Abstract
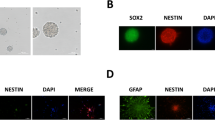
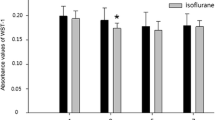
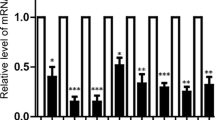
Sevoflurane is a safe and well-known inhaled anesthetic. Given that sevoflurane can be delivered to developing fetuses through the mother, it is critical to determine whether this agent affects fetal neurodevelopment. Recent research has sought to determine whether sevoflurane affects fetal brain development when the mother is exposed during the second to third trimester of pregnancy, considered to be the crucial period for the development of nervous system. However, even though the first trimester is a critical period for fetal organogenesis and the most susceptible time to teratogen exposure, research regarding the effects of sevoflurane on organogenesis, especially on brain development, is insufficient. In the present study, human embryonic stem cells (hESC)-derived cerebral organoids were exposed to sevoflurane during the time corresponding to the first trimester to investigate the effect of early sevoflurane exposure on fetal brain development, specifically the processes of neuronal differentiation and maturation. Organoid size exposed to the intermediate concentration of sevoflurane did not differ from control, immunofluorescence demonstrated that sevoflurane temporarily decreased the size of SOX2 + /N-cad + ventricular zone structures only during the mid-time point, and upregulated expression of TUJ1 and MAP2 only during the early time point. However, all markers returned to normal levels, and organoids formed normal cortical structures at the late time point. Our results suggest that maternal sevoflurane exposure during the first trimester of pregnancy can cause abnormal neuronal differentiation in the fetal brain. However, considering the recovery observed in later periods, sevoflurane exposure might not have lasting impacts on fetal brain development.



Similar content being viewed by others
References
Alwan S, Chambers CD (2015) Identifying human teratogens: an update. J Pediatr Genet 4(2):39–41. https://doi.org/10.1055/s-0035-1556745
Arai Y, Taverna E (2017) Neural progenitor cell polarity and cortical development. Front Cell Neurosci 11:384. https://doi.org/10.3389/fncel.2017.00384
Brambrink AM, Evers AS, Avidan MS, Farber NB, Smith DJ, Zhang X, Olney JW (2010) Isoflurane-induced neuroapoptosis in the neonatal rhesus macaque brain. Anesthesiology 112(4):834–841. https://doi.org/10.1097/ALN.0b013e3181d049cd
Brioni JD, Varughese S, Ahmed R, Bein B (2017) A clinical review of inhalation anesthesia with sevoflurane: from early research to emerging topics. J Anesth 31(5):764–778. https://doi.org/10.1007/s00540-017-2375-6
Broccoli V, Giannelli SG, Mazzara PG (2014) Modeling physiological and pathological human neurogenesis in the dish. Front Neurosci 8:183. https://doi.org/10.3389/fnins.2014.00183
Chai D, Cheng Y, Jiang H (2019) Fundamentals of fetal toxicity relevant to sevoflurane exposures during pregnancy. Int J Dev Neurosci 72:31–35. https://doi.org/10.1016/j.ijdevneu.2018.11.001
Cui FH, Li J, Li KZ, Xie YG, Zhao XL (2021) Effects of sevoflurane exposure during different stages of pregnancy on the brain development of rat offspring. J Anesth 35(5):654–662. https://doi.org/10.1007/s00540-021-02972-2
Davidson AJ, Disma N, de Graaff JC, Withington DE, Dorris L, Bell G, McCann ME (2016) Neurodevelopmental outcome at 2 years of age after general anaesthesia and awake-regional anaesthesia in infancy (GAS): an international multicentre, randomised controlled trial. The Lancet 387(10015):239–250. https://doi.org/10.1016/s0140-6736(15)00608-x
Di Lullo E, Kriegstein AR (2017) The use of brain organoids to investigate neural development and disease. Nat Rev Neurosci 18(10):573–584. https://doi.org/10.1038/nrn.2017.107
Esper T, Wehner M, Meinecke CD, Rueffert H (2015) Blood/gas partition coefficients for isoflurane, sevoflurane, and desflurane in a clinically relevant patient population. Anesth Analg 120(1):45–50. https://doi.org/10.1213/ANE.0000000000000516
Fang F, Song R, Ling X, Peng M, Xue Z, Cang J (2017) Multiple sevoflurane anesthesia in pregnant mice inhibits neurogenesis of fetal hippocampus via repressing transcription factor Pax6. Life Sci 175:16–22. https://doi.org/10.1016/j.lfs.2017.03.003
FDA Drug Safety Communication (2017) FDA review results in new warnings about using general anesthetics and sedation drugs in young children and pregnant women. https://www.fda.gov/drugs/drug-safety-and-availability/fda-drug-safety-communication-fda-review-results-new-warnings-about-using-general-anesthetics-and.
