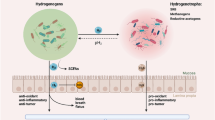
Abstract
Radiotherapy is one of the main treatments for localized primary cancer in patients. Cardiotoxicity and lung injury are two of the main side effects of oxidative stress following radiotherapy in patients with thoracic region cancer. Gliclazide (GLZ) as an antihyperglycemic drug has antioxidant, anti-inflammatory, and anti-apoptotic activities. This study aimed to evaluate the effect of GLZ in cardiotoxicity and lung injury induced by irradiation (IR). In this experimental study, 64 mice were divided into eight groups: control, GLZ (5, 10, and 25 mg/kg), IR (6 Gy), and IR + GLZ (in three doses). GLZ was administrated for 8 consecutive successive days and mice were exposed with IR on the 9th day of study. On the 10th day of study, tissue biochemical assay and at 14th day of study, histopathological assay were performed to evaluate for cardiotoxicity and lung injury. The findings revealed that IR induces atypical features in heart and lung histostructure, and oxidative stress (an increase of MDA, PC levels, and decrease of GSH content) in these tissues. GLZ administration preserved heart and lung damages and improves oxidative stress markers in mice. Data have authenticated that GLZ could protect heart and lung histostructure against oxidative stress-induced injury through inhibiting oxidative stress.






Similar content being viewed by others
Data availability
The data sets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Fuchs J, Urla C, Sparber-Sauer M, Schuck A, Leuschner I, Klingebiel T, et al. Treatment and outcome of patients with localized intrathoracic and chest wall rhabdomyosarcoma: a report of the cooperative weichteilsarkom studiengruppe (CWS). J Cancer Res Clin Oncol. 2018;144(5):925–34.
Rahi MS, Parekh J, Pednekar P, Parmar G, Abraham S, Nasir S, et al. Radiation-induced lung injury—current perspectives and management. Clin Pract. 2021;11(3):410–29.
Giuranno L, Ient J, De Ruysscher D, Vooijs MA. Radiation-induced lung injury (RILI). Front Oncol. 2019;9:877.
Bansal N, Blanco JG, Sharma UC, Pokharel S, Shisler S, Lipshultz SE. Cardiovascular diseases in survivors of childhood cancer. Cancer Metastasis Rev. 2020;39(1):55–68.
Gramatyka M, Sokół M. Radiation metabolomics in the quest of cardiotoxicity biomarkers: the review. Int J Radiat Biol. 2020;96(3):349–59.
Ping Z, Peng Y, Lang H, Xinyong C, Zhiyi Z, Xiaocheng W, et al. Oxidative stress in radiation-induced cardiotoxicity. Oxidative Med Cell Longev. 2020;2020(1):15.
Spetz J, Moslehi J, Sarosiek K. Radiation-induced cardiovascular toxicity: mechanisms, prevention, and treatment. Curr Treat Options Cardiovasc Med. 2018;20(4):1–11.
Soliman AF, Anees LM, Ibrahim DM. Cardioprotective effect of zingerone against oxidative stress, inflammation, and apoptosis induced by cisplatin or gamma radiation in rats. Naunyn Schmiedeberg’s Arch Pharmacol. 2018;391(8):819–32.
Nuszkiewicz J, Woźniak A, Szewczyk-Golec K. Ionizing radiation as a source of oxidative stress—the protective role of melatonin and vitamin D. Int J Mol Sci. 2020;21(16):5804.
Meky NH, Haggag AM, Kamal AM, Ahmed ZA. The protective effect of L-carnitine against gamma irradiation-induced cardiotoxicity in male albino rats. Egypt Acad J Biol Sci. 2017;9(2):9–20.
Vona R, Gambardella L, Cittadini C, Straface E, Pietraforte D. Biomarkers of oxidative stress in metabolic syndrome and associated diseases. Oxidative Med Cell Longev. 2019;2019(1):19.
