Abstract
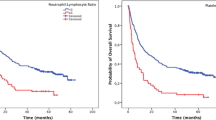
Accurate predictors of survival for patients with advanced gastric cancer treated with neoadjuvant chemotherapy are currently lacking. In this study, we aimed to evaluate the prognostic significance of the neutrophil–lymphocyte ratio (NLR) in patients with stage III–IV gastric cancer who received neoadjuvant chemotherapy FOLFOX 4 as neoadjuvant chemotherapy. We enrolled 70 patients with stage III–IV cancer stomach in this study. Patients received FOLFOX 4 as neoadjuvant chemotherapy. Blood sample was collected before chemotherapy. The NLR was divided into two groups: high (>3) and low (≤3). Univariate analysis on progression-free survival (PFS) and overall survival (OS) was performed using the Kaplan–Meier and log-rank tests, and multivariate analysis was conducted using the Cox proportional hazards regression model. The toxicity was evaluated according to National Cancer Institute Common Toxicity Criteria. The univariate analysis showed that PFS and OS were both worse for patients with high NLR than for those with low NLR before chemotherapy (median PFS 28 and 44 months, respectively, P = 0.001; median OS 30 and 48 months, P = 0.001). Multivariate analysis showed that NLRs before chemotherapy were independent prognostic factors of OS but not for progression-free survival. NLR may serve as a potential biomarker for survival prognosis in patients with stage III–IV gastric cancer receiving neoadjuvant chemotherapy. The FOLFOX 4 demonstrated an acceptable toxicity.




Similar content being viewed by others
References
Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D. Global cancer statistics. CA Cancer J Clin. 2011;61:69.
Siegel R, Ma J, Zou Z, Jemal A. Cancer statistics, 2014. CA Cancer J Clin. 2014;64:9.
Carlomagno C, Matano E, Bianco R, Cimminiello C, Prudente A, Pagliarulo C, et al. Adjuvant FOLFOX-4 in patients with radically resected gastric cancer: tolerability and prognostic factors. Exp Ther Med. 2010;1:611–7.
Shirai O, Ohmiya N, Taguchi A, Nakamura M, Kawashima H, Miyahara R, et al. P53, P21 and P73 gene polymorphisms in gastric carcinoma. Hepatogastroenterology. 2010;11:1595–601.
Kim KH, Kwon HC, Oh SY, Kim SH, Lee S, Kwon KA, et al. Clinicopathological significance of ERCC1, thymidylate synthase and glutathione S-transferase P1 expression for advanced gastric cancer patients receiving adjuvant 5-Fu and cisplatin chemotherapy. Biomarkers. 2011;11:74–82.
Elinav E, Nowarski R, Thaiss CA, Hu B, Jin C, Flavell RA. Inflammation-induced cancer: crosstalk between tumors, immune cells and microorganisms. Nat Rev Cancer. 2013;13(11):759–71.
Borsig L, Wolf MJ, Roblek M, Lorentzen A, Heikenwalder M. Inflammatory chemokines and metastasis–tracing the accessory. Oncogene. 2014;33(25):3217–24.
Hirashima M, Higuchi S, Sakamoto K, Nishiyama T, Okada H. The ratio of neutrophils to lymphocytes and the phenotypes of neutrophils in patients with early gastric cancer. J Cancer Res Clin Oncol. 1998;11:329–34.
Yamanaka T, Matsumoto S, Teramukai S, Ishiwata R, Nagai Y, Fukushima M. The baseline ratio of neutrophils to lymphocytes is associated with patient prognosis in advanced gastric cancer. Oncology. 2007;11:215–20.
Shimada H, Tajiguchi N, Kainuma O, Soda H, Ikeda A, Cho A, Miyazaki A, Gunji H, Yamamoto H, Nagata M. High preoperative neutrophil-lymphocyte ratio predicts poor survival in patients with gastric cancer. Gastric Cancer. 2010;11:170–6.
Aliustaoglu M, Bilici A, Ustaalioglu BB, Konya V, Gucun M, Seker M, et al. The effect of peripheral blood values on prognosis of patients with locally advanced gastric cancer before treatment. Med Oncol. 2010;11:1060–5.
Jung MR, Park YK, Jeong O, Seon JW, Ryu SY, Kim DY, et al. Elevated preoperative neutrophil to lymphocyte ratio predicts poor survival following resection in late stage gastric cancer. J Surg Oncol. 2011;11:504–10.
