Abstract
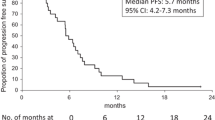
We investigated the efficacy and safety of a new second-line chemotherapy of combining folinic acid, 5-fluorouracil and irinotecan (FOLFIRI) with both panitumumab and bevacizumab to treat patients with metastatic colorectal cancer (mCRC). Patients with mCRC and unsuccessful previous oxaliplatin-based chemotherapy were included in the study. The FOLFIRI arm was given FOLFIRI only. The FOLFIRI+PB arm was given panitumumab (3 mg/kg) and bevacizumab (3 mg/kg) plus FOLFIRI every other week. Between 2009 and 2013, 155 and 137 patients were included in the FOLFIRI arm and FOLFIRI+PB arm, respectively. The response rate was 40.1 % for FOLFIRI+PB arm versus 30.1 % for FOLFIRI arm. The disease-controlled rate in FOLFIRI+PB arm was improved to 62.2 from 50.2 % in FOLFIRI arm. The median overall survival was 13.9 months in FOLFIRI+PB arm as compared to 10.7 months in FOLFIRI arm. A series of adverse events were comparable between two arms, whereas some of the antibody therapy-associated toxicities were observed in FOLFIRI+PB arm. The new strategy of combining panitumumab and bevacizumab with FOLFIRI as second-line chemotherapy for patients with mCRC is safe and feasible.

Similar content being viewed by others
References
Jemal A, Siegel R, Xu J, Ward E. Cancer statistics, 2010. CA Cancer J Clin. 2010;60(5):277–300.
Huxley RR, Ansary-Moghaddam A, Clifton P, Czernichow S, Parr CL, Woodward M. The impact of dietary and lifestyle risk factors on risk of colorectal cancer: a quantitative overview of the epidemiological evidence. Int J Cancer. 2009;125(1):171–80.
Fuchs CS, Marshall J, Mitchell E, Wierzbicki R, Ganju V, Jeffery M, Schulz J, Richards D, Soufi-Mahjoubi R, Wang B, et al. Randomized, controlled trial of irinotecan plus infusional, bolus, or oral fluoropyrimidines in first-line treatment of metastatic colorectal cancer: results from the BICC-C Study. J Clin Oncol. 2007;25(30):4779–86.
Fuchs CS, Moore MR, Harker G, Villa L, Rinaldi D, Hecht JR. Phase III comparison of two irinotecan dosing regimens in second-line therapy of metastatic colorectal cancer. J Clin Oncol. 2003;21(5):807–14.
Roque IFM, Sola I, Martin-Richard M, Lopez JJ, Bonfill Cosp X. Second-line chemotherapy in advanced and metastatic CRC. Cochrane Database Syst Rev. 2009;2:CD00687.
Cohn AL, Shumaker GC, Khandelwal P, Smith DA, Neubauer MA, Mehta N, Richards D, Watkins DL, Zhang K, Yassine MR. An open-label, single-arm, phase 2 trial of panitumumab plus FOLFIRI as second-line therapy in patients with metastatic colorectal cancer. Clin Colorectal Cancer. 2011;10(3):171–7.
Stintzing S, Fischer von Weikersthal L, Decker T, Vehling-Kaiser U, Jager E, Heintges T, Stoll C, Giessen C, Modest DP, Neumann J, et al. FOLFIRI plus cetuximab versus FOLFIRI plus bevacizumab as first-line treatment for patients with metastatic colorectal cancer-subgroup analysis of patients with KRAS: mutated tumours in the randomised German AIO study KRK-0306. Ann Oncol 2012;23(7):1693–99.
Mitchell EP, Piperdi B, Lacouture ME, Shearer H, Iannotti N, Pillai MV, Xu F, Yassine M. The efficacy and safety of panitumumab administered concomitantly with FOLFIRI or Irinotecan in second-line therapy for metastatic colorectal cancer: the secondary analysis from STEPP (Skin Toxicity Evaluation Protocol With Panitumumab) by KRAS status. Clin Colorectal Cancer. 2011;10(4):333–9.
Hurwitz HI, Fehrenbacher L, Hainsworth JD, Heim W, Berlin J, Holmgren E, Hambleton J, Novotny WF, Kabbinavar F. Bevacizumab in combination with fluorouracil and leucovorin: an active regimen for first-line metastatic colorectal cancer. J Clin Oncol. 2005;23(15):3502–8.
Moriwaki T, Bando H, Takashima A, Yamazaki K, Esaki T, Yamashita K, Fukunaga M, Miyake Y, Katsumata K, Kato S, et al. Bevacizumab in combination with irinotecan, 5-fluorouracil, and leucovorin (FOLFIRI) in patients with metastatic colorectal cancer who were previously treated with oxaliplatin-containing regimens: a multicenter observational cohort study (TCTG 2nd-BV study). Med Oncol. 2012;29(4):2842–8.
Peeters M, Price TJ, Cervantes A, Sobrero AF, Ducreux M, Hotko Y, Andre T, Chan E, Lordick F, Punt CJ, et al. Randomized phase III study of panitumumab with fluorouracil, leucovorin, and irinotecan (FOLFIRI) compared with FOLFIRI alone as second-line treatment in patients with metastatic colorectal cancer. J Clin Oncol. 2010;28(31):4706–13.
Hocking CM, Price TJ. Panitumumab in the management of patients with KRAS wild-type metastatic colorectal cancer. Therap Adv Gastroenterol. 2014;7(1):20–37.
Petrelli F, Borgonovo K, Cabiddu M, Ghilardi M, Lonati V, Maspero F, Sauta MG, Beretta GD, Barni S. FOLFIRI-bevacizumab as first-line chemotherapy in 3500 patients with advanced colorectal cancer: a pooled analysis of 29 published trials. Clin Colorectal Cancer. 2013;12(3):145–51.
Beretta GD, Petrelli F, Stinco S, Cabiddu M, Ghilardi M, Squadroni M, Borgonovo K, Barni S. FOLFIRI+bevacizumab as second-line therapy for metastatic colorectal cancer pretreated with oxaliplatin: a pooled analysis of published trials. Med Oncol. 2013;30(1):486.
Eisenhauer EA, Therasse P, Bogaerts J, Schwartz LH, Sargent D, Ford R, Dancey J, Arbuck S, Gwyther S, Mooney M, et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer. 2009;45(2):228–47.
Tournigand C, Andre T, Achille E, Lledo G, Flesh M, Mery-Mignard D, Quinaux E, Couteau C, Buyse M, Ganem G, et al. FOLFIRI followed by FOLFOX6 or the reverse sequence in advanced colorectal cancer: a randomized GERCOR study. J Clin Oncol. 2004;22(2):229–37.
Muro K, Boku N, Shimada Y, Tsuji A, Sameshima S, Baba H, Satoh T, Denda T, Ina K, Nishina T, et al. Irinotecan plus S-1 (IRIS) versus fluorouracil and folinic acid plus irinotecan (FOLFIRI) as second-line chemotherapy for metastatic colorectal cancer: a randomised phase 2/3 non-inferiority study (FIRIS study). Lancet Oncol. 2010;11(9):853–60.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Xie, S., Han, G., Fan, Z. et al. Safety and efficacy of second-line treatment with folinic acid, 5-fluorouracil and irinotecan (FOLFIRI) in combination of panitumumab and bevacizumab for patients with metastatic colorectal cancer. Med Oncol 31, 35 (2014). https://doi.org/10.1007/s12032-014-0035-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12032-014-0035-3