Cikman O, Taysi S, Gulsen M, Demir E, Akan M, Diril H, et al. The radioprotective effects of caffeic acid phenethyl ester and thymoquinone on oxidative and nitrosative stress in liver tissue of rats exposed to total head irradiation. West Indian Med J. 2016. https://doi.org/10.7727/wimj.2014.176.
Derindağ G, Akgül HM, Kiziltunc A, Özkan Hİ, Özmen HK, Akgül N. Evaluation of saliva glutathione, glutathione peroxidase, and malondialdehyde levels inhead-neck radiotherapy patients. Turk J Med Sci. 2021;51(2):644–9.
Zhou B, Liu S, Yin H, Qi M, Hong M, Ren G-B. Development of Gliclazide ionic liquid and the transdermal patches: an effective and noninvasive sustained release formulation to achieve hypoglycemic effects. Eur J Pharm Sci. 2021;164:105915.
Taghizadeh F, Hosseinimehr SJ, Zargari M, Karimpour Malekshah A, Mirzaei M, Talebpour AF. Alleviation of cisplatin-induced hepatotoxicity by gliclazide: involvement of oxidative stress and caspase-3 activity. Pharmacol Res Perspect. 2021;9(3):e00788.
Mafra CAdCC, Vasconcelos RC, Medeiros CACXd, Leitão RFdC, Brito GAdC, Costa DVdS, et al. Gliclazide prevents 5-FU-induced oral mucositis by reducing oxidative stress, inflammation, and P-selectin adhesion molecules. Front Physiol. 2019;10:327.
Wu Y-b, Shi L-l, Wu Y-j, Xu W-h, Wang L, Ren M-s. Protective effect of gliclazide on diabetic peripheral neuropathy through Drp-1 mediated-oxidative stress and apoptosis. Neurosci Lett. 2012;523(1):45–9.
Zhang Y-W, Wang X, Ren X, Zhang M. Involvement of glucose-regulated protein 78 and spliced X-box binding protein 1 in the protective effect of gliclazide in diabetic nephropathy. Diabetes Res Clin Pract. 2018;146:41–7.
Taghizadeh F, Hosseinimehr SJ, Zargari M, Karimpour Malekshah A, Talebpour Amiri FB. Gliclazide attenuates cisplatin-induced nephrotoxicity through inhibiting NF-κB and caspase-3 activity. IUBMB Life. 2020;72(9):2024–33.
Tan F, Li H, Ma M, Yu Y. Protective effect of treatment with low-dose gliclazide in a model of middle cerebral artery occlusion and reperfusion in rats. Brain Res. 2014;1560:83–90.
Arafa E-SA, Mohamed WR, Zaher DM, Omar HA. Gliclazide attenuates acetic acid-induced colitis via the modulation of PPARγ, NF-κB and MAPK signaling pathways. Toxicol Appl Pharmacol. 2020;391:114919.
Pouri M, Shaghaghi Z, Ghasemi A, Hosseinimehr SJ. Radioprotective effect of gliclazide as an anti-hyperglycemic agent against genotoxicity induced by ionizing radiation on human lymphocytes. Cardiovasc Hematol Agents Med Chem. 2019;17(1):40–6.
Pan W-Q, Wang S-F, Ding B-P, Huang Z-G. Protective effects of gliclazide on myocardium of diabetic rats and its mechanism. Chin J Appl Physiol. 2020;36(5):402.
Wu Z, Wang X, Yang R, Liu Y, Zhao W, Si J, et al. Effects of carbon ion beam irradiation on lung injury and pulmonary fibrosis in mice. Exp Ther Med. 2013;5(3):771–6.
Taghizadeh F, Hosseinimehr SJ, Zargari M, Karimpour Malekshah A, Talebpour Amiri FB. Gliclazide attenuates cisplatin-induced nephrotoxicity through inhibiting NF-kappaB and caspase-3 activity. IUBMB Life. 2020;72(9):2024–33. https://doi.org/10.1002/iub.2342.
Spector T. Refinement of the coomassie blue method of protein quantitation. A simple and linear spectrophotometric assay for less than or equal to 0.5 to 50 microgram of protein. Anal Biochem. 1978;86(1):142–6.
