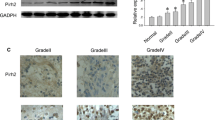
Abstract
Proline and serine–rich coiled-coil 1 (PSRC1) has been reported to function as an oncogene in several cancers by regulating mitosis, while there are few reports on the role of PSRC1 in lower-grade glioma (LGG). Thus, this study collected 22 samples and 1126 samples from our institution and several databases, respectively, to explore the function of PSRC1 in LGG. First, the analysis of clinical characteristics showed that PSRC1 was always highly expressed in more malignant clinical characteristics of LGG, such as higher WHO grade, recurrence type, and IDH wild type. Second, the prognosis analysis revealed that the high expression of PSRC1 was an independent risk factor contributing to the shorter overall survival of LGG patients. Third, the analysis of DNA methylation showed that the expression of PSRC1 was associated with its 8 DNA methylation sites, overall negatively regulated by its DNA methylation level in LGG. Fourth, the analysis of immune correlation revealed that the expression of PSRC1 was positively correlated with the infiltration of 6 immune cells and the expression of 4 well-known immune checkpoints in LGG, respectively. Finally, co-expression analysis and KEGG analysis showed the 10 genes most related to PSRC1 and the signaling pathways involved by PSRC1 in LGG, respectively, such as MAPK signaling pathway and focal adhesion. In conclusion, this study identified the pathogenic role of PSRC1 in the pathological progression of LGG, expanding the molecular understanding of PSRC1, and provided a biomarker and potential immunotherapeutic target for the treatment of LGG.





Similar content being viewed by others
Data Availability
The data set analyzed during the current study is available in the TCGA database (https://cancergenome.nih.gov/) and the CGGA database (http://www.cgga.org.cn/) and the GEO database (https://www.ncbi.nlm.nih.gov/geo/). Besides, the datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Boussiotis V, Charest A (2018) Immunotherapies for malignant glioma. Oncogene 37:1121. https://doi.org/10.1038/s41388-017-0024-z
Chen T, Yang S, Xu J, Lu W, Xie X (2020) Transcriptome sequencing profiles of cervical cancer tissues and SiHa cells. Funct Integr Genomics 20:211. https://doi.org/10.1007/s10142-019-00706-y
Duffau H, Taillandier L (2015) New concepts in the management of diffuse low-grade glioma: proposal of a multistage and individualized therapeutic approach. Neuro Oncol 17:332. https://doi.org/10.1093/neuonc/nou153
Fife B, Pauken K (2011) The role of the PD-1 pathway in autoimmunity and peripheral tolerance. Ann N Y Acad Sci 1217:45. https://doi.org/10.1111/j.1749-6632.2010.05919.x
Gorman J, Colgan J (2014) Regulation of T cell responses by the receptor molecule Tim-3. Immunol Res 59:56. https://doi.org/10.1007/s12026-014-8524-1
Graziano S, Gonzalo S (2017) Mechanisms of oncogene-induced genomic instability. Biophys Chem 225:49. https://doi.org/10.1016/j.bpc.2016.11.008
Guo K, Hu L, Xi D, Zhao J, Liu J, Luo T, Ma Y, Lai W, Guo Z (2018) PSRC1 overexpression attenuates atherosclerosis progression in apoE mice by modulating cholesterol transportation and inflammation. J Mol Cell Cardiol 116:69. https://doi.org/10.1016/j.yjmcc.2018.01.013
Gupta M, Sarojamma V, Reddy M, Shaik J, Vadde R (2019) Computational biology: toward early detection of pancreatic cancer. Crit Rev Oncog 24:191. https://doi.org/10.1615/CritRevOncog.2019031335
Han M, Xu W (2017) EMP3 is induced by TWIST1/2 and regulates epithelial-to-mesenchymal transition of gastric cancer cells. Tumour Biol 39:1010428317718404. https://doi.org/10.1177/1010428317718404
Hanahan D, Weinberg R (2011) Hallmarks of cancer: the next generation. Cell 144:646. https://doi.org/10.1016/j.cell.2011.02.013
