Abstract
Genomic instability is the hallmark of cancer. Checkpoint kinase-1 (Chk1) is required for cell cycle delay after DNA damage or blocked DNA replication. Chk1-depleted tumor cells undergo premature mitosis and apoptosis. Here we analyzed the depletion of Chk1 in normal somatic cells in the absence of DNA damage in order to investigate alternative cell cycle checkpoint mechanism(s). By means of adenoviruses, flow cytometry, immunofluorescence and Western blotting, Chk1-depleted mouse embryonic fibroblasts (MEFs) were investigated. Chk1−/− MEFs arrested at the S/G2 boundary of the cell cycle with decreased protein levels of many cell cycle key players. Cyclin B1 was predominantly cytoplasmic. Interestingly, overexpression of nuclear dominant Cyclin B1 leads to nuclear translocation and premature mitosis. Chk1−/− MEFs exhibited the absence of double-strand breaks, yet cells showed delayed DNA damage recovery with pan-nuclear immunostaining pattern of Histone H2AX. Activation of this checkpoint would elicit a senescent-like phenotype. Taken together, our elaborated data revealed the existence of an additional S/M checkpoint functioning via γH2AX signaling and cytoplasmic retention of Cyclin B1 in somatic cells.







Similar content being viewed by others
Abbreviations
- Chk1:
-
Checkpoint kinase 1
- MEF:
-
Mouse embryonic fibroblast
- Cdk1:
-
Cyclin-dependent kinase 1
- DSB:
-
Double-strand break
- Lac-z:
-
Gene encoding β-galactosidase
- ATM:
-
Ataxia telangiectasia mutated
- ATR:
-
Ataxia telangiectasia mutated and Rad3 related
- SAHF:
-
Senescence-associated heterochromatin foci
References
Abu-Odeh M, Hereema NA, Aqeilan RI. WWOX modulates the ATR-mediated DNA damage checkpoint response. Oncotarget. 2016;7(4):4344–55. doi:10.18632/oncotarget.6571.
Wayne J, Brooks T, Massey AJ. Inhibition of Chk1 with the small molecule inhibitor V158411 induces DNA damage and cell death in an unperturbed S-phase. Oncotarget. 2016. doi:10.18632/oncotarget.13119.
Shimada M, Nakanishi M. DNA damage checkpoints and cancer. J Mol Histol. 2006;37(5–7):253–60. doi:10.1007/s10735-006-9039-4.
Chen LLP, Evans TC Jr, Ettwiller LM. DNA damage is a pervasive cause of sequencing errors, directly confounding variant identification. Science. 2017;355(6326):752–6. doi:10.1126/science.aai8690.
Shimada M, Niida H, Zineldeen DH, Tagami H, Tanaka M, Saito H, et al. Chk1 is a histone H3 threonine 11 kinase that regulates DNA damage-induced transcriptional repression. Cell. 2008;132(2):221–32. doi:10.1016/j.cell.2007.12.013.
Visconti R, Della Monica R, Grieco D. Cell cycle checkpoint in cancer: a therapeutically targetable double-edged sword. J Exp Clin Cancer Res. 2016;35(1):153. doi:10.1186/s13046-016-0433-9.
Mulner-Lorillon OCH, Morales J, Bellé R, Cormier P. MAPK/ERK activity is required for the successful progression of mitosis in sea urchin embryos. Dev Biol. 2017;421(2):194–203. doi:10.1016/j.ydbio.2016.11.018.
Liu Q, Guntuku S, Cui XS, Matsuoka S, Cortez D, Tamai K, et al. Chk1 is an essential kinase that is regulated by Atr and required for the G(2)/M DNA damage checkpoint. Genes Dev. 2000;14(12):1448–59.
Massey AJ, Stokes S, Browne H, Foloppe N, Fiumana A, Scrace S, et al. Identification of novel, in vivo active Chk1 inhibitors utilizing structure guided drug design. Oncotarget. 2015;6(34):35797–812. doi:10.18632/oncotarget.5929.
Benada J, Macurek L. Targeting the checkpoint to kill cancer cells. Biomolecules. 2015;5(3):1912–37. doi:10.3390/biom5031912.
Niida H, Tsuge S, Katsuno Y, Konishi A, Takeda N, Nakanishi M. Depletion of Chk1 leads to premature activation of Cdc2-cyclin B and mitotic catastrophe. J Biol Chem. 2005;280(47):39246–52. doi:10.1074/jbc.M505009200.
Patil M, Pabla N, Dong Z. Checkpoint kinase 1 in DNA damage response and cell cycle regulation. Cell Mol Life Sci. 2013;70(21):4009–21. doi:10.1007/s00018-013-1307-3.