Farzipour S, Amiri FT, Mihandoust E, Shaki F, Noaparast Z, Ghasemi A, et al. Radioprotective effect of diethylcarbamazine on radiation-induced acute lung injury and oxidative stress in mice. J Bioenerg Biomembr. 2020;52(1):39–46.
Tahamtan R, Shabestani Monfared A, Tahamtani Y, Tavassoli A, Akmali M, Mosleh-Shirazi MA, et al. Radioprotective effect of melatonin on radiation-induced lung injury and lipid peroxidation in rats. Cell J. 2015;17(1):111–20.
Zordoky BN, Radin MJ, Heller L, Tobias A, Matise I, Apple FS, et al. The interplay between genetic background and sexual dimorphism of doxorubicin-induced cardiotoxicity. Cardio-Oncology. 2016;2(1):1–11.
Sarhan H, Naoum L. Protective role of royal jelly against gamma radiation induced oxidative stress, cardio-toxicity and organ dysfunctions in male rats. Egypt J Hosp Med. 2020;78(1):62–7.
Ibrahim DM, Radwan RR, Fattah SMA. Antioxidant and antiapoptotic effects of sea cucumber and valsartan against doxorubicin-induced cardiotoxicity in rats: the role of low dose gamma irradiation. J Photochem Photobiol B. 2017;170:70–8.
Wu X, Ji H, Wang Y, Gu C, Gu W, Hu L, et al. Melatonin alleviates radiation-induced lung injury via regulation of miR-30e/NLRP3 axis. Oxidative Med Cell Longev. 2019;1:14.
Türkkan G, Willems Y, Hendriks LE, Mostard R, Conemans L, Gietema HA, et al. Idiopathic pulmonary fibrosis: current knowledge, future perspectives and its importance in radiation oncology. Radiother Oncol. 2021;155:269–77.
Hanania AN, Mainwaring W, Ghebre YT, Hanania NA, Ludwig M. Radiation-induced lung injury: assessment and management. Chest. 2019;156(1):150–62.
de Brito AA, da Silveira EC, Rigonato-Oliveira NC, Soares SS, Brandao-Rangel MAR, Soares CR, et al. Low-level laser therapy attenuates lung inflammation and airway remodeling in a murine model of idiopathic pulmonary fibrosis: relevance to cytokines secretion from lung structural cells. J Photochem Photobiol B. 2020;203:111731.
Nam J-K, Kim A-R, Choi S-H, Kim J-H, Han SC, Park S, et al. Pharmacologic inhibition of HIF-1α attenuates radiation-induced pulmonary fibrosis in a preclinical image guided radiation therapy. Int J Radiat Oncol Biol Phys. 2021;109(2):553–66.
Loubani M, Fowler A, Standen NB, Galiñanes M. The effect of gliclazide and glibenclamide on preconditioning of the human myocardium. Eur J Pharmacol. 2005;515(1–3):142–9.
Acknowledgements
This study was the subject of a Pharm D. thesis of Soroush Arzani as a student of Mazandaran University of Medical Sciences, Sari, Iran.
Funding
This study was supported by a grant from Mazandaran University of Medical Sciences, Sari, Iran (ID#8619). Seyed Jalal Hosseinimehr was received this grant.
Author information
Authors and Affiliations
Contributions
SJH designed this study. SA, FTA, SF, and SJH contributed to experiments. FTA and SJH designed and wrote the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interest
The authors declare that they have no competing interest.
Ethical approval
This experimental animal study was confirmed by the Research and Ethics Committee of Mazandaran University of Medical Sciences (ID#IR.MAZUMS.REC.1400.8619). All methods were carried out in accordance with relevant guidelines and regulations of University.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Talebpour Amiri, F., Arzani, S., Farzipour, S. et al. Radioprotective effects of gliclazide against irradiation-induced cardiotoxicity and lung injury through inhibiting oxidative stress. Med Oncol 39, 199 (2022). https://doi.org/10.1007/s12032-022-01803-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12032-022-01803-y