Gascoigne DA, Serdyukova NA, Aksenov DP (2021) Early development of the GABAergic system and the associated risks of neonatal anesthesia. Int J Mol Sci 22(23). https://doi.org/10.3390/ijms222312951
Grenier K, Kao J, Diamandis P (2020) Three-dimensional modeling of human neurodegeneration: brain organoids coming of age. Mol Psychiatry 25(2):254–274. https://doi.org/10.1038/s41380-019-0500-7
Gumusoglu SB, Stevens HE (2019) Maternal inflammation and neurodevelopmental programming: a review of preclinical outcomes and implications for translational psychiatry. Biol Psychiatry 85(2):107–121. https://doi.org/10.1016/j.biopsych.2018.08.008
Hong YJ, Do JT (2019) Neural lineage differentiation from pluripotent stem cells to mimic human brain tissues. Front Bioeng Biotechnol 7:400. https://doi.org/10.3389/fbioe.2019.00400
Jiang M, Tang T, Liang X, Li J, Qiu Y, Liu S, Cang J (2021) Maternal sevoflurane exposure induces temporary defects in interkinetic nuclear migration of radial glial progenitors in the fetal cerebral cortex through the Notch signalling pathway. Cell Prolif 54(6):e13042. https://doi.org/10.1111/cpr.13042
Kelava I, Lancaster MA (2016) Dishing out mini-brains: current progress and future prospects in brain organoid research. Dev Biol 420(2):199–209. https://doi.org/10.1016/j.ydbio.2016.06.037
Kotani N, Takahashi S, Sessler DI, Hashiba E, Kubota T, Hashimoto H, Matsuki A (1999) Volatile anesthetics augment expression of proinflammatory cytokines in rat alveolar macrophages during mechanical ventilation. Anesthesiology 91(1):187–197. https://doi.org/10.1097/00000542-199907000-00027
Lancaster MA, Knoblich JA (2014) Generation of cerebral organoids from human pluripotent stem cells. Nat Protoc 9(10):2329–2340. https://doi.org/10.1038/nprot.2014.158
Lancaster MA, Renner M, Martin CA, Wenzel D, Bicknell LS, Hurles ME, Knoblich JA (2013) Cerebral organoids model human brain development and microcephaly. Nature 501(7467):373–379. https://doi.org/10.1038/nature12517
Lee CT, Bendriem RM, Wu WW, Shen RF (2017) 3D brain organoids derived from pluripotent stem cells: promising experimental models for brain development and neurodegenerative disorders. J Biomed Sci 24(1):59. https://doi.org/10.1186/s12929-017-0362-8
Lee JR, Loepke AW (2018) Does pediatric anesthesia cause brain damage? - Addressing parental and provider concerns in light of compelling animal studies and seemingly ambivalent human data. Korean J Anesthesiol 71(4):255–273. https://doi.org/10.4097/kja.d.18.00165
Li X, Jiang X, Gao Q, Zhao P (2021) FOXO3 regulates sevoflurane-induced neural stem cell differentiation in fetal rats. Cell Mol Neurobiol. https://doi.org/10.1007/s10571-021-01055-w
Liu S, Fang F, Song R, Gao X, Jiang M, Cang J (2018) Sevoflurane affects neurogenesis through cell cycle arrest via inhibiting wnt/beta-catenin signaling pathway in mouse neural stem cells. Life Sci 209:34–42. https://doi.org/10.1016/j.lfs.2018.07.054
Longinus EN, Chinwe O, Yvonne DBO (2012) Case report: the obstetric patient going for non-obstetric surgery. Jos J Med 6