Jackman M, Lindon C, Nigg EA, Pines J. Active cyclin B1–Cdk1 first appears on centrosomes in prophase. Nat Cell Biol. 2003;5(2):143–8. doi:10.1038/ncb918.
Krämer A, Mailand N, Lukas C, Syljuåsen RG, Wilkinson CJ, Nigg EA, et al. Centrosome-associated Chk1 prevents premature activation of cyclin-B-Cdk1 kinase. Nat Cell Biol. 2004;6(9):884–91. doi:10.1038/ncb1165.
Horton JK, Stefanick DF, Kedar PS, Wilson SH. ATR signaling mediates an S-phase checkpoint after inhibition of poly(ADP-ribose) polymerase activity. DNA Repair (Amst). 2007;6(6):742–50. doi:10.1016/j.dnarep.2006.12.015.
Tsaponina O, Chabes A. Pre-activation of the genome integrity checkpoint increases DNA damage tolerance. Nucleic Acids Res. 2013;41(22):10371–8. doi:10.1093/nar/gkt820.
Naruyama H, Shimada M, Niida H, Zineldeen DH, Hashimoto Y, Kohri K, et al. Essential role of Chk1 in S phase progression through regulation of RNR2 expression. Biochem Biophys Res Commun. 2008;374(1):79–83. doi:10.1016/j.bbrc.2008.06.112.
Jin P, Hardy S, Morgan DO. Nuclear localization of cyclin B1 controls mitotic entry after DNA damage. J Cell Biol. 1998;141(4):875–85.
Niida H, Katsuno Y, Banerjee B, Hande MP, Nakanishi M. Specific role of Chk1 phosphorylations in cell survival and checkpoint activation. Mol Cell Biol. 2007;27(7):2572–81. doi:10.1128/MCB.01611-06.
Zineldeen DH, Wagih AA, Nakanishi M. Cloning and functional characterization of Ptpcd2 as a novel cell cycle related protein tyrosine phosphatase that regulates mitotic exit. Asian Pac J Cancer Prev. 2013;14(6):3669–76.
Olive PL, Banáth JP. The comet assay: a method to measure DNA damage in individual cells. Nat Protoc. 2006;1(1):23–9. doi:10.1038/nprot.2006.5.
Końca K, Lankoff A, Banasik A, Lisowska H, Kuszewski T, Góźdź S, et al. A cross-platform public domain PC image-analysis program for the comet assay. Mutat Res. 2003;534(1–2):15–20.
Park JH, Park EJ, Lee HS, Kim SJ, Hur SK, Imbalzano AN, et al. Mammalian SWI/SNF complexes facilitate DNA double-strand break repair by promoting gamma-H2AX induction. EMBO J. 2006;25(17):3986–97. doi:10.1038/sj.emboj.7601291.
Zineldeen DH, Shimada M, Niida H, Katsuno Y, Nakanishi M. Ptpcd-1 is a novel cell cycle related phosphatase that regulates centriole duplication and cytokinesis. Biochem Biophys Res Commun. 2009;380(3):460–6. doi:10.1016/j.bbrc.2009.01.113.
Kim YJ, Ketter R, Steudel WI, Feiden W. Prognostic significance of the mitotic index using the mitosis marker anti-phosphohistone H3 in meningiomas. Am J Clin Pathol. 2007;128(1):118–25. doi:10.1309/HXUNAG34B3CEFDU8.
Dziegielewski J, Melendy T, Beerman TA. Bleomycin-induced alterations in DNA replication: relationship to DNA damage. Biochemistry. 2001;40(3):704–11.
de Vries HI, Uyetake L, Lemstra W, Brunsting JF, Su TT, Kampinga HH, et al. Grp/DChk1 is required for G2-M checkpoint activation in Drosophila S2 cells, whereas Dmnk/DChk2 is dispensable. J Cell Sci. 2005;118(Pt 9):1833–42. doi:10.1242/jcs.02309.
Yi XTY, Lin X, Dai Y, Yang T, Yue X, Jiang X, Li X, Jiang DS, Andrade KC, Chang J. Histone methyltransferase Setd2 is critical for the proliferation and differentiation of myoblasts. Biochim Biophys Acta. 2017;1864(4):697–707. doi:10.1016/j.bbamcr.2017.01.012.
Lefebvre FABBL, Bergalet J, Lécuyer E. Biochemical fractionation of time-resolved drosophila embryos reveals similar transcriptomic alterations in replication checkpoint and histone mRNA processing mutants. J Mol Biol. 2017. doi:10.1016/j.jmb.2017.01.022.
