Abstract
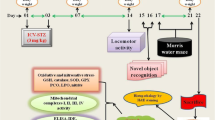
Insulin signaling disruption and caspase-3 cleavage play a pathologic role in Alzheimer’s disease (AD). Evidence suggested that cinnamaldehyde (Cin), the major component of cinnamon, has the ability to act as a neuroprotective agent. However, little evidence is available to demonstrate its effectiveness in regulating the insulin and caspase-3 signaling pathways and underlying molecular mechanisms. Therefore, the present study was conducted to correlate the molecular mechanisms of these signaling pathways and Cin treatment on animal behavioral performance in an intracerebroventricular (ICV)-streptozotocin (STZ, 3 mg/kg) model. The sporadic AD rat model was treated with Cin (10 and 100 mg/kg; intraperitoneal, i.p) daily for 2 weeks. Novel object recognition (NOR), Morris water maze (MWM), and elevated plus maze (EPM) tests were performed to assess recognition/spatial memory and anxiety-like behavior, respectively. Hippocampal Aβ aggregation was assessed using Congo red staining. The activity of hippocampal caspase-3 and IRS-1/Akt/GSK-3β signaling pathways were analyzed using the Western blot technique. The results revealed that Cin (100 mg/kg, effective dose) improved recognition/spatial memory deficits and anxiety-like behavior. In addition, Cin negated the effects of STZ on Aβ aggregation and caspase-3 cleavage in the hippocampus. Furthermore, the Western blot method showed that hippocampal IRS-1/AKT/GSK-3β phosphorylation was altered in ICV-STZ animal model, while Cin modulated this signaling pathway through decreasing Phospho.IRS-1Ser307/Total.IRS-1 ratio and also increasing Phospho.AktSer473/Total.Akt and Phospho.GSK-3βSer9/Total.GSK-3β ratios. These findings suggest that Cin is involved in the regulation of hippocampal IRS-1/AKT/GSK-3β and caspase-3 pathways in a sporadic AD model, and modulation of these signaling pathways also influences the animal behavioral performance.
Graphical Abstract












Similar content being viewed by others
Data Availability
The data that support the finding of this study are available from the senior author (rghasemi60@sbmu.ac.ir) upon reasonable request.
Abbreviations
- AD:
-
Alzheimer’s disease
- i.p:
-
Intraperitoneal
- Aβ:
-
Amyloid-β
- ICV:
-
Intracerebroventricular
- STZ:
-
Streptozotocin
- MWM:
-
Morris water maze
- NOR:
-
Novel object recognition
- EPM:
-
Elevated plus maze
- IR:
-
Insulin receptor
- IRS:
-
Insulin receptor substrate
- PI3K:
-
Phosphoinositide 3-kinase
- Akt:
-
A serine/threonine protein kinase
- IGF:
-
Insulin-like growth factor
- GSK-3:
-
Glycogen synthase kinase-3
- Cin:
-
Cinnamaldehyde
- HRP:
-
Horseradish peroxidase
- ECL:
-
Enhanced chemiluminescence
- PVDF:
-
Polyvinylidene difluoride
- PBS:
-
Phosphate buffer saline
- OAT:
-
Open arm time
- CAT:
-
Close arm time
- OAE:
-
Open arm entry
- CAE:
-
Close arm entry
- Q:
-
Quadrant
- LN2 :
-
Liquid nitrogen
- DG:
-
Dentate gyrus
References
Agrawal R, Tyagi E, Shukla R, Nath C (2009) A study of brain insulin receptors, AChE activity and oxidative stress in rat model of ICV STZ induced dementia. Neuropharmacology 56(4):779–787. https://doi.org/10.1016/j.neuropharm.2009.01.005
