Abstract
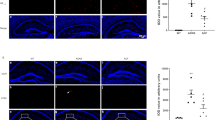
Alzheimer’s disease (AD) is a severe neurological illness that causes memory loss and is a global problem. The calcium hypothesis recently steadily evolved in AD. The prospective targets for calcium homeostasis therapy, however, are limited, and gene expression-level research connected to calcium homeostasis in AD remains hazy. In this study, we analyzed the microarray dataset (GSE132903) taken from the Gene Expression Omnibus (GEO) database to investigate calcium homeostasis-related genes for AD. Using immunoblot analysis, we examined the association of ITPKB with inflammation in AD. Additionally, the immunofluorescence technique was employed to assess the impact of pharmacological inhibition of ITPKB on the amyloid-β (Aβ) plaque deposition in APP/PS1 mice. This article’s further exploration of calcium homeostasis-related genes has propelled the validation of the calcium homeostasis theory in AD.









Similar content being viewed by others
Data Availability
Data is provided within the manuscript or supplementary information files.
References
Agostini M, Fasolato C (2016) When, where and how? Focus on neuronal calcium dysfunctions in Alzheimer’s Disease. Cell Calcium 60:289–298. https://doi.org/10.1016/j.ceca.2016.06.008
Bajrai LH, Sohrab SS, Alandijany TA et al (2021) Gene expression profiling of early acute febrile stage of dengue infection and its comparative analysis with Streptococcus pneumoniae infection. Front Cell Infect Microbiol 11. https://doi.org/10.3389/fcimb.2021.707905
Baleriola J, Walker CA, Jean YY et al (2014) Axonally synthesized ATF4 transmits a neurodegenerative signal across brain regions. Cell 158:1159–1172. https://doi.org/10.1016/j.cell.2014.07.001
Bezprozvanny I, Mattson MP (2008) Neuronal calcium mishandling and the pathogenesis of Alzheimer’s disease. Trends Neurosci 31:454–463. https://doi.org/10.1016/j.tins.2008.06.005
Brancaccio D, Tetta C, Gallieni M et al (2002) Inflammation, CRP, calcium overload and a high calcium-phosphate product: a “liaison dangereuse.” Nephrol Dial Transplant 17:201–203. https://doi.org/10.1093/ndt/17.2.201
Briggs CA, Chakroborty S, Stutzmann GE (2017) Emerging pathways driving early synaptic pathology in Alzheimer’s disease. Biochem Biophys Res Commun 483:988–997. https://doi.org/10.1016/j.bbrc.2016.09.088
Calsolaro V, Edison P (2016) Neuroinflammation in Alzheimer’s disease: current evidence and future directions. Alzheimers Dement 12:719–732. https://doi.org/10.1016/j.jalz.2016.02.010
Chang KA, Kim SH, Sakaki Y et al (1999) Inhibition of the NGF and IL-1beta-induced expression of Alzheimer’s amyloid precursor protein by antisense oligonucleotides. J Mol Neurosci 12:69–74. https://doi.org/10.1385/jmn:12:1:69
Chen K, Yang LN, Lai C et al (2020) Role of Grina/Nmdara1 in the central nervous system diseases. Curr Neuropharmacol 18:861–867. https://doi.org/10.2174/1570159X18666200303104235
Copenhaver PF, Anekonda TS, Musashe D et al (2011) A translational continuum of model systems for evaluating treatment strategies in Alzheimer’s disease: isradipine as a candidate drug. Dis Model Mech 4:634–648. https://doi.org/10.1242/dmm.006841
Cramer E, Lenz U (2010) Association of progressively type-II censored order statistics. Journal of Statistical Planning and Inference 140:576–583. https://doi.org/10.1016/j.jspi.2009.07.020
Crossley CA, Rajani V, Yuan Q (2023) Modulation of L-type calcium channels in Alzheimer’s disease: a potential therapeutic target. Comput Struct Biotechnol J 21:11–20. https://doi.org/10.1016/j.csbj.2022.11.049
Fu WY, Ip NY (2023) The role of genetic risk factors of Alzheimer’s disease in synaptic dysfunction. Semin Cell Dev Biol 139:3–12. https://doi.org/10.1016/j.semcdb.2022.07.011
Gentleman RC, Carey VJ, Bates DM et al (2004) Bioconductor: open software development for computational biology and bioinformatics. Genome Biol 5:R80. https://doi.org/10.1186/gb-2004-5-10-r80
Hickman SE, Allison EK, El Khoury J (2008) Microglial dysfunction and defective beta-amyloid clearance pathways in aging Alzheimer’s disease mice. J Neurosci 28:8354–8360. https://doi.org/10.1523/JNEUROSCI.0616-08.2008
Kanehisa M (2019) Toward understanding the origin and evolution of cellular organisms. Protein Sci 28:1947–1951. https://doi.org/10.1002/pro.3715
Kanehisa M, Goto S (2000) KEGG: kyoto encyclopedia of genes and genomes. Nucleic Acids Res 28:27–30. https://doi.org/10.1093/nar/28.1.27
Kanehisa M, Furumichi M, Sato Y et al (2023) KEGG for taxonomy-based analysis of pathways and genomes. Nucleic Acids Res 51:D587–D592. https://doi.org/10.1093/nar/gkac963
Khan S, Barve KH, Kumar MS (2020) Recent advancements in pathogenesis, diagnostics and treatment of Alzheimer’s disease. Curr Neuropharmacol 18:1106–1125. https://doi.org/10.2174/1570159x18666200528142429
