Abstract
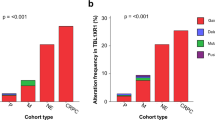
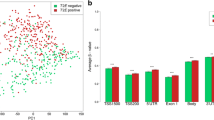
The copy number gain of genes in chromosomal region 8q21–24 has been demonstrated to be associated with genesis and progression of prostate cancer (PCa). The aim of this study was to identify novel and effective molecular markers in this chromosomal region for PCa. The differentially expressed genes in PCa specimens were screened by gene microarray analysis, which was validated by RT-QPCR analysis. Then, the DNA qPCR analysis was carried out to detect the copy number changes of these differentially expressed genes. Moreover, the clinical significance of candidate markers (MYC and E2F5) in PCa were further determined. E2F5 and MYC were identified as candidate markers in PCa tissues and PCa cell lines. The DNA qPCR revealed the significant copy number gains of E2F5 and MYC in PCa tissues but not in PCa cell lines. In addition, Western blot analysis and immunohistochemical staining both found the significant higher expression of E2F5 and MYC proteins in PCa tissues than those in adjacent benign specimens (all P < 0.01). Moreover, the overexpression of E2F5 protein was significantly associated with a high Gleason score (P < 0.01), an advanced clinical stage (P = 0.01), a positive metastasis (P < 0.01) and PSA Failure (P < 0.01). The overexpression of MYC was more frequently found in PCa tissues with positive metastasis (P = 0.02) and PSA failure (P = 0.02). Interestingly, there was a close correlation in the expression level of MYC in PCa tissues with that of E2F5 (r s = 0.5, P ≤ 0.001). Our data offers the convincing evidence that the copy number gains of E2F5 and MYC may play an important role in genesis and progression of PCa. Especially, E2F5 may be a novel potential candidate marker for malignant PCa.





Similar content being viewed by others
References
Kim JH, Dhanasekaran SM, Mehra R, et al. Integrative analysis of genomic aberrations associated with prostate cancer progression. Cancer Res. 2007;67:8229–39.
Rubin MA, Varambally S, Beroukhim R, et al. Overexpression, amplification, and androgen regulation of TPD52 in prostate cancer. Cancer Res. 2004;64:3814–22.
Hav M, Libbrecht L, Ferdinande L, et al. MDM2 gene amplification and protein expressions in colon carcinoma: is targeting MDM2 a new therapeutic option? Virchows Arch. 2011;458:197–203.
Tørring N, Borre M, Sørensen KD, Andersen CL, Wiuf C, Ørntoft TF. Genome-wide analysis of allelic imbalance in prostate cancer using the Affymetrix 50 K SNP mapping array. Br J Cancer. 2007;96:499–506.
Hawthorn L, Luce J, Stein L, Rothschild J. Integration of transcript expression, copy number and LOH analysis of infiltrating ductal carcinoma of the breast. BMC Cancer. 2010;10:460.
Quinn DI, Henshall SM, Sutherland RL. Molecular markers of prostate cancer outcome. Eur J Cancer. 2005;41:858–87.
Kote-Jarai Z, Olama AA, Giles GG, et al. Seven prostate cancer susceptibility loci identified by a multi-stage genome-wide association study. Nat Genet. 2011;43:785–91.
Wolter H, Trijic D, Gottfried HW, Mattfeldt T. Chromosomal changes in incidental prostatic carcinomas detected by comparative genomic hybridization. Eur Urol. 2002;41:328–34.
Taylor BS, Schultz N, Hieronymus H, et al. Integrative genomic profiling of human prostate cancer. Cancer Cell. 2010;18:11–22.
Chen JH, He HC, Jiang FN, et al. Analysis of the specific pathways and networks of prostate cancer for gene expression profiles in Chinese Population. Med Oncol. 2012;29:1972–84.
Han ZD, Zhang YQ, He HC, et al. Identification of novel serological tumor markers for human prostate cancer using integrative transcriptome and proteome analysis. Med Oncol. 2012;29:2877–88.
Zhong WD, Liang YX, Lin SX, Lin SX, Li L, et al. Expression of CD147 is associated with prostate cancer progression. Int J Cancer. 2012;130:300–8.