Akhtar A, Bishnoi M, Sah SP (2020a) Sodium orthovanadate improves learning and memory in intracerebroventricular-streptozotocin rat model of Alzheimer’s disease through modulation of brain insulin resistance induced tau pathology. Brain Res Bull 164:83–97. https://doi.org/10.1016/j.brainresbull.2020.08.001
Akhtar A, Dhaliwal J, Saroj P, Uniyal A, Bishnoi M, Sah SP (2020b) Chromium picolinate attenuates cognitive deficit in ICV-STZ rat paradigm of sporadic Alzheimer’s-like dementia via targeting neuroinflammatory and IRS-1/PI3K/AKT/GSK-3β pathway. Inflammopharmacology 28(2):385–400. https://doi.org/10.1007/s10787-019-00681-7
Bagheri-Mohammadi S (2021a) Microglia in Alzheimer’s disease: the role of stem cell-microglia interaction in brain homeostasis. Neurochem Res 46(2):141–148. https://doi.org/10.1007/s11064-020-03162-4
Bagheri-Mohammadi S (2021b) Stem cell-based therapy as a promising approach in Alzheimer’s disease: current perspectives on novel treatment. Cell Tissue Bank 22(3):339–353. https://doi.org/10.1007/s10561-020-09896-3
Bagheri-Mohammadi S, Alani B, Karimian M, Moradian-Tehrani R, Noureddini M (2019) Intranasal administration of endometrial mesenchymal stem cells as a suitable approach for Parkinson’s disease therapy. Mol Biol Rep 46(4):4293–4302. https://doi.org/10.1007/s11033-019-04883-8
Chen Y, Liang Z, Blanchard J, Dai CL, Sun S, Lee MH, Grundke-Iqbal I, Iqbal K, Liu F, Gong CX (2013) A non-transgenic mouse model (icv-STZ mouse) of Alzheimer’s disease: similarities to and differences from the transgenic model (3xTg-AD mouse). Mol Neurobiol 47(2):711–725. https://doi.org/10.1007/s12035-012-8375-5
Choi YH (2021) Trans-cinnamaldehyde protects C2C12 myoblasts from DNA damage, mitochondrial dysfunction and apoptosis caused by oxidative stress through inhibiting ROS production. Genes Genomics 43(4):303–312. https://doi.org/10.1007/s13258-020-00987-9
Craft S (2005) Insulin resistance syndrome and Alzheimer’s disease: age-and obesity-related effects on memory, amyloid, and inflammation. Neurobiol Aging 26(1):65–69. https://doi.org/10.1016/j.neurobiolaging.2005.08.021
De la Monte SM, Wands JR (2008) Alzheimer’s disease is type 3 diabetes—evidence reviewed. J Diabetes Sci Technol 2(6):1101–1113. https://doi.org/10.1177/193229680800200619
Dos Santos JP, Vizuete A, Hansen F, Biasibetti R, Gonçalves CA (2018) Early and persistent O-GlcNAc protein modification in the streptozotocin model of Alzheimer’s disease. J Alzheimer’s Dis 61(1):237–249. https://doi.org/10.3233/JAD-170211
Doyle AA, Stephens JC (2019) A review of cinnamaldehyde and its derivatives as antibacterial agents. Fitoterapia 139:104405. https://doi.org/10.1016/j.fitote.2019.104405
Fu W, Ruangkittisakul A, MacTavish D, Shi JY, Ballanyi K, Jhamandas JH (2012) Amyloid β (Aβ) peptide directly activates amylin-3 receptor subtype by triggering multiple intracellular signaling pathways. J Biol Chem 287(22):18820–18830. https://doi.org/10.1074/jbc.M111.331181
Ghasemi R, Dargahi L, Haeri A, Moosavi M, Mohamed Z, Ahmadiani A (2013) Brain insulin dysregulation: implication for neurological and neuropsychiatric disorders. Mol Neurobiol 47(3):1045–1065. https://doi.org/10.1007/s12035-013-8404-z
