Abstract
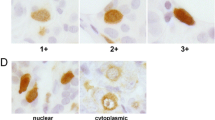
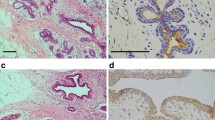
Plakophilins (PKP) are desmosomal plague proteins, which belong to the p120ctn subfamily of armadillo repeat containing proteins. We aimed to analyze the role of plakophilins in breast cancer and its clinical progress. We have performed immunohistochemical study of the PKP1,2,3 in breast carcinoma. The study included 108 patients with breast cancer and 26 age- and sex-matched healthy controls. We investigated the associations between staining intensity and some clinicopathologic features like tumor size, axillary node status, stage, lymphovascular invasion, perineural invasion, grade, hormone receptor status, and c-erb B2. The mean age of patients was 46 years (22–78). In breast cancer, compared with normal tissue, PKP1 and PKP2 expressions were indifferent (P > 0.05), but PKP3 expression was significantly increased in breast cancer (P = 0.0014). Although PKP1 and PKP2 expression levels were not correlated with clinicopathological parameters, increased PKP3 expression was positively correlated with node positivity and grade (P = 0.000, P = 0.000). Conclusion: Overexpressed PKP3 is likely to be an essential contributor to a growth-promoting pathway and to aggressive features of breast cancer.

Similar content being viewed by others
References
Schwarz J, Ayim A, Schmidt A, Jäger S, Koch S, Baumann R, et al. Differential expression of desmosomal plakophilins in various types of carcinomas: correlation with cell type and differentiation. Hum Pathol. 2006;37:613–22.
Schmidt A. Jager S.Plakophilins hard-work in the desmosome recreation in the nucleus? Eur J Cell Biol. 2005;84:189–204.
Schmidt A, Langbein L, Prätzel S, Rode M, Rackwitz HR, Franke WW. Plakophilin 3–a novel cell-type-specific desmosomal plaque protein. Differentiation. 1999;5:291–306.
Heid HW, Schmidh A, Zimbelmann R, et al. Cell type-spesific desmosomal plaque proteins of the plakoglobin family:plakophilin 1. Differentiation. 1994;58:113–31.
Mertens C, Kuhn C, Franke WW. Plakophilins 2a and 2b: constitutive proteins of dual location in the karyoplasm and the desmosomal plaque. J Cell Biol. 1996;4:1009–25.
Villaret DB, Wang T, Dillon D, Xu J, Sivam D, Cheever MA, et al. Identification of genes overexpressed in head and neck squamous cell carcinoma using a combination of complementary DNA subtraction and microarray analysis. Laryngoscope. 2000;3:374–81.
Mertens C, Kuhn C, Moll R, Schwetlick I, Franke WW. Desmosomal plakophilin 2 as a differentiation marker in normal and malignant tissues. Differentiation. 1999;5:277–90.
Bass-Zubeck A, Godsel LM, Delmar M, Green KJ. Plakophilins: multifunctional scaffolds for adhesion and signaling. Curr Opin Cell Biol. 2009;21:708–16.
Bonne S, van Hengel J, Nollet F, Kools P, van Roy F. Plakophilin-3 a novel armadillo-like protein present in nuclei and desmosomes of epithelial cells. J Cell Sci. 1999;112:2265–76.
Bonne S, Gilbert B, Hatzfeld M, Chen X, Green KJ, Van Roy F. Defining desmosomal plakophilin-3 interactions. J Cell Biol. 2003;161:403–16.
Furukawa C, Daigo Y, Ishikawa N, Kato T, Ito T, Tsuchiya E, et al. Plakophilin 3 oncogene as prognostic marker and therapeutic target for lung caner. Cancer Res. 2005;65:7102–10.
Hatzfeld M. Plakophilins: multifunctional protein sor just regulators of desmosomal adhesion? Biochim Biophys Acta. 2007;1773:69–77.
South AP. Plakophilin 1: an important stabilizer of desmosomes. Clin Exp Derm. 2004;29:161–7.
Papagerakis S, Shabana AH, Depondt J, Gehanno P, Forest N. Immunohistochemical localization of plakophilins (PKP1, PKP2, PKP3, and p0071) in primary oropharyngeal tumors: correlation with clinical parameters. Hum Pathol. 2003;34:565–72.
Cheung IY, Feng Y, Danis K. Novel markers of subclinical disease for Ewing family tumors from gene expression profiling. Clin Cancer Res. 2007;13:6978–83.
Mertens C, Kuhn C, Moll R, Schwetlick I, Franke WW. Desmosomal plakophilin 2 as a differentiation marker in normal and malignant tissues. Differentiation. 1999;5:277–90.
Kundu ST, Gosavi P, Khapare N, Patel R, Hosing AS, Maru GB, Ingle A, DeCaprio JA, Dalal SN. Plakophilin3 downregulation leads to a decrease in cell adhesion and promotes metastasis. Int J Cancer. 2008;123:2303–14.
Aigner K, Descovich L, Mikula M, Sultan A, Dampier B, Bonné S, van Roy F, Mikulits W, Schreiber M, Brabletz T, Sommergruber W, Schweifer N, Wernitznig A, Beug H, Foisner R, Eger A. The transcription factor ZEB1 (deltaEF1) represses Plakophilin 3 during human cancer progression. FEBS Lett. 2007;17:1617–24.
Goodfellow IG, Roberts LO. Eukaryotic initiation factor 4E. Int J Biochem Cell Biol. 2008;40:2675–80.
Hinton TM, Coldwell MJ, Carpenter GA, Morley SJ, Pain VM. Functional analysis of individual binding activities of the scaffold protein eIF4G. J Biol Chem. 2007;282:1695–708.
De Benedetti A, Graff JR. eIF-4E expression and its role in malignancies and metastases. Oncogene. 2004;23:3189–99.
Breuninger S, Reidenbach S, Sauer CG, Ströbel P, Pfitzenmaier J, Trojan L, Hofmann I. Desmosomal plakophilins in the prostate and prostatic adenocarcinomas: implications for diagnosis and tumor progression. Am J Pathol. 2010;176(5):2509–19. Epub 2010 Mar 26.
Hatzfeld M, Wolf A. A role of plakophilins in the regulation of translation. Cell Cycle. 2010;9:2973–8.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Demirag, G.G., Sullu, Y. & Yucel, I. Expression of Plakophilins (PKP1, PKP2, and PKP3) in breast cancers. Med Oncol 29, 1518–1522 (2012). https://doi.org/10.1007/s12032-011-0071-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12032-011-0071-1