Abstract
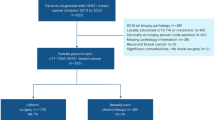
It has been shown that breast cancer patients with N3a (10 positive lymph nodes) had a poor prognosis. We planned to investigate the clinical outcome BC patients who presented with N3a disease and had no evidence of systemic metastasis at the time of diagnosis. We made a retrospective chart review of breast cancer patients who had ≥10 positive lymph nodes and received adjuvant systemic therapy in Marmara University Hospital between 1998 and 2008. We recorded clinical, pathologic and treatment characteristics of the patients and analyzed the survival outcome. We identified 73 patients with N3a disease who were treated in Marmara University Hospital between 1998 and 2008. The median age was 52. Most (75%) of the patients had invasive ductal histology, 75% had T2/T3 tumors, 36% had grade 3 tumors. The median number of metastatic lymph nodes was 15. Estrogen and progesterone receptors were both positive in 61% and both negative in 16+ tumors. Her-2/neu status was assessed in 68% of the tumors; 18% of patients had 3+ and 50% had negative scores. Six patients had triple negative tumors. All patients except one received adjuvant chemotherapy and radiotherapy. Seventy-four percent of patients received anthracycline/taxane-based chemotherapy. Fifty-nine patients received adjuvant endocrine therapy, 42% them received aromatase inhibitors. Five of the 13 Her-2 positive patients received adjuvant trastuzumab. With a median follow-up of 47 months, 5-year disease and overall survival rates were 66 and 81%, respectively. Twenty-four patients had relapsed and 14 patients died. Her-2 status and the number of lymph nodes (<20 vs. ≥20) had significant impact on disease-free survival in the univariate analysis (P = 0.03 and 0.05, respectively) and Her-2 retained its significant impact on disease-free survival in the multivariate analysis (P = 0.05). The prognosis of BC patients with N3a disease has changed favorably in the past decade with the current standards of care.


Similar content being viewed by others
References
van ‘t Veer LJ, et al. Gene expression profiling predicts clinical outcome of breast cancer. Nature. 2002;415:530–6.
van de Vijver MJ, et al. A gene-expression signature as a predictor of survival in breast cancer. N Engl J Med. 2002;347(25):1999–2009.
Carter CL, Allen C, DE Henson. Relation of tumor size, lymph node status, and survival in 24,740 breast cancer cases. Cancer. 1989;63(1):181–7.
Nemoto T, et al. Management and survival of female breast cancer: results of a national survey by the American College of Surgeons. Cancer. 1980;45(12):2917–24.
Singletary SE, et al. Revision of the American Joint Committee on Cancer staging system for breast cancer. J Clin Oncol. 2002;20(17):3628–36.
Early Breast Cancer Trialists’ CollaborativeGroup (EBCTCG). Effects of chemotherapy and hormonal therapy for early breast cancer on recurrence and 15-year survival: an overview of the randomised trials. Lancet. 2005;365:1687–717.
Bria E, et al. Benefit of taxanes as adjuvant chemotherapy for early breast cancer. Cancer. 2006;106:2337–44.
De Laurentiis M, et al. Taxane-based combinations as adjuvant chemotherapy of early breast cancer: a meta-analysis of randomized trials. J Clin Oncol. 2008;26:44–53.
Ferguson T, et al. Taxanes for adjuvant treatment of early breast cancer. Cochrane Database Syst Rev. 2007; Issue 4.
Walker MS, et al. The natural history of breast cancer with more than 10 positive nodes. Am J Surg. 1995;169:575–9.
Buzdar AU, et al. Clinical course of patients with breast cancer with ten or more positive nodes who were treated with doxorubicin-containing adjuvant therapy. Cancer. 1992;69(2):448–52.
Schmoor C, Sauerbrei W, Bastert G, Bojar H, Schumacher M, German Breast Cancer Study Group. Long-term prognosis of breast cancer patients with 10 or more positive lymph nodes treated with CMF. Eur J Cancer. 2001;37(9):1123–31.
Peters WP, et al. Prospective, randomized comparison of high-dose chemotherapy with stem-cell support versus intermediate-dose chemotherapy after surgery and adjuvant chemotherapy in women with high-risk primary breast cancer: a report of CALGB 9082, SWOG 9114, and NCIC MA–13. J Clin Oncol. 2005;23:2191.