Hajisoltani R, Karimi SA, Rahdar M, Davoudi S, Borjkhani M, Hosseinmardi N, Behzadi G, Janahmadi M (2019) Hyperexcitability of hippocampal CA1 pyramidal neurons in male offspring of a rat model of autism spectrum disorder (ASD) induced by prenatal exposure to valproic acid: a possible involvement of Ih channel current. Brain Res 1708:188–199. https://doi.org/10.1016/j.brainres.2018.12.011
Hooper C, Killick R, Lovestone S (2008) The GSK3 hypothesis of Alzheimer’s disease. J Neurochem 104(6):1433–1439. https://doi.org/10.1111/j.1471-4159.2007.05194.x
Javadpour P, Askari S, Rashidi FS, Dargahi L, Ahmadiani A, Ghasemi R (2021) Imipramine alleviates memory impairment and hippocampal apoptosis in STZ-induced sporadic Alzheimer’s rat model: possible contribution of MAPKs and insulin signaling. Behav Brain Res 408:113260. https://doi.org/10.1016/j.bbr.2021.113260
Kazerouni A, Nazeri M, Karimzadeh A, SoukhakLari R, Moezi L, Pirsalami F, Moosavi M (2020) Sub-chronic oral cinnamaldehyde treatment prevents scopolamine-induced memory retrieval deficit and hippocampal Akt and MAPK dysregulation in male mice. Neurol Res 42(2):99–107. https://doi.org/10.1080/01616412.2019.1709142
Lee HK, Kumar P, Fu Q, Rosen KM, Querfurth HW (2009) The insulin/Akt signaling pathway is targeted by intracellular β-amyloid. Mol Biol Cell 20(5):1533–1544. https://doi.org/10.1091/mbc.e08-07-0777
Lee J, Kim MS (2007) The role of GSK3 in glucose homeostasis and the development of insulin resistance. Diabetes Res Clin Pract 77(3):S49-57. https://doi.org/10.1016/j.diabres.2007.01.033
Lueptow LM (2017) Novel object recognition test for the investigation of learning and memory in mice. J vis Exp 30(126):e55718. https://doi.org/10.3791/55718
Lv C, Yuan X, Zeng HW, Liu RH, Zhang WD (2017) Protective effect of cinnamaldehyde against glutamate-induced oxidative stress and apoptosis in PC12 cells. Eur J Pharmacol 815:487–494. https://doi.org/10.1016/j.ejphar.2017.09.009
Mattson MP (2004) Pathways towards and away from Alzheimer’s disease. Nature 430(7000):631–639. https://doi.org/10.1038/nature02621
Mendes CT, Mury FB, de Sá ME, Alberto FL, Forlenza OV, Dias-Neto E, Gattaz WF (2009) Lithium reduces Gsk3b mRNA levels: implications for Alzheimer disease. Eur Arch Psychiatry Clin Neurosci 259(1):16–22. https://doi.org/10.1007/s00406-008-0828-5
Miedel CJ, Patton JM, Miedel AN, Miedel ES, Levenson JM (2017) Assessment of spontaneous alternation, novel object recognition and limb clasping in transgenic mouse models of amyloid-β and tau neuropathology. J Vis Exp: JoVE (123). https://doi.org/10.3791/55523
Moreira PI (2012) Alzheimer’s disease and diabetes: an integrative view of the role of mitochondria, oxidative stress, and insulin. J Alzheimers Dis 30(s2):S199-215. https://doi.org/10.3233/JAD-2011-111127
Mustafa HN (2020) Neuro-amelioration of cinnamaldehyde in aluminum-induced Alzheimer’s disease rat model. J Histotechnol 43(1):11–20. https://doi.org/10.1080/01478885.2019.1652994
Noureddini M, Bagheri-Mohammadi S (2021) Adult hippocampal neurogenesis and Alzheimer’s disease: novel application of mesenchymal stem cells and their role in hippocampal neurogenesis. Int J Mol Cell Med 10(1):1. https://doi.org/10.22088/IJMCM.BUMS.10.1.1
O’Neill C (2013) PI3-kinase/Akt/mTOR signaling: impaired on/off switches in aging, cognitive decline and Alzheimer’s disease. Exp Gerontol 48(7):647–653. https://doi.org/10.1016/j.exger.2013.02.025
