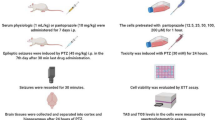
Abstract
The transporter hypothesis is one of the most popular hypotheses of drug-resistant epilepsy (DRE). P-glycoprotein (P-gp), a channel protein at the blood–brain barrier (BBB), plays an important role in the transport of some anti-seizure drugs from brain tissue into vessels, which reduces drug concentrations and diminishes the effects of drug treatment. We performed this study to test whether P-gp is overexpressed in DRE and identify ways to prevent and reverse DRE. In this study, we established a phenytoin (PHT)-resistant mouse model and revealed that P-gp was overexpressed at the BBB in PHT-resistant mice. The P-gp inhibitor nimodipine decreased the resistance of phenytoin. Antioxidative preventive treatment with N-acetylcysteine (NAC) prevented the mice from entering a PHT-resistant state, and NAC therapy tended to reverse PHT resistance into sensitivity. We were also able to induce PHT resistance by activating the Nrf2/P-gp pathway, which indicates that oxidative stress plays an important role in drug resistance. Taken together, these findings suggest that antioxidative therapy may be a promising strategy for overcoming DRE.







Similar content being viewed by others
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Amin ML (2013) P-glycoprotein inhibition for optimal drug delivery. Drug Target Insights 7:27–34. https://doi.org/10.4137/DTI.S12519
Asadi-Pooya AA, Razavizadegan SM, Abdi-Ardekani A et al (2013) Adjunctive use of verapamil in patients with refractory temporal lobe epilepsy: a pilot study. Epilepsy Behav 29:150–154. https://doi.org/10.1016/j.yebeh.2013.07.006
Ashraf T, Kao A, Bendayan R (2014) Functional expression of drug transporters in glial cells: potential role on drug delivery to the CNS. Adv Pharmacol 71:45–111. https://doi.org/10.1016/bs.apha.2014.06.010
Bales KR, O’Neill SM, Pozdnyakov N et al (2016) Passive immunotherapy targeting amyloid-beta reduces cerebral amyloid angiopathy and improves vascular reactivity. Brain 139:563–577. https://doi.org/10.1093/brain/awv313
Bauer B, Hartz AM, Pekcec A et al (2008) Seizure-induced up-regulation of P-glycoprotein at the blood-brain barrier through glutamate and cyclooxygenase-2 signaling. Mol Pharmacol 73:1444–1453. https://doi.org/10.1124/mol.107.041210
Beaulieu E, Demeule M, Ghitescu L et al (1997) P-glycoprotein is strongly expressed in the luminal membranes of the endothelium of blood vessels in the brain. Biochem J 326:539–544. https://doi.org/10.1042/bj3260539
Binvignat O, Olloquequi J (2020) Excitotoxicity as a target against neurodegenerative processes. Curr Pharm Des 26:1251–1262. https://doi.org/10.2174/1381612826666200113162641
Boulay AC, Saubamea B, Decleves X et al (2015) Purification of mouse brain vessels. J Vis Exp e53208. https://doi.org/10.3791/53208
Chen Z, Brodie MJ, Liew D et al (2018) Treatment outcomes in patients with newly diagnosed epilepsy treated with established and new antiepileptic drugs: a 30-year longitudinal cohort study. JAMA Neurol 75:279–286. https://doi.org/10.1001/jamaneurol.2017.3949
Chen Z, Shi T, Zhang L et al (2016) Mammalian drug efflux transporters of the ATP binding cassette (ABC) family in multidrug resistance: a review of the past decade. Cancer Lett 370:153–164. https://doi.org/10.1016/j.canlet.2015.10.010