Hsieh P, Chang J, Sun W, Hsieh S, Wang M, Wang F (2007) p53 downstream target DDA3 is a novel microtubule-associated protein that interacts with end-binding protein EB3 and activates beta-catenin pathway. Oncogene 26:4928. https://doi.org/10.1038/sj.onc.1210304
Hsieh W, Hsieh S, Chen C, Wang F (2008) Human DDA3 is an oncoprotein down-regulated by p53 and DNA damage. Biochem Biophys Res Commun 369:567. https://doi.org/10.1016/j.bbrc.2008.02.047
Jang C, Wong J, Coppinger J, Seki A, Yates J, Fang G (2008) DDA3 recruits microtubule depolymerase Kif2a to spindle poles and controls spindle dynamics and mitotic chromosome movement. J Cell Biol 181:255. https://doi.org/10.1083/jcb.200711032
Jones P, Ohtani H, Chakravarthy A, De Carvalho D (2019) Epigenetic therapy in immune-oncology. Nat Rev Cancer 19:151. https://doi.org/10.1038/s41568-019-0109-9
Juergens R, Wrangle J, Vendetti F, Murphy S, Zhao M, Coleman B, Sebree R, Rodgers K, Hooker C, Franco N, Lee B, Tsai S, Delgado I, Rudek M, Belinsky S, Herman J, Baylin S, Brock M, Rudin C (2011) Combination epigenetic therapy has efficacy in patients with refractory advanced non-small cell lung cancer. Cancer Discov 1:598. https://doi.org/10.1158/2159-8290.Cd-11-0214
Kim S, Jang C (2016) ANKRD53 interacts with DDA3 and regulates chromosome integrity during mitosis. Biochem Biophys Res Commun 470:484. https://doi.org/10.1016/j.bbrc.2016.01.144
Kulis M, Esteller M (2010) DNA methylation and cancer. Adv Genet 70:27. https://doi.org/10.1016/b978-0-12-380866-0.60002-2
Louis D, Perry A, Reifenberger G, von Deimling A, Figarella-Branger D, Cavenee W, Ohgaki H, Wiestler O, Kleihues P, Ellison D (2016) The 2016 World Health Organization classification of tumors of the central nervous system: a summary. Acta Neuropathol 131:803. https://doi.org/10.1007/s00401-016-1545-1
Ma Q, Zhang Y, Liang H, Zhang F, Liu F, Chen S, Hu Y, Jiang L, Hao Y, Li M, Liu Y (2018a) EMP3, which is regulated by miR-663a, suppresses gallbladder cancer progression via interference with the MAPK/ERK pathway. Cancer Lett 430:97. https://doi.org/10.1016/j.canlet.2018.05.022
Ma X, Shang F, Ni W, Zhu J, Luo B, Zhang Y (2018b) MicroRNA-338-5p plays a tumor suppressor role in glioma through inhibition of the MAPK-signaling pathway by binding to FOXD1. J Cancer Res Clin Oncol 144:2351. https://doi.org/10.1007/s00432-018-2745-y
Mazor G, Levin L, Picard D, Ahmadov U, Carén H, Borkhardt A, Reifenberger G, Leprivier G, Remke M, Rotblat B (2019) The lncRNA TP73-AS1 is linked to aggressiveness in glioblastoma and promotes temozolomide resistance in glioblastoma cancer stem cells. Cell Death Dis 10:246. https://doi.org/10.1038/s41419-019-1477-5
Moore L, Le T, Fan G (2013) DNA methylation and its basic function. Neuropsychopharmacology 38:23. https://doi.org/10.1038/npp.2012.112
Oishi Y, Watanabe Y, Yoshida Y, Sato Y, Hiraishi T, Oikawa R, Maehata T, Suzuki H, Toyota M, Niwa H, Suzuki M, Itoh F (2012) Hypermethylation of Sox17 gene is useful as a molecular diagnostic application in early gastric cancer. Tumour Biol 33:383. https://doi.org/10.1007/s13277-011-0278-y
Paluch E, Aspalter I, Sixt M (2016) Focal adhesion-independent cell migration. Annu Rev Cell Dev Biol 32:469. https://doi.org/10.1146/annurev-cellbio-111315-125341
Qemo I, Porter L (2019) Cell cycle dynamics in glioma cancer stem cells. Methods Mol Biol 1869:117. https://doi.org/10.1007/978-1-4939-8805-1_11
Raffaello A, De Stefani D, Sabbadin D, Teardo E, Merli G, Picard A, Checchetto V, Moro S, Szabò I, Rizzuto R (2013) The mitochondrial calcium uniporter is a multimer that can include a dominant-negative pore-forming subunit. EMBO J 32:2362. https://doi.org/10.1038/emboj.2013.157
Sakaguchi S, Mikami N, Wing J, Tanaka A, Ichiyama K, Ohkura N (2020) Regulatory T cells and human disease. Annu Rev Immunol 38:541. https://doi.org/10.1146/annurev-immunol-042718-041717
Stresemann C, Lyko F (2008) Modes of action of the DNA methyltransferase inhibitors azacytidine and decitabine. Int J Cancer 123:8. https://doi.org/10.1002/ijc.23607
