Abstract
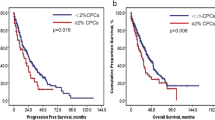
In this study, the impact of plasma cell maturity on the prognoses of multiple myeloma (MM) patients in the era of novel agents was investigated. Myeloma cell maturity was classified via immunophenotyping: myeloma cells showing mature plasma cell 1 (MPC-1)-positive and CD49e-positive cells were considered mature type; MPC-1-positive and CD49e-negative cells were considered intermediate type; and MPC-1-negative cells were considered immature type. This study included 87 newly diagnosed MM patients who were initially treated with bortezomib and/or chemotherapy. Myeloma cell maturity was a critical factor affecting overall survival (OS) in the cohort, with median OS not reached in mature-type, 50 months in intermediate-type, and 20 months in immature-type cells. Multivariate analysis showed that immature type and stage III according to the International Staging System were both independent prognostic factors affecting OS. The findings of this study demonstrate the clinical importance of myeloma cell classification according to immunophenotyping using MPC-1 and CD49e antibodies to determine patient prognosis in this era of novel therapeutic agents.

Similar content being viewed by others
References
Richardson PG, Barlogie B, Berenson J, Singhal S, Jagannath S, Irwin D, et al. A phase 2 study of bortezomib in relapsed, refractory myeloma. N Engl J Med. 2003;348:2609–17.
Richardson PG, Sonneveld P, Schuster MW, Irwin D, Stadtmauer EA, Facon T, et al. Bortezomib or high-dose dexamethasone for relapsed multiple myeloma. N Engl J Med. 2005;352:2487–98.
Dimopoulos M, Spencer A, Attal M, Prince HM, Harousseau J-L, Dmoszynska A, et al. Lenalidomide plus dexamethasone for relapsed or refractory multiple myeloma. N Engl J Med. 2007;357:2123–32.
San Miguel JF, Schlag R, Khuageva NK, Dimopoulos MA, Shpilberg O, Kropff M, et al. Persistent overall survival benefit and no increased risk of second malignancies with bortezomib-melphalan-prednisone versus melphalan-prednisone in patients with previously untreated multiple myeloma. J Clin Oncol. 2013;31:448–55.
Mateos M-V, Richardson PG, Schlag R, Khuageva NK, Dimopoulos MA, Shpilberg O, et al. Bortezomib plus melphalan and prednisone compared with melphalan and prednisone in previously untreated multiple myeloma: updated follow-up and impact of subsequent therapy in the phase III VISTA trial. J Clin Oncol. 2010;28:2259–66.
Avet-Loiseau H, Leleu X, Roussel M, Moreau P, Guerin-Charbonnel C, Caillot D, et al. Bortezomib plus dexamethasone induction improves outcome of patients with t(4;14) myeloma but not outcome of patients with del(17p). J Clin Oncol. 2010;28:4630–4.
Chang H, Trieu Y, Qi X, Xu W, Stewart KA, Reece D. Bortezomib therapy response is independent of cytogenetic abnormalities in relapsed/refractory multiple myeloma. Leuk Res. 2007;31:779–82.
Jagannath S, Richardson PG, Sonneveld P, Schuster MW, Irwin D, Stadtmauer EA, et al. Bortezomib appears to overcome the poor prognosis conferred by chromosome 13 deletion in phase 2 and 3 trials. Leukemia. 2007;21:151–7.
Sagaster V, Ludwig H, Kaufmann H, Odelga V, Zojer N, Ackermann J, et al. Bortezomib in relapsed multiple myeloma: response rates and duration of response are independent of a chromosome 13q-deletion. Leukemia. 2007;21:164–8.
Bartl R, Frisch B, Burkhardt R, Fateh-Moghadam A, Mahl G, Gierster P, et al. Bone marrow histology in myeloma: its importance in diagnosis, prognosis, classification and staging. Br J Haematol. 1982;51:361–75.
Bartl R, Frisch B, Fateh-Moghadam A, Kettner G, Jaeger K, Sommerfeld W. Histologic classification and staging of multiple myeloma: a retrospective and prospective study of 674 cases. Am J Clin Pathol. 1987;87:342–55.
Harada H, Kawano MM, Huang N, Harada Y, Iwato K, Tanabe O, et al. Phenotypic difference of normal plasma cells from mature myeloma cells. Blood. 1993;81:2658–63.