Chi X, Huang C, Li R et al (2017) Inhibition of mTOR pathway by rapamycin decreases P-glycoprotein expression and spontaneous seizures in pharmacoresistant epilepsy. J Mol Neurosci 61:553–562. https://doi.org/10.1007/s12031-017-0897-x
De Sarro G, Ferreri G, Gareri P et al (2003) Comparative anticonvulsant activity of some 2,3-benzodiazepine derivatives in rodents. Pharmacol Biochem Behav 74:595–602. https://doi.org/10.1016/s0091-3057(02)01040-7
Enrique A, Goicoechea S, Castano R et al (2017) New model of pharmacoresistant seizures induced by 3-mercaptopropionic acid in mice. Epilepsy Res 129:8–16. https://doi.org/10.1016/j.eplepsyres.2016.10.012
Fiest KM, Sauro KM, Wiebe S et al (2017) Prevalence and incidence of epilepsy: a systematic review and meta-analysis of international studies. Neurology 88:296–303. https://doi.org/10.1212/WNL.0000000000003509
Gaitatzis A, Johnson AL, Chadwick DW et al (2004) Life expectancy in people with newly diagnosed epilepsy. Brain 127:2427–2432. https://doi.org/10.1093/brain/awh267
Gil-Martins E, Barbosa DJ, Silva V et al (2020) Dysfunction of ABC transporters at the blood-brain barrier: Role in neurological disorders. Pharmacol Ther 213:107554. https://doi.org/10.1016/j.pharmthera.2020.107554
He SM, Li R, Kanwar JR et al (2011) Structural and functional properties of human multidrug resistance protein 1 (MRP1/ABCC1). Curr Med Chem 18:439–481. https://doi.org/10.2174/092986711794839197
Hocht C, Lazarowski A, Gonzalez NN et al (2007) Nimodipine restores the altered hippocampal phenytoin pharmacokinetics in a refractory epileptic model. Neurosci Lett 413:168–172. https://doi.org/10.1016/j.neulet.2006.11.075
Janmohamed M, Lawn N, Spilsbury K et al (2021) Starting a new anti-seizure medication in drug-resistant epilepsy: Add-on or substitute? Epilepsia 62:228–237. https://doi.org/10.1111/epi.16765
Kalilani L, Sun X, Pelgrims B et al (2018) The epidemiology of drug-resistant epilepsy: a systematic review and meta-analysis. Epilepsia 59:2179–2193. https://doi.org/10.1111/epi.14596
Kwan P, Brodie MJ (2000) Early identification of refractory epilepsy. N Engl J Med 342:314–319. https://doi.org/10.1056/NEJM200002033420503
Laxer KD, Trinka E, Hirsch LJ et al (2014) The consequences of refractory epilepsy and its treatment. Epilepsy Behav 37:59–70. https://doi.org/10.1016/j.yebeh.2014.05.031
Lazarowski A, Ramos AJ, Garcia-Rivello H et al (2004) Neuronal and glial expression of the multidrug resistance gene product in an experimental epilepsy model. Cell Mol Neurobiol 24:77–85. https://doi.org/10.1023/b:cemn.0000012726.43842.d2
Lhatoo SD, Johnson AL, Goodridge DM et al (2001) Mortality in epilepsy in the first 11 to 14 years after diagnosis: multivariate analysis of a long-term, prospective, population-based cohort. Ann Neurol 49:336–344
Li Y, Chen J, Zeng T et al (2014) Expression of HIF-1alpha and MDR1/P-glycoprotein in refractory mesial temporal lobe epilepsy patients and pharmacoresistant temporal lobe epilepsy rat model kindled by coriaria lactone. Neurol Sci 35:1203–1208. https://doi.org/10.1007/s10072-014-1681-0
McCagh J, Fisk JE, Baker GA (2009) Epilepsy, psychosocial and cognitive functioning. Epilepsy Res 86:1–14. https://doi.org/10.1016/j.eplepsyres.2009.04.007
Mohanraj R, Norrie J, Stephen L et al (2006) Mortality in adults with newly diagnosed and chronic epilepsy: a retrospective comparative study. Lancet Neurol 5:481–487. https://doi.org/10.1016/s1474-4422(06)70448-3