Pei JJ, Khatoon S, An WL, Nordlinder M, Tanaka T, Braak H, Tsujio I, Takeda M, Alafuzoff I, Winblad B, Cowburn RF (2003) Role of protein kinase B in Alzheimer’s neurofibrillary pathology. Acta Neuropathol 105(4):381–392. https://doi.org/10.1007/s00401-002-0657-y
Pham HM, Xu A, Schriner SE, Sevrioukov EA, Jafari M (2018) Cinnamaldehyde improves lifespan and healthspan in Drosophila melanogaster models for Alzheimer’s disease. Biomed Res Int. https://doi.org/10.1155/2018/3570830
Roth KA (2001) Caspases, apoptosis, and Alzheimer disease: causation, correlation, and confusion. J Neuropathol Exp Neurol 60(9):829–838. https://doi.org/10.1093/jnen/60.9.829
Roy A, Sharma S, Nag TC, Katyal J, Gupta YK, Jain S (2022) Cognitive dysfunction and anxiety resulted from synaptic downscaling, hippocampal atrophy and ventricular enlargement in icv streptozotocin injected male Wistar rats. bioRxiv. https://doi.org/10.1101/2022.04.04.486747
Saeed M, Ghadiri A, Hadizadeh F, Attaranzadeh A, Alavi MS, Etemad L (2018) Cinnamaldehyde improves methamphetamine-induced spatial learning and memory deficits and restores ERK signaling in the rat prefrontal cortex. Iran J Basic Med Sci 21(12):1316. https://doi.org/10.22038/IJBMS.2018.35368.8427
Sarkar S, Raymick J, Cuevas E, Rosas-Hernandez H, Hanig J (2020) Modification of methods to use Congo-red stain to simultaneously visualize amyloid plaques and tangles in human and rodent brain tissue sections. Metab Brain Dis 35(8):1371–1383. https://doi.org/10.1007/s11011-020-00608-0
Slee EA, Adrain C, Martin SJ (2001) Executioner caspase-3,-6, and-7 perform distinct, non-redundant roles during the demolition phase of apoptosis. J Biol Chem 276(10):7320–7326. https://doi.org/10.1074/jbc.M008363200
SoukhakLari R, Borhani-Haghighi A, Farsadrooh A, Moezi L, Pirsalami F, Kazerouni A, Safari A, Moosavi M (2019) The effect of cinnamaldehyde on passive avoidance memory and hippocampal Akt, ERK and GSK-3β in mice. Eur J Pharmacol 859:172530. https://doi.org/10.1016/j.ejphar.2019.172530
Suzanne M (2009) Insulin resistance and Alzheimer’s disease. BMB Rep 42(8):475
Townsend M, Mehta T, Selkoe DJ (2007) Soluble Aβ inhibits specific signal transduction cascades common to the insulin receptor pathway. J Biol Chem 282(46):33305–33312. https://doi.org/10.1074/jbc.M610390200
Tramutola A, Lanzillotta C, Di Domenico F, Head E, Butterfield DA, Perluigi M, Barone E (2020) Brain insulin resistance triggers early onset Alzheimer disease in Down syndrome. Neurobiol Dis 137:104772. https://doi.org/10.1016/j.nbd.2020.104772
Tyagi A, Pugazhenthi S (2021) Targeting insulin resistance to treat cognitive dysfunction. Mol Neurobiol. https://doi.org/10.1007/s12035-021-02283-3
Vahidinia Z, Alipour N, Atlasi MA, Naderian H, Beyer C, Azami Tameh A (2017) Gonadal steroids block the calpain-1-dependent intrinsic pathway of apoptosis in an experimental rat stroke model. Neurol Res 39(1):54–64. https://doi.org/10.1080/01616412.2016.1250459
Vila M, Przedborski S (2003) Targeting programmed cell death in neurodegenerative diseases. Nat Rev Neurosci 4(5):365–375. https://doi.org/10.1038/nrn1100
Zhang L, Zhang Z, Fu Y, Yang P, Qin Z, Chen Y, Xu Y (2016) Trans-cinnamaldehyde improves memory impairment by blocking microglial activation through the destabilization of iNOS mRNA in mice challenged with lipopolysaccharide. Neuropharmacology 110:503–518. https://doi.org/10.1016/j.neuropharm.2016.08.013