Otsuyama K, Asaoku H, Kawano MM. An increase in MPC-1- and MPC-1-CD45+ immature myeloma cells in the progressive states of bone marrow plasmacytosis: the revised phenotypic classification of monoclonal marrow plasmacytosis (MOMP-2005). Int J Hematol. 2006;83:39–43.
Kawano MM, Mihara K, Tsujimoto T, Huang N, Kuramoto A. A new phenotypic classification of bone marrow plasmacytosis. Int J Hematol. 1995;61:179–88.
Fujii R, Ishikawa H, Mahmoud MS, Asaoku H, Kawano MM. MPC-1-CD49e- immature myeloma cells include CD45+ subpopulations that can proliferate in response to IL-6 in human myelomas. Br J Haematol. 1999;105:131–40.
Bene MC, Castoldi G, Knapp W, Ludwig WD, Matutes E, Orfao A, et al. Proposals for the immunological classification of acute leukemias” European group for the immunological characterization of leukemias (EGIL). Leukemia. 1995;9:1783–6.
Iriyama N, Asou N, Miyazaki Y, Yamaguchi S, Sato S, Sakura T, et al. Normal karyotype acute myeloid leukemia with the CD7+ CD15 + CD34+ HLA-DR+ immunophenotype is a clinically distinct entity with a favorable outcome. Ann Hematol. 2014;93:957–63.
Kanda Y. Investigation of the freely available easy-to-use software “EZR” for medical statistics. Bone Marrow Transpl. 2013;48:452–8.
Kuroda Y, Sakai A, Okikawa Y, Munemasa S, Katayama Y, Hyodo H, et al. The maturation of myeloma cells correlates with sensitivity to chemotherapeutic agents. Int J Hematol. 2005;81:335–41.
Niesvizky R, Flinn IW, Rifkin R, Gabrail N, Charu V, Clowney B, et al. Community-based phase IIIB trial of three UPFRONT bortezomib-based myeloma regimens. J Clin Oncol. 2015;33:3921–9.
Kumar S, Flinn I, Richardson PG, Hari P, Callander N, Noga SJ, et al. Randomized, multicenter, phase 2 study (EVOLUTION) of combinations of bortezomib, dexamethasone, cyclophosphamide, and lenalidomide in previously untreated multiple myeloma. Blood. 2012;119:4375–82.
Mujtaba T, Dou QP. Advances in the understanding of mechanisms and therapeutic use of bortezomib. Discov Med. 2011;12:471–80.
Vincenz L, Jäger R, O’Dwyer M, Samali A. Endoplasmic reticulum stress and the unfolded protein response: targeting the Achilles heel of multiple myeloma. Mol Cancer Ther. 2013;12:831–43.
Obeng EA, Carlson LM, Gutman DM, Harrington WJ, Lee KP, Boise LH. Proteasome inhibitors induce a terminal unfolded protein response in multiple myeloma cells. Blood. 2006;107:4907–16.
Lin K-I, Tunyaplin C, Calame K. Transcriptional regulatory cascades controlling plasma cell differentiation. Immunol Rev. 2003;194:19–28.
Iwakoshi NN, Lee A-H, Vallabhajosyula P, Otipoby KL, Rajewsky K, Glimcher LH. Plasma cell differentiation and the unfolded protein response intersect at the transcription factor XBP-1. Nat Immunol. 2003;4:321–9.
Munshi NC, Hideshima T, Carrasco D, Shammas M, Auclair D, Davies F, et al. Identification of genes modulated in multiple myeloma using genetically identical twin samples. Blood. 2004;103:1799–806.
Bagratuni T, Wu P, Gonzalez de Castro D, Davenport EL, Dickens NJ, Walker BA, et al. XBP1s levels are implicated in the biology and outcome of myeloma mediating different clinical outcomes to thalidomide-based treatments. Blood. 2010;116:250–3.
Gambella M, Rocci A, Passera R, Gay F, Omedè P, Crippa C, et al. High XBP1 expression is a marker of better outcome in multiple myeloma patients treated with bortezomib. Haematologica. 2014;99:e14–6.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
KM and YH received lecture fees and honoraria from Celgene K.K. and Janssen Pharmaceutical K.K. MT received honoraria from Janssen Pharmaceutical K.K. The remaining authors have no competing interests to declare.
Rights and permissions
About this article
Cite this article
Iriyama, N., Miura, K., Hatta, Y. et al. Plasma cell maturity as a predictor of prognosis in multiple myeloma. Med Oncol 33, 87 (2016). https://doi.org/10.1007/s12032-016-0803-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12032-016-0803-3