Tallman MS, et al. Conventional adjuvant chemotherapy with or without high-dose chemotherapy and autologous stem-cell transplantation in high-risk breast cancer. N Engl J Med. 2003;349:17.
Zander AR, et al. High-dose chemotherapy with autologous hematopoietic stem-cell support compared with standard-dose chemotherapy in breast cancer patients with 10 or more positive lymph nodes: first results of a randomized trial. J Clin Oncol. 2004;22:2273.
Fumoleau P, et al. Intensification of adjuvant chemotherapy: 5-year results of a randomized trial comparing conventional doxorubicin and cyclophosphamide with high-dose mitoxantrone and cyclophosphamide with filgrastim in operable breast cancer with 10 or more involved axillary nodes. J Clin Oncol. 2001;19(3):612–20.
Eiermann W, German Adjuvant Breast Cancer Group, et al. Dose-intensified epirubicin versus standard-dose epirubicin/cyclophosphamide followed by CMF in breast cancer patients with 10 or more positive lymph nodes: results of a randomised trial (GABG-IV E-93)–the German Adjuvant Breast Cancer Group. Eur J Cancer. 2010;46(1):84–94.
Tsuchiya A, Kanno M, Abe R. The impact of lymph node metastases on the survival of breast cancer patients with ten or more positive lymph nodes. Surg Today. 1997;27(10):902–6.
Faneyte IF, et al. Predicting early failure after adjuvant chemotherapy in high-risk breast cancer patients with extensive lymph node involvement. Clin Cancer Res. 2004;10(13):4457–63.
Diab SG, et al. Radiation therapy, survival in breast cancer patients with 10 or more positive axillary lymph nodes treated with mastectomy. J Clin Oncol. 1998;16(5):1655–60.
Gianni AM, et al. Efficacy, toxicity, applicability of high-dose sequential chemotherapy as adjuvant treatment in operable breast cancer with 10 or more involved axillary nodes: five-year results. J Clin Oncol. 1997;15(6):2312–21.
Peters WP, et al. High-dose chemotherapy, autologous bone marrow support as consolidation after standard-dose adjuvant therapy for high-risk primary breast cancer. J Clin Oncol. 1993;11(6):1132–43.
Crump M, Goss PE, Prince M, Girouard C. Outcome of extensive evaluation before adjuvant therapy in women with breast cancer, 10 or more positive axillary lymph nodes. J Clin Oncol. 1996;14(1):66–9.
Peto R, et al., Early Breast Cancer Trialists’ Collaborative Group. The worldwide overview: new results for systemic adjuvant therapies. San Antonio breast cancer symposium. 2007. Abstr P–1.
Citron ML, Berry DA, Cirrincione C, et al. Randomized trial of dose-dense versus conventionally scheduled and sequential versus concurrent combination chemotherapy as postoperative adjuvant treatment of node-positive primary breast cancer: first report of Intergroup Trial C9741/Cancer and Leukemia Group B Trial 9741. J Clin Oncol. 2003;21(8):1431–9.
Hudis C. CAGB 9344/CAGB C9741: what we have learnt? Eur J Cancer. 2006;Supp 4(5):10–2.
Ignatiadis M, Desmedt C, Sotiriou C, de Azambuja E, Piccart M. HER-2 as a target for breast cancer therapy. Clin Cancer Res. 2009;15(6):1848–52.
Jabro G, et al. The importance of local-regional radiotherapy with conventional or high-dose chemotherapy in the management of breast cancer patients with > or =10 positive axillary nodes. Int J Radiat Oncol Biol Phys. 1999;44(2):273–80.
Dowsett M, et al. Meta-analysis of breast cancer outcomes in adjuvant trials of aromatase inhibitors versus tamoxifen. J Clin Oncol. 2010;28(3):509–18.
Sørlie T, et al. Gene expression patterns of breast carcinomas distinguish tumor subclasses with clinical implications. Proc Natl Acad Sci USA. 2001;98(19):10869–74.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Basaran, G., Devrim, C., Caglar, H.B. et al. Clinical outcome of breast cancer patients with N3a (≥10 positive lymph nodes) disease: has it changed over years?. Med Oncol 28, 726–732 (2011). https://doi.org/10.1007/s12032-010-9516-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12032-010-9516-1