Korkotian E, Oni-Biton E, Segal M (2017) The role of the store-operated calcium entry channel Orai1 in cultured rat hippocampal synapse formation and plasticity. J Physiol 595:125–140. https://doi.org/10.1113/JP272645
Lefever S, Anckaert J, Volders PJ et al (2017) decodeRNA- predicting non-coding RNA functions using guilt-by-association. Database (Oxford) 2017. https://doi.org/10.1093/database/bax042
Miller AT, Dahlberg C, Sandberg ML et al (2015) Inhibition of the inositol kinase Itpkb augments calcium signaling in lymphocytes and reveals a novel strategy to treat autoimmune disease. PLoS ONE 10:e01310710 https://doi.org/10.1371/journal.pone.0131071
Moradi Majd R, Mayeli M, Rahmani F (2020) Pathogenesis and promising therapeutics of Alzheimer disease through eIF2alpha pathway and correspondent kinases. Metab Brain Dis 35:1241–1250. https://doi.org/10.1007/s11011-020-00600-8
Pan XD, Zhu YG, Lin N et al (2011) Microglial phagocytosis induced by fibrillar beta-amyloid is attenuated by oligomeric beta-amyloid: implications for Alzheimer’s disease. Mol Neurodegener 6:45. https://doi.org/10.1186/1750-1326-6-45
Paris D, Bachmeier C, Patel N et al (2011) Selective antihypertensive dihydropyridines lower Abeta accumulation by targeting both the production and the clearance of Abeta across the blood-brain barrier. Mol Med 17:149–162. https://doi.org/10.2119/molmed.2010.00180
Pchitskaya E, Popugaeva E, Bezprozvanny I (2018) Calcium signaling and molecular mechanisms underlying neurodegenerative diseases. Cell Calcium 70:87–94. https://doi.org/10.1016/j.ceca.2017.06.008
Rinn JL, Chang HY (2012) Genome regulation by long noncoding RNAs. Annu Rev Biochem 81:145–166. https://doi.org/10.1146/annurev-biochem-051410-092902
Ryan KC, Ashkavand Z, Norman KR (2020) The role of mitochondrial calcium homeostasis in Alzheimer’s and related diseases. Int J Mol Sci. https://doi.org/10.3390/ijms21239153
Ryu JC, Zimmer ER, Rosa-Neto P et al (2019) Consequences of metabolic disruption in Alzheimer’s disease pathology. Neurotherapeutics 16:600–610. https://doi.org/10.1007/s13311-019-00755-y
Schwartz L, Peres S, Jolicoeur M et al (2020) Cancer and Alzheimer’s disease: intracellular pH scales the metabolic disorders. Biogerontology 21:683–694. https://doi.org/10.1007/s10522-020-09888-6
Stygelbout V, Leroy K, Pouillon V et al (2014) Inositol trisphosphate 3-kinase B is increased in human Alzheimer brain and exacerbates mouse Alzheimer pathology. Brain 137:537–552. https://doi.org/10.1093/brain/awt344
Subramanian A, Tamayo P, Mootha VK et al (2005) Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc Natl Acad Sci USA 102:15545–15550. https://doi.org/10.1073/pnas.0506580102
Sun S, Zhang H, Liu J et al (2014) Reduced synaptic STIM2 expression and impaired store-operated calcium entry cause destabilization of mature spines in mutant presenilin mice. Neuron 82:79–93. https://doi.org/10.1016/j.neuron.2014.02.019
Tropea MR, Gulisano W, Vacanti V et al (2022) Nitric oxide/cGMP/CREB pathway and amyloid-beta crosstalk: from physiology to Alzheimer’s disease. Free Radic Biol Med 193:657–668. https://doi.org/10.1016/j.freeradbiomed.2022.11.022
Unger MS, Li E, Scharnagl L et al (2020) CD8(+) T-cells infiltrate Alzheimer’s disease brains and regulate neuronal- and synapse-related gene expression in APP-PS1 transgenic mice. Brain Behav Immun 89:67–86. https://doi.org/10.1016/j.bbi.2020.05.070
Yu G, Wang LG, Han Y et al (2012) clusterProfiler: an R package for comparing biological themes among gene clusters. OMICS 16:284–287. https://doi.org/10.1089/omi.2011.0118
Zhang G, Wang Z, Hu H et al (2021) Microglia in Alzheimer’s disease: a target for therapeutic intervention. Front Cell Neurosci 15. https://doi.org/10.3389/fncel.2021.749587
Zhou B, Tian R (2018) Mitochondrial dysfunction in pathophysiology of heart failure. J Clin Investig 128:3716–3726. https://doi.org/10.1172/jci120849
Funding
The key project of the Precision Medicine Joint Fund of the Natural Science Foundation of Hebei Province, Grant No. H2021206021, The 2022 Government-funded Provincial Medical Outstanding Talent Project.
Author information
Authors and Affiliations
Contributions
YH, FX, XR, and ML: methodology, data collection, and analysis, and manuscript preparation; ZZ and ZZ: study design and data interpretation. QW: manuscript revision. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics Approval
The study was approved by the Ethics Committee in the Third Hospital of Hebei Medical University (B2020-260R, 23 May 2022).
Consent to Participate
Informed consent was obtained from all individual participants included in the study.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hu, Y., Zhao, Z., Xu, F. et al. Transcriptome and Animal Model Integration Reveals Inhibition of Calcium Homeostasis-Associated Gene ITPKB Alleviates Amyloid Plaque Deposition. J Mol Neurosci 74, 42 (2024). https://doi.org/10.1007/s12031-024-02221-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12031-024-02221-7