Abstract
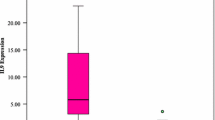
Human leukocyte antigen G (HLA-G) is a non-classical major histocompatibility class Ib antigen with multiple immune regulatory functions including the induction of immune tolerance in malignancies. The goal of our study was to investigate the expression of membrane form of HLA-G in acute lymphoblastic leukemia (ALL) before and after therapy in a trial to evaluate its role as a tumor escape mechanism and prognosis. So we measured its expression by reverse transcription (RT)-PCR in peripheral blood mononuclear cells of 25 (ALL) patients and 15 healthy controls and correlated our findings with a variety of clinical and laboratory variables and two important cytokines, IL-10 and INF-γ, and with natural killer (NK) cells. Serum levels of IL-10 and INF-γ were measured by ELISA. NK cells were quantitated by flow cytometry. The best cutoff values for the investigated markers were determined by ROC curve. The current study showed that membrane-bound HLA-G expression levels and positivity rates above the cutoff value 0.37 were significantly higher in ALL patients at diagnosis compared to after therapy and both showed significant higher levels than in normal control group (P < 0.01). Moreover, IL-10 and INF-γ serum levels were significantly elevated in ALL patients at time of diagnosis compared to healthy controls with a significant reduction in their levels in ALL patients after receiving chemotherapy. Membrane HLA-G expression showed a significant positive correlation with lactate dehydrogenase, peripheral and bone marrow blast cells and with IL-10 and INF-γ. The positive correlation of membrane HLA-G expression with both IL-10 and INF-γ serum levels supports the speculation that both cytokines may be involved in the control of HLA-G expression. HLA-G showed a negative correlation with NK cells confirming its importance in tumor escape through down-regulation of NK cells. In conclusion, HLA-G expression could be used as a prognostic tumor marker to monitor disease state and improvement in ALL.


Similar content being viewed by others
References
Faderl S, O’Brien S, Pui CH, Stock W, Wetzler M, Hoelzer D, Kantarjian HM. Adult acute lymphoblastic leukemia: concepts and strategies. Cancer. 2010;116(5):1165–76.
Akinloye O, Adegbenro M, Olaniyi J, Rahamon S, Anifowose A, Awosika E, Arinola G. Plasma interferon-gamma and IL-4, immunoglobulin classes and nitric oxide in nigerians with acute leukaemia. ©Sierra Leone J Biomed Res. 2011;3(3):138–43.
Mocellin S, Wang E, Marincola FM. Cytokines and immune response in the tumor microenvironment. J Immunother. 2001;24:392–407.
Carosella ED, Moreau P, Lemaoult J, Rouas-Freiss N. HLA-G: from biology to clinical benefits. Trends Immunol. 2008;29(3):125–32.
LeMaoult J, Zafaranloo K, Le Danff C, Carosella ED. HLA-G up-regulates ILT2, ILT3, ILT4, and KIR2DL4 in antigen presenting cells, NK cells, and T cells. FASEB J. 2005;19(6):662–4.
Kovats S, Main EK, Librach C, et al. A class I antigen, HLA-G, expressed in human trophoblasts. Science. 1990;248:220–3.
Blaschitz A, Lenfant F, Mallet V, et al. Endothelial cells in chorionic fetal vessels of first trimester placenta express HLA-G. Eur J Immunol. 1997;27:3380–8.
Yang Y, Chu W, Geraghty DE, Hunt JS. Expression of HLA-G in human mononuclear phagocytes and selective induction by IFN-gamma. J Immunol. 1996;156:4224–31.
Crisa L, McMaster MT, Ishii JK, et al. Identification of a thymic epithelial cell subset sharing expression of the class Ib HLA-G molecule with fetal trophoblasts. J Exp Med. 1997;186:289–98.
Le Rond S, Le Maoult J, Créput C, Menier C, Deschamps M, Le Friec G, Amiot L, Durrbach A, Dausset J, Carosella ED. Rouas-FreissN. Alloreactive CD4+ and CD8+ T cells express the immunotolerant HLA-G molecule in mixed lymphocyte reactions: in vivo implications in transplanted patients. Eur J Immunol. 2004;34(3):649–60.
Cabestre A, Moreau P, Riteau B, et al. HLA-G expression in human melanoma cells: protection from NK cytolysis. J Reprod Immunol. 1999;43:183–93.
