Abstract
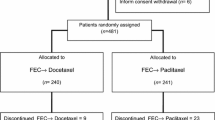
This trial compared 6 cycles of fluorouracil, epirubicin, and cyclophosphamide (FEC) with a sequential regimen of 3 cycles of FEC followed by 3 cycles of docetaxel (FEC-D) as adjuvant treatment for women with node-positive or/and T3 or T4 breast cancer. Between January 2006 and January 2010, 657 patients with operable breast cancer were randomly assigned to either FEC every 21 days for 6 cycles, or 3 cycles of FEC followed by 3 cycles of docetaxel, both given every 21 days. Radiotherapy was mandatory for all patients who had undergone breast conserving surgery. Radiation to the chest wall, supraclavicular area, was recommended following mastectomy. Hormone-receptor–positive patients received tamoxifen for 5 years after chemotherapy. The primary end point was 5-year disease-free survival (DFS). Median follow-up was 61 months. Five-year DFS rates were 74 % with FEC and 78 % with FEC-D (P = 0.013). Multivariate analysis adjusted for prognostic factors showed a 17 % reduction in the relative risk of relapse with FEC-D. Five-year overall survival rates were 85 % with FEC and 89.4 % with FEC-D, demonstrating a 27 % reduction in the relative risk of death (P = 0.014). The incidence of grade 3–4 neutropenia, the need for hematopoietic growth factor, and incidence of nausea/vomiting were higher with FEC. Docetaxel was associated with more febrile neutropenia, stomatitis, edema, and nail disorders. Though rare overall, there were fewer cardiac events after FEC-D, attributable mainly to the lower anthracycline cumulative dose. Sequential adjuvant chemotherapy with FEC followed by docetaxel significantly improves disease-free and overall survival in node-positive or/and T3 or T4 breast cancer patients. Although the magnitude of the benefit observed with FEC-D, differences in the toxicity profiles of FEC and FEC-D may influence the choice of treatment for patients.


Similar content being viewed by others
References
Early Breast Cancer Trialists’ Collaborative Group (EBCTCG). Effects of chemotherapy and hormonal therapy for early breast cancer on recurrence and 15-year survival: an overview of the randomized trail. Lancet. 2005;365:1687–717.
Martin M, Pienkowski T, Mackey J, et al. Adjuvant docetaxel for node-positive breast cancer. N Engl J Med. 2005;352:2302–13.
Bonneterre J, Roche H, Kerbrat P, et al. Epirubicin increases long-term survival in adjuvant chemotherapy of patients with poor-prognosis, node-positive, early breast cancer: 10-year follow-up results of the French adjuvant study group 05 randomized trial. J Clin Oncol. 2005;23:2686–93.
Chevallier B, Fumoleau P, Kerbrat P, et al. Docetaxel is a major cytotoxic drug for the treatment of advanced breast cancer: a phase II trial of the clinical screening cooperative group of the European organization for research and treatment of cancer. J Clin Oncol. 1995;13:314–22.
Valero V, Holmes FA, Walters RS, et al. Phase II trial of docetaxel: a new, highly effective antineoplastic agent in the management of patients with anthracycline-resistant metastatic breast cancer. J Clin Oncol. 1995;13:2886–94.
Ravdin PM, Burris HA 3rd, Cook G, et al. Phase II trial of docetaxel in advanced anthracycline resistant or anthracenedione-resistant breast cancer. J Clin Oncol. 1995;13:2879–85.
Luck HJ, Thomssen C, Du Bois A, et al. Metastatic breast cancer: experience with the combination paclitaxel plus epirubicin. Oncology (WillistonPark). 1998;12(suppl 1):36–9.
Conte PF, Gennari A, Salvadori B, et al. Paclitaxel plus epirubicin in advanced breast cancer. Oncology (Williston Park). 1998;12(suppl 1):40–4.
Misset JL, Dieras V, Gruia G, et al. Dosefinding study of docetaxel and doxorubicin in firstline treatment of patients with metastatic breast cancer. Ann Oncol. 1999;10:553–60.
Nabholtz JM, Mackey JR, Smylie M, et al. Phase II study of docetaxel, doxorubicin, and cyclophosphamide as first-line chemotherapy for metastatic breast cancer. J Clin Oncol. 2001;19:314–21.
Viens P, Roché H, Kerbrat P, et al. Epirubicin–docetaxel combination in first-line chemotherapy for patients with metastatic breast cancer: final results of a dose-finding and efficacy study. Am J Clin Oncol. 2001;24:328–35.
Venturini M, Michelotti A, Papaldo P, et al. Identification of the highest dose of docetaxel associable with active doses of epirubicin: results from a dose-finding study in advanced breast cancer patients. Ann Oncol. 2001;12:1097–106.
Morales S, Lorenzo A, Ramos M, et al. Docetaxel plus epirubicin is a highly active, well tolerated, first-line chemotherapy for metastatic breast cancer: results of a large, multicentre phase II study. Cancer Chemother Pharmacol. 2004;53:75–81.
Biganzoli L, Cufer T, Bruning P, et al. Doxorubicin and paclitaxel versus doxorubicin and cyclophosphamide as first-line chemotherapy in metastatic breast cancer: the European organisation for research and treatment of cancer multicenter phase III trial 10961. J Clin Oncol. 2002;20:3114–21.