Lu Y, Wu X, Dong Y, Xu Z, Zhang Y, Xie Z (2010) Anesthetic sevoflurane causes neurotoxicity differently in neonatal naive and Alzheimer disease transgenic mice. Anesthesiology 112(6):1404–1416. https://doi.org/10.1097/ALN.0b013e3181d94de1
Luhmann HJ, Fukuda A, Kilb W (2015) Control of cortical neuronal migration by glutamate and GABA. Front Cell Neurosci 9:4. https://doi.org/10.3389/fncel.2015.00004
Nie H, Peng Z, Lao N, Dong H, Xiong L (2013) Effects of sevoflurane on self-renewal capacity and differentiation of cultured neural stem cells. Neurochem Res 38(8):1758–1767. https://doi.org/10.1007/s11064-013-1074-4
Nishiyama T, Yamashita K, Yokoyama T (2005) Stress hormone changes in general anesthesia of long duration: isoflurane-nitrous oxide vs sevoflurane-nitrous oxide anesthesia. J Clin Anesth 17(8):586–591. https://doi.org/10.1016/j.jclinane.2005.03.009
Oliveira B, Cerag Yahya A, Novarino G (2019) Modeling cell-cell interactions in the brain using cerebral organoids. Brain Res 1724:146458. https://doi.org/10.1016/j.brainres.2019.146458
Petroff OA (2002) GABA and glutamate in the human brain. Neuroscientist 8(6):562–573. https://doi.org/10.1177/1073858402238515
Qiu J, Shi P, Mao W, Zhao Y, Liu W, Wang Y (2015) Effect of apoptosis in neural stem cells treated with sevoflurane. BMC Anesthesiol 15:25. https://doi.org/10.1186/s12871-015-0018-8
Sachs A, Guglielminotti J, Miller R, Landau R, Smiley R, Li G (2017) Risk factors and risk stratification for adverse obstetrical outcomes after appendectomy or cholecystectomy during pregnancy. JAMA Surg 152(5):436–441. https://doi.org/10.1001/jamasurg.2016.5045
Satomoto M, Sun Z, Adachi YU, Makita K (2018) Neonatal sevoflurane exposure induces adulthood fear-induced learning disability and decreases glutamatergic neurons in the basolateral amygdala. J Neurosurg Anesthesiol 30(1):59–64. https://doi.org/10.1097/ANA.0000000000000387
Shan Y, Yang F, Tang Z, Bi C, Sun S, Zhang Y, Liu H (2018) Dexmedetomidine ameliorates the neurotoxicity of sevoflurane on the immature brain through the BMP/SMAD signaling pathway. Front Neurosci 12:964. https://doi.org/10.3389/fnins.2018.00964
Silbereis JC, Pochareddy S, Zhu Y, Li M, Sestan N (2016) The cellular and molecular landscapes of the developing human central nervous system. Neuron 89(2):248–268. https://doi.org/10.1016/j.neuron.2015.12.008
Sun LS, Li G, Miller TL, Salorio C, Byrne MW, Bellinger DC, McGowan FX (2016) Association between a single general anesthesia exposure before age 36 months and neurocognitive outcomes in later childhood. JAMA 315(21):2312–2320. https://doi.org/10.1001/jama.2016.6967
Suong DNA, Imamura K, Inoue I, Kabai R, Sakamoto S, Okumura T, Inoue H (2021) Induction of inverted morphology in brain organoids by vertical-mixing bioreactors. Commun Biol 4(1):1213. https://doi.org/10.1038/s42003-021-02719-5
Tambalo M, Lodato S (2020) Brain organoids: human 3D models to investigate neuronal circuits assembly, function and dysfunction. Brain Res 1746:147028. https://doi.org/10.1016/j.brainres.2020.147028
Watanabe M, Buth JE, Vishlaghi N, de la Torre-Ubieta L, Taxidis J, Khakh BS, Novitch BG (2017) Self-organized cerebral organoids with human-specific features predict effective drugs to combat Zika virus infection. Cell Rep 21(2):517–532. https://doi.org/10.1016/j.celrep.2017.09.047