Sun Y, Liu W, Liu T, Feng X, Yang N, Zhou H (2015) Signaling pathway of MAPK/ERK in cell proliferation, differentiation, migration, senescence and apoptosis. J Recept Signal Transduct Res 35:600. https://doi.org/10.3109/10799893.2015.1030412
Tang Q, Cheng J, Cao X, Surowy H, Burwinkel B (2016) Clin Epigenetics Clinical Epigenetics 8:115. https://doi.org/10.1186/s13148-016-0282-6
Van Tongelen A, Loriot A, De Smet C (2017) Oncogenic roles of DNA hypomethylation through the activation of cancer-germline genes. Cancer Lett 396:130. https://doi.org/10.1016/j.canlet.2017.03.029
Wang B, Yang B, Yang H, Wang J, Hu S, Gao Y, Bu X (2018) MicroRNA-499a decelerates glioma cell proliferation while accelerating apoptosis through the suppression of Notch1 and the MAPK signaling pathway. Brain Res Bull 142:96. https://doi.org/10.1016/j.brainresbull.2018.06.005
Wang X, Zhou R, Xiong Y, Zhou L, Yan X, Wang M, Li F, Xie C, Zhang Y, Huang Z, Ding C, Shi K, Li W, Liu Y, Cao Z, Zhang Z, Zhou S, Chen C, Zhang Y, Chen L, Wang Y (2021) Sequential fate-switches in stem-like cells drive the tumorigenic trajectory from human neural stem cells to malignant glioma. Cell Res 31:684. https://doi.org/10.1038/s41422-020-00451-z
Youssef G, Miller J (2020) Lower grade gliomas. Curr Neurol Neurosci Rep 20:21. https://doi.org/10.1007/s11910-020-01040-8
Yu Q, Wang X, Luo J, Wang S, Fang X, Yu J, Ling Z (2012) CDH1 methylation in preoperative peritoneal washes is an independent prognostic factor for gastric cancer. J Surg Oncol 106:765. https://doi.org/10.1002/jso.23116
Yue H, Xu Q, Xie S (2018) High EMP3 expression might independently predict poor overall survival in glioblastoma and its expression is related to DNA methylation. Medicine 97:e9538. https://doi.org/10.1097/md.0000000000009538
Zheng Z, Luan X, Zha J, Li Z, Wu L, Yan Y, Wang H, Hou D, Huang L, Huang F, Zheng H, Ge L, Guan H (2017) TNF-α inhibits the migration of oral squamous cancer cells mediated by miR-765-EMP3-p66Shc axis. Cell Signal 34:102. https://doi.org/10.1016/j.cellsig.2017.03.009
Zhou Z, Li H, Liu F (2018) DNA methyltransferase inhibitors and their therapeutic potential. Curr Top Med Chem 18:2448. https://doi.org/10.2174/1568026619666181120150122
Acknowledgements
Thanks to Henan Provincial People's Hospital for supporting this study.
Funding
This work was supported by the Key Project of Henan Natural Science Foundation (232300421123) and the Tackling key problems of science and technology in Henan Province (222102310002, 232102311159) and the Henan Postdoctoral Fund (2021) and the Medical Science and technology research plan in Henan Province (LHGJ20210013) and the Project of Action for Postgraduate Training Innovation and Quality Improvement of Henan University (Grant ID: SYLYC2022174). The funding source was not involved in collecting, analyzing and interpreting the data.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Zhendong Liu, Wenjia Liang and Qingyun Zhu contributed equally to this work and shared first authorship; Zhendong Liu and Yanzheng Gao designed the research; Wenjia Liang, Xingbo Cheng and Rongjun Qian performed the research; Wenjia Liang and Qingyun Zhu wrote the paper. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics Approval and Consent to Participate
The study was conducted according to the guidelines of the Declaration of Helsinki and approved by the ethics committee of Henan Provincial People's hospital (2020, ethical review No. 107). Informed consent was obtained from all subjects involved in the study.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, Z., Liang, W., Zhu, Q. et al. PSRC1 Regulated by DNA Methylation Is a Novel Target for LGG Immunotherapy. J Mol Neurosci 73, 516–528 (2023). https://doi.org/10.1007/s12031-023-02133-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-023-02133-y