Ribeiro FR, Henrique R, Martins AT, Jerónimo C, Teixeira MR. Relative copy number gain of MYC in diagnostic needle biopsies is an independent prognostic factor for prostate cancer patients. Eur Urol. 2007;52:116–25.
Umemura S, Shirane M, Takekoshi S, et al. Overexpression of E2F–5 correlates with a pathological basal phenotype and a worse clinical outcome. Br J Cancer. 2009;100:764–71.
Jackson-Cook C, Zou Y, Turner K, Astbury C, Ware J. A novel tumorigenic human prostate epithelial cell line (M2205): molecular cytogenetic characterization demonstrates C-MYC amplification and jumping translocations. Cancer Genet Cytogenet. 2003;141:56–64.
Reiter RE, Sato I, Thomas G, et al. Co-amplification of prostate stem cell antigen (PSCA) and MYC in locally advanced prostate cancer. Genes Chromosom Cancer. 2000;27:95–103.
Chen H, Liu W, Roberts W, et al. 8q24 allelic imbalance and MYC gene copy number in primary prostate cancer. Prostate Cancer Prostatic Dis. 2010;13:238–43.
Pomerantz MM, Beckwith CA, Regan MM, et al. Evaluation of the 8q24 prostate cancer risk locus and MYC expression. Cancer Res. 2009;69:5568–74.
Gurel B, Iwata T, Koh CM, et al. Nuclear MYC protein overexpression is an early alteration in human prostate carcinogenesis. Mod Pathol. 2008;21:1156–67.
Ahmadiyeh N, Pomerantz MM, Grisanzio C, et al. 8q24 prostate, breast, and colon cancer risk loci show tissue-specific long-range interaction with MYC. Proc Natl Acad Sci USA. 2010;107:9742–6.
Koh CM, Bieberich CJ, Dang CV, Nelson WG, Yegnasubramanian S, De Marzo AM. MYC and prostate cancer. Genes Cancer. 2010;1:617–28.
Reimer D, Sadr S, Wiedemair A, et al. Clinical relevance of E2F family members in ovarian cancer–an evaluation in a training set of 77 patients. Clin Cancer Res. 2007;13:144–51.
Campanero MR, Armstrong M, Flemington E. Distinct cellular factors regulate the c-myb promoter through its E2F element. Mol Cell Biol. 1999;19:8442–50.
Trimarchi JM, Lees JA. Sibling rivalry in the E2F family. Nat Rev Mol Cell Biol. 2002;3:11–20.
Polanowska J, Le Cam L, Orsetti B, et al. Human E2F5 gene is oncogenic in primary rodent cells and is amplified in human breast tumors. Genes Chromosom Cancer. 2000;28:126–30.
Kowalik TF. Smad about E2F. TGFbeta repressionof c-Myc via a Smad3/E2F/p107 complex. Mol Cell. 2002;10:7–8.
Xie L, Law BK, Aakre ME, et al. Transforming growth factor beta-regulated gene expression in a mouse mammary gland epithelial cell line. Breast Cancer Res. 2003;5:R187–98.
Chen CR, Kang Y, Siegel PM, Massagué J. E2F4/5 and p107 as Smad cofactors linking the TGFbeta receptor to c-myc repression. Cell. 2002;110:19–32.
Acknowledgments
This work was supported by grants from the National Natural Science Foundation of China (81170699), the Guangzhou Municipal Science and Technology Key Project (11C23150711), the Key Projects of Bureau of Health in Guangzhou Municipality (201102A212015), Projects of Guangdong Key Laboratory of Clinical Molecular Medicine and Diagnostics, and the Projects of Guangdong Key Laboratory of Urology (2010A060801016).
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
Jin Zhao, Xin-yang Wu and Xiao-hui Ling contributed equally to this article.
Rights and permissions
About this article
Cite this article
Zhao, J., Wu, Xy., Ling, Xh. et al. Analysis of genetic aberrations on chromosomal region 8q21–24 identifies E2F5 as an oncogene with copy number gain in prostate cancer. Med Oncol 30, 465 (2013). https://doi.org/10.1007/s12032-013-0465-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12032-013-0465-3