Yang Z, Lv J, Li X, Meng Q, Yang Q, Ma W, Ke ZJ (2017) Sevoflurane decreases self-renewal capacity and causes c-Jun N-terminal kinase-mediated damage of rat fetal neural stem cells. Sci Rep 7:46304. https://doi.org/10.1038/srep46304
Yin X, Xu JC, Cho GS, Kwon C, Dawson TM, Dawson VL (2019) Neurons derived from human induced pluripotent stem cells integrate into rat brain circuits and maintain both excitatory and inhibitory synaptic activities. eNeuro 6(4). https://doi.org/10.1523/ENEURO.0148-19.2019
Yu CH, Weng SF, Ho CH, Chen YC, Chen JY, Chang YJ, Chu CC (2018) Pregnancy outcomes following nonobstetric surgery during gestation: a nationwide population-based case-control study in Taiwan. BMC Pregnancy Childbirth 18(1):460. https://doi.org/10.1186/s12884-018-2079-4
Yu Q, Feng N, Hu Y, Luo F, Zhao W, Zhao W, Liu Z, Li M, Xu L, Wu L, Liu Y (2019) Suberoylanilide hydroxamic acid (SAHA) alleviates the learning and memory impairment in rat offspring caused by maternal sevoflurane exposure during late gestation. J Toxicol Sci 44 (3):177–189. https://doi.org/10.2131/jts.44.177
Zhang J, Chen Z, Luo X, Yang Z (2020a) TrkC overexpression protects sevoflurane-induced neurotoxicity in human induced pluripotent stem cell-derived neurons. Dev Neurosci 42(2–4):105–113. https://doi.org/10.1159/000510326
Zhang Y, Wu Z, Li X, Wan Y, Zhang Y, Zhao P (2020b) Maternal sevoflurane exposure affects differentiation of hippocampal neural stem cells by regulating miR-410-3p and ATN1. Stem Cell Res Ther 11(1):423. https://doi.org/10.1186/s13287-020-01936-9
Zhu Y, Lv C, Liu J, Shang S, Jing W (2021) Effects of sevoflurane general anesthesia during early pregnancy on AIM2 expression in the hippocampus and parietal cortex of Sprague-Dawley offspring rats. Exp Ther Med 21(5):469. https://doi.org/10.3892/etm.2021.9900
Funding
This research was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF), funded by the Ministry of Education (2019R1I1A1A01064174); by the 3D-TissueChip Based Drug Discovery Platform Program through the Korea Evaluation Institute of Industrial Technology, funded by the Ministry of Commerce, Industry and Energy (20009773); and by a grant from the Korea Health Technology R&D Project through the Korea Health Industry Development Institute, funded by the Ministry of Health & Welfare, Republic of Korea (HI18C2458020021).
Author information
Authors and Affiliations
Contributions
J.Y. designed and supervised the study. J.A.L planned and performed all experiments. J.A.L, D.H.B, and Y.S.B analyzed data and wrote the manuscript. W.H.C and C.H.C reviewed the manuscript.
Corresponding authors
Ethics declarations
Conflict of Interest
The authors declare no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lee, J.A., Bae, D.H., Choi, W.H. et al. Effects of Sevoflurane Exposure on Fetal Brain Development Using Cerebral Organoids. J Mol Neurosci 72, 2440–2450 (2022). https://doi.org/10.1007/s12031-022-02080-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-022-02080-0