Urosevic M, Willers J, Mueller B, Kempf W, Burg G, Dummer R. HLA-G protein up-regulation in primary cutaneous lymphomas is associated with interleukin-10 expression in large cell T-cell lymphomas and indolent B-cell lymphomas. Blood. 2002;99:609–17.
Urosevic M, Kurrer MO, Kamarashev J, et al. Human leukocyte antigen G up-regulation in lung cancer associates with high-grade, human histology, leukocyte antigen class I loss and interleukin-10 production. Am J Pathol. 2001;159:817–24.
Ye SR, Yang H, Li K, Dong DD, Lin XM, Yie SM. Human leukocyte antigen G expression: as a significant prognostic indicator for patients with colorectal cancer. Mod Pathol. 2007;20:375–83.
Colombo MP, Trinchieri G. Cytokines and cancer. Cytokine Growth Factor Rev. 2002;13:93–4.
Wu S, Gessner R, Taube T, von Stackelberg A, Henze G, Seeger K. Expression of interleukin-10 splicing variants is a positive prognostic feature in relapsed childhood acute lymphoblastic leukemia. J Clin Oncol. 2005;23:3038–42.
Cloppenborg T, Stanulla M, Zimmermann M, Schrappe M, Welte K, Klein C. Immunosurveillance of childhood ALL: polymorphic interferon-gamma Alleles are associated with age at diagnosis and clinical risk groups. Leukaemia. 2005;19:44–8.
Onno M, Le Friec G, Pangault C, Amiot L, Guilloux V, Drenou B, et al. Modulation of HLA-G antigens expression in myelomonocylic cells. Hum Immunol. 2000;61:1086–94.
Farag SS, Caligiuri MA. Human natural killer cell development and biology. Blood Rev. 2006;20:123–37.
Fregni G, Perie A, Avril M, Caignard A. NK cells sense tumors, course of disease and treatments consequences for NK-based therapies. Onco Immunol. 2012;1(1):38–47.
Soderstrom K, Corliss B, Lanier LL, Phillips JH. CD94/NKG2 is the predominant inhibitory receptor involved in recognition of HLA-G by decidual and peripheral blood NK cells. J Immunol. 1997;159:1072–5.
Vardiman JW, Thiele J, Arber DA, Brunning RD, Borowitz MJ, Porwit A, Harris NL, Le Beau MM, Hellström-Lindberg E, Tefferi A, Bloomfield CD. The 2008 revision of the world health organization (WHO) classification of myeloid neoplasms and acute leukemia: rationale and important changes. Blood. 2009;114(5):937–51.
Hoelzer D, Thiel E, Loffler H, et al. Intensified therapy in acute lymphoblastic and acute undifferentiated leukemia in adults. Blood. 1984;64(1):38–47.
Nilsson C, Aboud S, Karlen K, Hejdeman B, Urassa W, Biberfeld G. Optimal blood mononuclear cell isolation procedures for gamma interferon enzyme-linked immunospot testing of healthy Swedish and Tanzanian subjects. Clin Vaccine Immunol. 2008;15(4):585–9.
Yie SM. Hu Z human leukocyte antigen-G (HLA-G) as a marker for diagnosis, prognosis and tumor immune escape in human malignancies. Histol Histopathol. 2011;26(3):409–20.
Nuckel H, Rebmann V, Durig J, Duhrsen U, Grosse-Wilde H. HLA-G expression is associated-with an unfavorable outcome and immunodeficiency in chronic lymphocytic leukemia. Blood. 2005;105(4):1694–8.
Yan WH, Lin A, Chen BG, et al. Unfavorable clinical implications for HLA-G expression in acute myeloid leukemia. J Cell Mol Med. 2008;12(3):889–98.
Rezvany M, Kazemi A, Hajifathali A, Kaviani S, Mellstedt H. Analysis of HLA-G gene expression in B-Lymphocytes from chronic lymphocytic leukemia patients. Iran Biomed J. 2007;11(2):125–9.
Erikci AA, Karagoz B, Ozyurt M, Ozturk A, Kilic S, Bilgi O. HLA-G expression in B chronic lymphocytic leukemia: a new prognostic marker? Hematology. 2009;14(2):101–5.
Agaugue′ S Carosella E, Rouas-Freiss N. Role of HLA-G in tumor escape through expansion of myeloid-derived suppressor cells and cytokine balance in favor of Th2 versus Th1/Th17. Blood. 2011;117(26):7021–31.