Moshe SL, Perucca E, Ryvlin P et al (2015) Epilepsy: new advances. Lancet 385:884–898. https://doi.org/10.1016/S0140-6736(14)60456-6
Murray CJ, Vos T, Lozano R et al (2012) Disability-adjusted life years (DALYs) for 291 diseases and injuries in 21 regions, 1990–2010: a systematic analysis for the Global Burden of Disease Study 2010. Lancet 380:2197–2223. https://doi.org/10.1016/S0140-6736(12)61689-4
Pascaud C, Garrigos M, Orlowski S (1998) Multidrug resistance transporter P-glycoprotein has distinct but interacting binding sites for cytotoxic drugs and reversing agents. Biochem J 333(Pt 2):351–358. https://doi.org/10.1042/bj3330351
Racine RJ (1972) Modification of seizure activity by electrical stimulation. II Motor Seizure Electroencephalogr Clin Neurophysiol 32:281–294. https://doi.org/10.1016/0013-4694(72)90177-0
Reddy DS (2020) Brain structural and neuroendocrine basis of sex differences in epilepsy. Handb Clin Neurol 175:223–233. https://doi.org/10.1016/b978-0-444-64123-6.00016-3
Reyes RC, Cittolin-Santos GF, Kim JE et al (2016) Neuronal glutathione content and antioxidant capacity can be normalized in situ by n-acetyl cysteine concentrations attained in human cerebrospinal fluid. Neurotherapeutics 13:217–225. https://doi.org/10.1007/s13311-015-0404-4
Shao Y, Wang C, Hong Z et al (2016) Inhibition of p38 mitogen-activated protein kinase signaling reduces multidrug transporter activity and anti-epileptic drug resistance in refractory epileptic rats. J Neurochem 136:1096–1105. https://doi.org/10.1111/jnc.13498
Sharp CD, Hines I, Houghton J et al (2003) Glutamate causes a loss in human cerebral endothelial barrier integrity through activation of NMDA receptor. Am J Physiol Heart Circ Physiol 285:H2592-2598. https://doi.org/10.1152/ajpheart.00520.2003
Sharp CD, Houghton J, Elrod JW et al (2005) N-methyl-D-aspartate receptor activation in human cerebral endothelium promotes intracellular oxidant stress. Am J Physiol Heart Circ Physiol 288:H1893-1899. https://doi.org/10.1152/ajpheart.01110.2003
Silva R, Vilas-Boas V, Carmo H et al (2015) Modulation of P-glycoprotein efflux pump: induction and activation as a therapeutic strategy. Pharmacol Ther 149:1–123. https://doi.org/10.1016/j.pharmthera.2014.11.013
Song X, Gong Z, Liu K et al (2020) Baicalin combats glutamate excitotoxicity via protecting glutamine synthetase from ROS-induced 20S proteasomal degradation. Redox Biol 34:101559. https://doi.org/10.1016/j.redox.2020.101559
Spatt J, Bauer G, Baumgartner C et al (2005) Predictors for negative attitudes toward subjects with epilepsy: a representative survey in the general public in Austria. Epilepsia 46:736–742. https://doi.org/10.1111/j.1528-1167.2005.52404.x
Stepien KM, Tomaszewski M, Tomaszewska J et al (2012) The multidrug transporter P-glycoprotein in pharmacoresistance to antiepileptic drugs. Pharmacol Rep 64:1011–1019. https://doi.org/10.1016/s1734-1140(12)70900-3
Summers MA, Moore JL, McAuley JW (2004) Use of verapamil as a potential P-glycoprotein inhibitor in a patient with refractory epilepsy. Ann Pharmacother 38:1631–1634. https://doi.org/10.1345/aph.1E068
Tang F, Hartz AMS, Bauer B (2017) Drug-resistant epilepsy: multiple hypotheses, few answers. Front Neurol 8:301. https://doi.org/10.3389/fneur.2017.00301
Trinka E (2003) Epilepsy: comorbidity in the elderly. Acta Neurol Scand Suppl 180:33–36. https://doi.org/10.1034/j.1600-0404.108.s180.5.x