Ha TK, Kim HJ, Kwon SJ. Does the new UICC/AJCC TNM staging system (7th Edition) improve assessing prognosis in gastric cancer compared to the old system (6th Edition)? J Korean Gastric Cancer Assoc. 2009;9:159–66.
National Cancer Institute NIoH. US department of health and human services common terminology criteria for adverse events CTCAE, version 4. Washington DC: National Cancer Institute; 2009.
Caussanel JP, Lévi F, Brienza S, Misset JL, Itzhaki M, Adam R, et al. Phase I trial of 5-day continuous venous infusion of oxaliplatin at circadian rhythm modulated rate compared with constant rate. J Natl Cancer Inst. 1990;82:1046–50.
Eisenhauer EA, Therasse P, Bogaerts J, Schwartz LH, Sargent D, Ford R, et al. New response evaluation criteria in solid tumours: revised RESIST guideline (version 1.1). Eur J Cancer. 2009;11:228–47.
Bland JM, Altman DG. The log rank test. BMJ. 2008;328(7447):1073.
Schuhmacher C, Reim D, Novotny A. Neoadjuvant treatment for gastric cancer. J Gastric Cancer. 2013;13(2):73–8.
Ji JF, Yu Z, Zhong XN, Wu XJ, Wu QZ, Bu D, et al. Oxaliplatin-based regimen as neoadjuvant chemotherapy for Chinese patients with advanced gastric cancer: preliminary results of a phase II study. J Clin Oncol. 2004;22(14S):4184.
Yan D, Dai H. FOLFOX regimen in the patients with locally advanced or metastatic gastric cancer. Zhonghua Zhong Liu Za Zhi. 2009;31(3):217–9.
De Vita F, Orditura M, Matano E, Bianco R, Carlomagno C, Infusino S, et al. A phase II study of biweekly oxaliplatin plus infusional 5-fluorouracil and folinic acid (FOLFOX-4) as first-line treatment of advanced gastric cancer patients. Br J Cancer. 2005;92(9):1644–9.
Oh SY, Kwon HC, Seo BG, Kim SH, Kim JS, Kim HJ. A phase II study of oxaliplatin with low dose leucovorin and bolus and continuous infusion 5-fluorouracil (modified FOLFOX-4) as first line therapy for patients with advanced gastric cancer. Acta Oncol. 2007;46(3):336–41.
Zhu X, Leaw J, Gu W, Qian Y, Du H, Wang B, et al. Phase II clinical trial of advanced and metastatic gastric cancer based on continuous infusion of 5-fluorouracil combined with epirubicin and oxaliplatin. J Cancer Res Clin Oncol. 2008;134(9):929–36.
Lee S, Oh SY, Kim SH, Lee JH, Kim MC, et al. Prognostic significance of neutrophil lymphocyte ratio and platelet lymphocyte ratio in advanced gastric cancer patients treated with FOLFOX chemotherapy. BMC Cancer. 2013;22(13):350.
Jin H, Zhang G, Liu X, Liu X, Chen C, Yu H, et al. Blood neutrophil-lymphocyte ratio predicts survival for stages III–IV gastric cancer treated with neoadjuvant chemotherapy. World J Surg Oncol. 2013;24(11):112.
Yuan D, Zhu K, Li K, Yan R, Jia Y, Dang C, Yuan D. The preoperative neutrophil-lymphocyte ratio predicts recurrence and survival among patients undergoing R0 resections of adenocarcinomas of the esophagogastric junction. J Surg Oncol. 2014;110(3):333–40.
Seo HY, Kim DS, Choi YS, Sung HJ, Park KH, Choi IK, et al. Treatment outcomes of oxaliplatin, 5-FU, and leucovorin as salvage therapy for patients with advanced or metastatic gastric cancer: a retrospective analysis. Cancer Chemother Pharmacol. 2009;63(3):433–9.
Kim YJ, Goh PG, Kim ES, Lee SY, Moon HS, Lee ES, et al. Comparison of the toxicities and efficacies of the combination chemotherapy regimens in advanced gastric cancer patients who achieved complete response after chemotherapy. Korean J Gastroenterol. 2011;58(6):311–7.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
el Aziz, L.M.A. Blood neutrophil–lymphocyte ratio predicts survival in locally advanced cancer stomach treated with neoadjuvant chemotherapy FOLFOX 4. Med Oncol 31, 311 (2014). https://doi.org/10.1007/s12032-014-0311-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12032-014-0311-2