Urosevic M, Dummer R. HLA-G and IL-10 expression in human cancer different stories with the same message. Semin Cancer Biol. 2003;13:337–42.
Drabko K, Bojarska-Junak A, Kowalczyk J. Serum concentration of IL-2, IL-4, IL-10 and TNF-α in children with acute lymphoblastic leukemia—possible role of oxidative stress. Eur J Immunol. 2008;33(3):146–9.
Bien E, Balcerska A, Adamkiewicz-Drozynska E, Rapala M, Krawczyk M, Stepinski J. Pre-treatment serum levels of interleukin-10, interleukin-12 and their ratio predict response to therapy and probability of event-free and overall survival in childhood soft tissue sarcomas, Hodgkin’s lymphomas and acute lymphoblastic leukemias. Clin Biochem. 2009;42:1144–57.
Haddad JJ, Fahlman CS. Redox and oxidant mediated regulation of interleukin-10: an anti-inflammatory, anti-oxidant cytokine? Biochem Biopys Res Commun. 2002;297:163–76.
Choi MS, Ray R, Zhaug Z, Mukherjee AB. IFN-gamma stimulates the expression of a novel secretoglobin that regulates chemotactic cell migration and invasion. J Immunol. 2004;172(7):4245–52.
Fujii H, Trudeau JD, Teachey DT, Fish JD, Grupp SA, Schultz KR, Reid GS. In vivo control of acute lymphoblastic leukemia by immunostimulatory CpG oligonucleotides. Blood. 2007;109(5):2008–13.
Varela N, Munoz-Pinedo C, Ruiz-Ruiz C, Robledo G, Pedroso M, López-Rivas A. Interferon-γ sensitizes human myeloid leukemia cells to death receptor-mediated apoptosis by a pleiotropic mechanism. J Biol Chem. 2001;276(21):17779–87.
Kholoussi M, Bayoumi F, El-Nady H. Estimation of serum interferon-gamma level in childhood acute lymphoblastic leukemia patients. J Med Sci. 2008;8(1):68–72.
Tuncer AM, Hiçsönmez G, Gümrük F, Sayli T, Güler E, Cetin M, Okur H. Serum TNF-alpha. gamma-INF. G-CSF and GM-CSF levels in neutropenic children with acute leukemia treated with short-course, high-dose methylprednisolone. Leuk Res. 1996;20(3):265–9.
Ghosh S, Bandyopadhyay S, Mallick A, Pal S, Vlasak R, Bhattacharya DK, Mandal C. Interferon-gamma promotes survival of lymphoblasts overexpressing 9-0-acetylated sialoglycoconjugates in childhood Acute Lymphoblastic Leukemia (ALL). J Cell Biochem. 2005;95(1):206–16.
Sheu J, Shih L. HLA-G and immune evasion in cancer cells. Formos Med Assoc. 2010;109(4):248–57.
Mizuno S, Emi N, Kasai M, Ishitani A, Saito H. Aberrant expression of HLA-G antigen in interferon gamma-stimulated acute myelogenous leukaemia. Br J Haematol. 2000;111:280–2.
Wagner SN, Rebmann V, Willers CP, Grosse-Wilde H, Goos M. Expression analysis of classic and non-classic HLA molecules before interferon alfa-2b treatment of melanoma. Lancet. 2000;356(9225):220–1.
Rouas-Freiss N, Goncalves RM, Menier C, Dausset J, Carosella ED. Direct evidence to support the role of HLA-G in protecting the fetus from maternal uterine natural killer cytolysis. Proc Natl Acad Sci USA. 1997;94:11520–5.
Jarosz M, Hak L, Wieckiewicz J, Balcerska A, Myoeliwska J. NK cells in children with acute lymphoblastic leukemia and non-Hodgkin lymphoma after cessation of intensive chemotherapy. Centr Eur J Immunol. 2009;34(2):94–9.
Alanko S, Salmi TT, Pelliniemi TT. Recovery of natural killer cells after chemotherapy for childhood acute lymphoblastic leukemia and solid tumors. Med Pediatr Oncol. 1995;24:373–8.
Conflict of interest
There is no conflict of interest of any of the authors with any establishment or persons who had a relation to this present work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Alkhouly, N., Shehata, I., Ahmed, M.B. et al. HLA-G expression in acute lymphoblastic leukemia: a significant prognostic tumor biomarker. Med Oncol 30, 460 (2013). https://doi.org/10.1007/s12032-013-0460-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12032-013-0460-8