Trinka E, Bauer G, Oberaigner W et al (2013) Cause-specific mortality among patients with epilepsy: results from a 30-year cohort study. Epilepsia 54:495–501. https://doi.org/10.1111/epi.12014
Wang X, Campos CR, Peart JC et al (2014) Nrf2 upregulates ATP binding cassette transporter expression and activity at the blood-brain and blood-spinal cord barriers. J Neurosci 34:8585–8593. https://doi.org/10.1523/jneurosci.2935-13.2014
Wang X, Huang S, Jiang Y et al (2018) Reactive astrocytes increase the expression of Pgp and Mrp1 via TNFalpha and NFkappaB signaling. Mol Med Rep 17:1198–1204. https://doi.org/10.3892/mmr.2017.7939
Weiss J, Kerpen CJ, Lindenmaier H et al (2003) Interaction of antiepileptic drugs with human P-glycoprotein in vitro. J Pharmacol Exp Ther 307:262–267. https://doi.org/10.1124/jpet.103.054197
Yu N, Di Q, Liu H et al (2011) Nuclear factor-kappa B activity regulates brain expression of P-glycoprotein in the kainic acid-induced seizure rats. Mediators Inflamm 2011:670613. https://doi.org/10.1155/2011/670613
Yu N, Liu H, Zhang YF et al (2014) Effects of brain IKKbeta gene silencing by small interfering RNA on P-glycoprotein expression and brain damage in the rat kainic acid-induced seizure model. CNS Neurol Disord Drug Targets 13:661–672. https://doi.org/10.2174/18715273113129990106
Yu N, Zhang YF, Zhang K et al (2017) Pregnane X receptor not nuclear factor-kappa B up-regulates P-glycoprotein expression in the brain of chronic epileptic rats induced by kainic acid. Neurochem Res 42:2167–2177. https://doi.org/10.1007/s11064-017-2224-x
Zhang C, Kwan P, Zuo Z et al (2010) In vitro concentration dependent transport of phenytoin and phenobarbital, but not ethosuximide, by human P-glycoprotein. Life Sci 86:899–905. https://doi.org/10.1016/j.lfs.2010.04.008
Acknowledgements
The authors are grateful to The Second Affiliated Hospital of Chongqing Medical University and Chongqing Key Laboratory of Neurology for providing facilities for this work.
Funding
This research was supported by the National Natural Science Foundation of China (82071458).
Author information
Authors and Affiliations
Contributions
Qiankun Liu: Performing the experiments; methodology; writing, original draft; writing, review and editing. You Wang: Ideas; writing, original draft; writing, review and editing; supervision. Dandan Tan: Methodology, performing the experiments. Yong Liu: Ideas, methodology, writing—original draft. Peng Zhang: Data curation, methodology. Limin Ma: Analyze or synthesize study data, writing—review and editing. Minxue Liang: Analyze or synthesize study data, performing the experiments. Yangmei Chen: Ideas, resources, writing—review and editing, Funding acquisition, project administration. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics Approval
All animal experiments complied with the National Institutes of Health Guide for the Care and Use of Laboratory Animals and the rules of the Animal Ethical Committee of Chongqing Medical University.
Competing Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, Q., Wang, Y., Tan, D. et al. The Prevention and Reversal of a Phenytoin-Resistant Model by N-acetylcysteine Therapy Involves the Nrf2/P-Glycoprotein Pathway at the Blood–Brain Barrier. J Mol Neurosci 72, 2125–2135 (2022). https://doi.org/10.1007/s12031-022-02056-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-022-02056-0