Luck H, Thomssen C, Untch M, et al. Multicentric phase III study in first line treatment of advanced metastatic breast cancer: epirubicin/paclitaxel vs epirubicin/cyclophosphamide—A study of the AGO breast cancer group. Proc Am Soc Clin Oncol. 2000;19:73a. (abstr 280).
Langley RE, Carmichael J, Jones AL, et al. Phase III trial of epirubicin plus paclitaxel compared with epirubicin plus cyclophosphamide as first-line chemotherapy for metastatic breast cancer: United Kingdom national cancer res institute trial AB01. J Clin Oncol. 2005;23:8322–30.
Nabholtz JM, Falkson C, Campos D, et al. Docetaxel and doxorubicin compared with doxorubicin and cyclophosphamide as first-line chemotherapy for metastatic breast cancer: results of a randomized, multicenter, phase III trial. J Clin Oncol. 2003;21:968–75.
Mackey JR, Paterson A, Dirix LY, et al. Final results of the phase III randomized trial comparing docetaxel, doxorubicin, and cyclophosphamide to FAC as first line chemotherapy for patients with metastatic breast cancer. Proc Am Soc Clin Oncol. 2002;21:35a. (abstr 137).
Bontenbal M, Creemers GJ, Braun HJ, et al. Phase II to III study comparing doxorubicin and docetaxel with fluorouracil, doxorubicin, and cyclophosphamide as first-line chemotherapy in patients with metastatic breast cancer: results of a Dutch community setting trial for the clinical trial Group of the comprehensive cancer centre. J Clin Oncol. 2005;23:7081–8.
Bonneterre J, Dieras V, Tubiana-Hulin M, et al. Phase II multicentre randomised study of docetaxel plus epirubicin vs 5-fluorouracil plus epirubicin and cyclophosphamide in metastatic breast cancer. Br J Cancer. 2004;91:1466–71.
Henderson IC, Berry DA, Demetri GD, et al. Improved outcomes from adding sequential paclitaxel but not from escalating doxorubicin dose in an adjuvant chemotherapy regimen for patients with node-positive primary breast cancer. J Clin Oncol. 2003;21:976–83.
Mamounas EP, Bryant J, Lembersky B, et al. Paclitaxel after doxorubicin plus cyclophosphamide as adjuvant chemotherapy for node-positive breast cancer: results from NSABP B-28. J Clin Oncol. 2005;23:3686–96.
Roché Henri, Fumoleau Pierre, Spielmann Marc, et al. Sequential adjuvant epirubicin-based and docetaxel chemotherapy for node-positive breast cancer patients: the FNCLCC PACS 01 Trial. J Clin Oncol. 2006;24:5664–71.
Ellis Paul, Barrett-Lee Peter, Johnson Lindsay, et al. Sequential docetaxel as adjuvant chemotherapy for early breast cancer (TACT): an open-label, phase III, randomised controlled trial. Lancet. 2009;373:1681–92.
Fumoleau P, Bonneterre J, Luporsi E, et al. Adjuvant chemotherapy for node-positive breast cancer patients: which is the reference today? J Clin Oncol. 2003;21:1190–2.
National Institutes of Health Consensus Development Panel. National Institutes of Health consensus development conference statement: adjuvant therapy for breast cancer, November 1–3, 2000. J Natl Cancer Inst. 2001;93:979–89.
Martin M, Rodriguez-Lescure A, Ruiz A, et al. Randomized phase 3 trial of fl uorouracil, epirubicin, and cyclophosphamide alone or followed by paclitaxel for early breast cancer. J Natl Cancer Inst. 2008;100:805–14.
Bonneterre J, Roche H, Kerbrat P, et al. Long-term cardiac follow-up in relapse-free patients after six courses of fluorouracil, epirubicin, and cyclophosphamide, with either 50 or 100 mg of epirubicin, as adjuvant therapy for node-positive breast cancer: French adjuvant study group. J Clin Oncol. 2004;22:3070–9.
Fumoleau P, Roche H, Kerbrat P, et al. Longterm cardiac toxicity after adjuvant epirubicin-based chemotherapy in early breast cancer: French adjuvant study group results. Ann Oncol. 2006;17:85–92.
Martin M, Mackey J, Vogel C. Benefi t from adjuvant taxanes and endocrine responsiveness in breast cancer. Breast. 2007;16(suppl 2):S127–31.
Andre F, Broglio K, Roche H, et al. Estrogen receptor expression and efficacy of docetaxel-containing adjuvant chemotherapy in patients with node-positive breast cancer: results from a pooled analysis. J Clin Oncol. 2008;26:2636–43.
Hugh J, Hanson J, Cheang M, et al. Breast cancer subtypes and response to docetaxel in node-positive breast cancer: use of an immunohistochemical defi nition in the BCIRG 001 trial. J Clin Oncol. 2009;27:1168–76.
Conflict of interest
The authors declare that they have no competing interests.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sakr, H., Hamed, R.H., Anter, A.H. et al. Sequential docetaxel as adjuvant chemotherapy for node-positive or/and T3 or T4 breast cancer: clinical outcome (Mansoura University). Med Oncol 30, 457 (2013). https://doi.org/10.1007/s12032-013-0457-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12032-013-0457-3