in neurodevelopmental disorders. Epigenomics. 2015;7(3):503–19. https://doi.org/10.2217/epi.15.1.
Oudhoff MJ, Antignano F, Chenery AL, Burrows K, Redpath SA, Braam MJ, et al. Intestinal epithelial cell-intrinsic deletion of Setd7 identifies role for developmental pathways in immunity to helminth infection. PLoS Pathog. 2016;12(9):e1005876. https://doi.org/10.1371/journal.ppat.1005876.
Zong Y, Shan H, Yin F, Ma X, Jiang C, Wang N, et al. Ddb1-Cullin4-Associated-Factor 1 in macrophages restricts the staphylococcus aureus-induced osteomyelitis. J Inflamm Res. 2021;14:1667–76. https://doi.org/10.2147/JIR.S307316.
Batista IAA, Helguero LA. Biological processes and signal transduction pathways regulated by the protein methyltransferase SETD7 and their significance in cancer. Signal Transduct Target Ther. 2018;3(1):19. https://doi.org/10.1038/s41392-018-0017-6.
Reinhardt RL, Khoruts A, Merica R, Zell T, Jenkins MK. Visualizing the generation of memory CD4 T cells in the whole body. Nature. 2001;410(6824):101–5. https://doi.org/10.1038/35065111.
Iovine B, Iannella ML, Bevilacqua MA. Damage-specific DNA binding protein 1 (DDB1) is involved in ubiquitin-mediated proteolysis of p27Kip1 in response to UV irradiation. Biochimie. 2011;93(5):867–75. https://doi.org/10.1016/j.biochi.2010.12.017.
Schrofelbauer B, Hakata Y, Landau NR. HIV-1 Vpr function is mediated by interaction with the damage-specific DNA-binding protein DDB1. Proc Natl Acad Sci USA. 2007;104(10):4130–5. https://doi.org/10.1073/pnas.0610167104.
Angers S, Li T, Yi X, MacCoss MJ, Moon RT, Zheng N. Molecular architecture and assembly of the DDB1-CUL4A ubiquitin ligase machinery. Nature. 2006;443(7111):590–3. https://doi.org/10.1038/nature05175.
Hesterberg RS, Beatty MS, Han Y, Fernandez MR, Akuffo AA, Goodheart WE, et al. Cereblon harnesses Myc-dependent bioenergetics and activity of CD8+ T lymphocytes. Blood. 2020;136(7):857–70. https://doi.org/10.1182/blood.2019003257.
Lidak T, Baloghova N, Korinek V, Sedlacek R, Balounova J, Kasparek P, et al. CRL4-DCAF12 ubiquitin ligase controls MOV10 RNA helicase during spermatogenesis and T cell activation. Int J Mol Sci. 2021;22(10):5394. https://doi.org/10.3390/ijms22105394.
Acknowledgements
We thank Bullet Edits Limited for the linguistic editing and proofreading of the manuscript.
Funding
This work was supported by the National Natural Science Foundation of China (32170975), Suzhou Science and Technology Project (SKY2022115), Scientific Research Project of Wuxi Municipal Health Commission (Q202201), and the Social Development-Science & Technology Demonstration Projects of Wuxi (N20201005).
Author information
Authors and Affiliations
Contributions
RW, XW, and LZ performed the experiments. LZ and YM supervised this research. LZ and YM wrote and revised the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no potential conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wu, R., Wu, X., Zou, L. et al. DDB1 regulates the activation-induced apoptosis of T cells via downregulating the expression of histone methyltransferase SETD7. Med Oncol 40, 146 (2023). https://doi.org/10.1007/s12032-023-02015-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12032-023-02015-8