Zhao WQ, Lacor PN, Chen H, Lambert MP, Quon MJ, Krafft GA, Klein WL (2009) Insulin receptor dysfunction impairs cellular clearance of neurotoxic oligomeric Aβ. J Biol Chem 284(28):18742–18753. https://doi.org/10.1074/jbc.M109.011015
Zhao Y, Deng H, Li K, Wang L, Wu Y, Dong X, Wang X, Chen Y, Xu Y (2019) Trans-cinnamaldehyde improves neuroinflammation-mediated NMDA receptor dysfunction and memory deficits through blocking NF-κB pathway in presenilin1/2 conditional double knockout mice. Brain Behav Immun 82:45–62. https://doi.org/10.1016/j.bbi.2019.07.032
Acknowledgements
This paper has been extracted from the Ph.D thesis (Dissertation, Medical physiology) written by Dr. Saeid Bagheri-Mohammadi in School of Medicine, Shahid Beheshti University of Medical Sciences (Registration number: 260) and supported by a Grant (Number: 25205) from Deputy of Research and Technology, Shahid Beheshti University of Medical Sciences, Tehran, Iran.
Funding
This work was supported by a Grant (Number: 25205) from Deputy of Research and Technology, Shahid Beheshti University of Medical Sciences, Tehran, Iran.
Author information
Authors and Affiliations
Contributions
Saeid Bagheri-Mohammadi: conceptualization, methodology, formal analysis, investigation, resources, validation, writing —review and editing. Rasoul Ghasemi: providing the complete research project, conceptualization, supervision, methodology, formal analysis, investigation, resources, validation, project administration, writing—review and editing. Maryam Moosavi: supervision, investigation, resources, validation, project administration, review and editing. Sahar Askari: validation, review and editing;. Behrang Alani: supervision, resources, project administration, and review and editing.
Corresponding authors
Ethics declarations
Ethics Approval
All of the experimental procedures were approved by the Ethical Committee for Research at Shahid Beheshti University of Medical Sciences (ID: IR.SBMU.MSP.REC.1399.385).
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlights
• This study investigated the molecular mechanism of hippocampal insulin signaling pathway and cinnamaldehyde treatment on behavioral performance in sporadic Alzheimer's disease model.
• Treatment with cinnamaldehyde ameliorated the STZ-induced recognition/spatial memory impairment.
• Central microinjection of STZ increased anxiety-like behavior, which was reversed by the administration of cinnamaldehyde.
• Cinnamaldehyde attenuates hippocampal Aβ aggregation induced by central microinjection of STZ.
• Cinnamaldehyde prevented cleavage of hippocampal caspase-3 in a sporadic Alzheimer's disease animal model.
• Cinnamaldehyde prevented hippocampal IRS-1, AKT, and GSK-3β signaling disruption induced by central microinjection of STZ.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Bagheri-Mohammadi, S., Askari, S., Alani, B. et al. Cinnamaldehyde Regulates Insulin and Caspase-3 Signaling Pathways in the Sporadic Alzheimer’s Disease Model: Involvement of Hippocampal Function via IRS-1, Akt, and GSK-3β Phosphorylation. J Mol Neurosci 72, 2273–2291 (2022). https://doi.org/10.1007/s12031-022-02075-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-022-02075-x