Hagting A, Jackman M, Simpson K, Pines J. Translocation of cyclin B1 to the nucleus at prophase requires a phosphorylation-dependent nuclear import signal. Curr Biol. 1999;9(13):680–9.
Li J, Meyer AN, Donoghue DJ. Nuclear localization of cyclin B1 mediates its biological activity and is regulated by phosphorylation. Proc Natl Acad Sci USA. 1997;94(2):502–7.
Santos SD, Wollman R, Meyer T, Ferrell JE. Spatial positive feedback at the onset of mitosis. Cell. 2012;149(7):1500–13. doi:10.1016/j.cell.2012.05.028.
Araujo AR, Gelens L, Sheriff RS, Santos SD. Positive feedback keeps duration of mitosis temporally insulated from upstream cell-cycle events. Mol Cell. 2016;64(2):362–75. doi:10.1016/j.molcel.2016.09.018.
Walsh S, Margolis SS, Kornbluth S. Phosphorylation of the cyclin b1 cytoplasmic retention sequence by mitogen-activated protein kinase and Plx. Mol Cancer Res. 2003;1(4):280–9.
Xiao Z, Xue J, Gu WZ, Bui M, Li G, Tao ZF, et al. Cyclin B1 is an efficacy-predicting biomarker for Chk1 inhibitors. Biomarkers. 2008;13(6):579–96. doi:10.1080/13547500802063240.
Bryant C, Scriven K, Massey AJ. Inhibition of the checkpoint kinase Chk1 induces DNA damage and cell death in human Leukemia and Lymphoma cells. Mol Cancer. 2014;13:147. doi:10.1186/1476-4598-13-147.
Del Nagro CJ, Choi J, Xiao Y, Rangell L, Mohan S, Pandita A, et al. Chk1 inhibition in p53-deficient cell lines drives rapid chromosome fragmentation followed by caspase-independent cell death. Cell Cycle. 2014;13(2):303–14. doi:10.4161/cc.27055.
Syljuåsen RG, Sørensen CS, Hansen LT, Fugger K, Lundin C, Johansson F, et al. Inhibition of human Chk1 causes increased initiation of DNA replication, phosphorylation of ATR targets, and DNA breakage. Mol Cell Biol. 2005;25(9):3553–62. doi:10.1128/MCB.25.9.3553-3562.2005.
Chastain PD, Brylawski BP, Zhou YC, Rao S, Chu H, Ibrahim JG, et al. DNA damage checkpoint responses in the S phase of synchronized diploid human fibroblasts. Photochem Photobiol. 2014. doi:10.1111/php.12361.
Bassing CH, Suh H, Ferguson DO, Chua KF, Manis J, Eckersdorff M, et al. Histone H2AX: a dosage-dependent suppressor of oncogenic translocations and tumors. Cell. 2003;114(3):359–70.
Celeste A, Difilippantonio S, Difilippantonio MJ, Fernandez-Capetillo O, Pilch DR, Sedelnikova OA, et al. H2AX haploinsufficiency modifies genomic stability and tumor susceptibility. Cell. 2003;114(3):371–83.
Zhu F, Zykova TA, Peng C, Zhang J, Cho YY, Zheng D, et al. Phosphorylation of H2AX at Ser139 and a new phosphorylation site Ser16 by RSK2 decreases H2AX ubiquitination and inhibits cell transformation. Cancer Res. 2011;71(2):393–403. doi:10.1158/0008-5472.CAN-10-2012.
McManus KJ, Hendzel MJ. ATM-dependent DNA damage-independent mitotic phosphorylation of H2AX in normally growing mammalian cells. Mol Biol Cell. 2005;16(10):5013–25. doi:10.1091/mbc.E05-01-0065.
Motoyama N, Naka K. DNA damage tumor suppressor genes and genomic instability. Curr Opin Genet Dev. 2004;14(1):11–6. doi:10.1016/j.gde.2003.12.003.
Stiff T, O’Driscoll M, Rief N, Iwabuchi K, Löbrich M, Jeggo PA. ATM and DNA-PK function redundantly to phosphorylate H2AX after exposure to ionizing radiation. Cancer Res. 2004;64(7):2390–6.
Murga M, Bunting S, Montaña MF, Soria R, Mulero F, Cañamero M, et al. A mouse model of ATR-Seckel shows embryonic replicative stress and accelerated aging. Nat Genet. 2009;41(8):891–8. doi:10.1038/ng.420.
Toledo LI, Murga M, Zur R, Soria R, Rodriguez A, Martinez S, et al. A cell-based screen identifies ATR inhibitors with synthetic lethal properties for cancer-associated mutations. Nat Struct Mol Biol. 2011;18(6):721–7. doi:10.1038/nsmb.2076.
Fragkos M, Jurvansuu J, Beard P. H2AX is required for cell cycle arrest via the p53/p21 pathway. Mol Cell Biol. 2009;29(10):2828–40. doi:10.1128/MCB.01830-08.
Rodriguez-Bravo V, Guaita-Esteruelas S, Salvador N, Bachs O, Agell N. Different S/M checkpoint responses of tumor and non tumor cell lines to DNA replication inhibition. Cancer Res. 2007;67(24):11648–56. doi:10.1158/0008-5472.can-07-3100.
Wang WT, Catto JW, Meuth M. Differential response of normal and malignant urothelial cells to CHK1 and ATM inhibitors. Oncogene. 2014. doi:10.1038/onc.2014.221.
Varmark H, Sparks CA, Nordberg JJ, Koppetsch BS, Theurkauf WE. DNA damage-induced cell death is enhanced by progression through mitosis. Cell Cycle. 2009;8(18):2951–63.
Smetana O, Široký J, Houlné G, Opatrný Z, Chabouté ME. Non-apoptotic programmed cell death with paraptotic-like features in bleomycin-treated plant cells is suppressed by inhibition of ATM/ATR pathways or NtE2F overexpression. J Exp Bot. 2012;63(7):2631–44. doi:10.1093/jxb/err439.
Klement K, Goodarzi AA. DNA double strand break responses and chromatin alterations within the aging cell. Exp Cell Res. 2014. doi:10.1016/j.yexcr.2014.09.003.
Russo I, Silver AR, Cuthbert AP, Griffin DK, Trott DA, Newbold RF. A telomere-independent senescence mechanism is the sole barrier to Syrian hamster cell immortalization. Oncogene. 1998;17(26):3417–26. doi:10.1038/sj.onc.1202261.
Deschênes-Simard X, Lessard F, Gaumont-Leclerc MF, Bardeesy N, Ferbeyre G. Cellular senescence and protein degradation: breaking down cancer. Cell Cycle. 2014;13(12):1840–58. doi:10.4161/cc.29335.
Chitikova ZV, Gordeev SA, Bykova TV, Zubova SG, Pospelov VA, Pospelova TV. Sustained activation of DNA damage response in irradiated apoptosis-resistant cells induces reversible senescence associated with mTOR downregulation and expression of stem cell markers. Cell Cycle. 2014;13(9):1424–39. doi:10.4161/cc.28402.
Acknowledgements
We are greatly thankful for Prof. Nakanishi M. (Tokyo University, Japan) for providing Chk1 MEFs, adenoviruses and antibodies and Prof. Noeman S. (Faculty of Medicine, Tanta University, Egypt) for critical reading of the manuscript and helpful discussion.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
All authors contributed sufficiently to this work. DHZ designed the research protocol, conducted experimental work, performed statistical analysis and drafted the manuscript, NMS helped in experimental work and data analysis, and SFL participated in molecular experiments. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest.
Ethical approval
All applicable international, national and/or institutional guidelines for the care and use of animals were followed. This article does not contain any studies with human participants performed by any of the authors.
Informed consent
No humans were involved.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Fig. 1
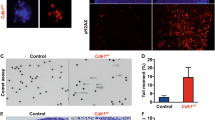
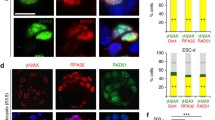
(A) Chk1flox/− MEFs were seeded into 10-cm dishes, serum starved in 0.5% FBS-containing medium at 50% for 2 days, then infected with Adeno-Cre, Adeno-Lac-Z viruses for 2 days, 15 h before the end of the second day of infection cells were released into 15% FBS medium containing 10uM Aphidicholine (to synchronize cells at S phase), on the end of the second day of infection (15 h after Aphidicholine block), NLS Cyclin B1, and tTA adenoviruses transduction was performed either alone or with Adeno-Cre (to deplete Chk1); 3 h after Cyclin B1/tTA transduction samples were collected at 3–9, 12, 24, 30 h post-transduction and trypsinized at the indicated time points and double stained with PI to visualize DNA content and FITC to detect pHH3 (mitotic index) by FACS, where a is Lac-Z, b; Cre, c; tTA/NLS alone and d; tTA/NLS/Cre. Percentages of pre-mitotic cells are encircled. B; Graph representing the mean percentages of pHH3 positive cells; error bars are ± SD. Data are representative of 3 independent experiments (TIFF 12130 kb)
Rights and permissions
About this article
Cite this article
Zineldeen, D.H., Shafik, N.M. & Li, S.F. Alternative Chk1-independent S/M checkpoint in somatic cells that prevents premature mitotic entry. Med Oncol 34, 70 (2017). https://doi.org/10.1007/s12032-017-0932-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12032-017-0932-3