Abstract
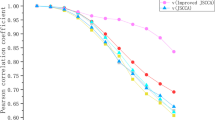
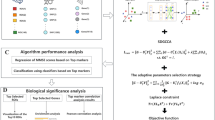
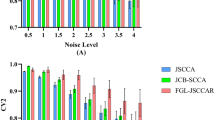
Image genetics mainly explores the pathogenesis of Alzheimer’s disease (AD) by studying the relationship between genetic data (such as SNP, gene expression data, and DNA methylation) and imaging data (such as structural MRI (sMRI), fMRI, and PET). Most of the existing research on brain imaging genomics uses two-way or three-way bi-multivariate methods to explore the correlation analysis between genes and brain imaging. However, many of these methods are still affected by the gradient domination or cannot take into account the effect of feature redundancy on the results, so that the typical correlation coefficient and program running speed are not significantly improved. In order to solve the above problems, this paper proposes a multi-constrained uncertainty-aware adaptive sparse multi-view canonical correlation analysis method (MC-unAdaSMCCA) to explore associations among SNPs, gene expression data, and sMRI; that is, based on traditional unAdaSMCCA, orthogonal constraints are imposed on the weights of the three data features through linear programming, which can reduce the redundancy of feature weights to improve the correlation between the data and reduce the complexity of the algorithm to significantly speed up the running speed of the program. Three adaptive sparse multi-view canonical correlation analysis methods are used as benchmarks to evaluate the difference between real neuroimaging data and synthetic data. Compared with the other three methods, our proposed method has obtained better or comparable typical correlation coefficients and typical weights. Moreover, the following experimental results show that the MC-unAdaSMCCA method cannot only identify biomarkers related to AD and mild cognitive impairment (MCI), but also has a strong ability to resist noise and process high-dimensional data. Therefore, our proposed method provides a reliable approach to multi-modal imaging genetic researches.

















Similar content being viewed by others
Availability of Data and Materials
The authors appreciate the Alzheimer’s Disease Neuroimaging Initiative (ADNI) for contributing data (SNPs, gene expression data. and sMRI).
References
Au R, Piers RJ, Lancashire L (2015) Back to the future: Alzheimer’s disease heterogeneity revisited. Alzheimers Dement (amst) 1(3):368–370. https://doi.org/10.1016/j.dadm.2015.05.006
Azorsa DO, Robeson RH, Frost D et al (2010) High-content siRNA screening of the kinome identifies kinases involved in Alzheimer’s disease-related tau hyperphosphorylation. BMC Genomics 11:25. https://doi.org/10.1186/1471-2164-11-25 (Published 2010 Jan 12)
Cai C, Huang C, Yang C et al (2020) Altered patterns of phase position connectivity in default mode subnetwork of subjective cognitive decline and amnestic mild cognitive impairment. Front Neurosci 14:185. https://doi.org/10.3389/fnins.2020.00185 (Published 2020 Mar 20)
Catricalà E, Polito C, Presotto L et al (2020) Neural correlates of naming errors across different neurodegenerative diseases: an FDG-PET study. Neurology 95(20):e2816–e2830. https://doi.org/10.1212/WNL.0000000000010967
Chanda K, Mukhopadhyay D (2020) LncRNA Xist X-chromosome instability and Alzheimer’s Ddisease. Curr Alzheimer Res 17(6):499–507. https://doi.org/10.2174/1567205017666200807185624
Chen J, Bushman FD, Lewis JD, Wu GD, Li H (2013a) Structure-constrained sparse canonical correlation analysis with an application to microbiome data analysis. Biostatistics 14(2):244–258. https://doi.org/10.1093/biostatistics/kxs038
Chen M, Gao C, Ren Z, Zhou HH (2013b) Sparse CCA via precision adjusted iterative thresholding. arXiv preprint arXiv:1311.6186
Davis EJ, Broestl L, Abdulai-Saiku S et al (2020) A second X chromosome contributes to resilience in a mouse model of Alzheimer’s disease. Sci Transl Med 12(558):eaaz5677. https://doi.org/10.1126/scitranslmed.aaz5677
de Jong LW, van der Hiele K, Veer IM et al (2008) Strongly reduced volumes of putamen and thalamus in Alzheimer’s disease: an MRI study. Brain 131(Pt 12):3277–3285. https://doi.org/10.1093/brain/awn278
De Roeck A, Van Broeckhoven C, Sleegers K (2019) The role of ABCA7 in Alzheimer’s disease: evidence from genomics, transcriptomics and methylomics. Acta Neuropathol 138(2):201–220. https://doi.org/10.1007/s00401-019-01994-1
Deng J, Zeng W, Kong W, Shi Y, Mou X, Guo J (2020) Multi-constrained joint non-negative matrix factorization with application to imaging genomic study of lung metastasis in soft tissue sarcomas. IEEE Trans Biomed Eng 67(7):2110–2118. https://doi.org/10.1109/TBME.2019.2954989
Du L, Huang H, Yan J et al (2016) Structured sparse canonical correlation analysis for brain imaging genetics: an improved GraphNet method. Bioinformatics 32(10):1544–1551. https://doi.org/10.1093/bioinformatics/btw033
Du L, Liu K, Yao X et al (2020a) Detecting genetic associations with brain imaging phenotypes in Alzheimer’s disease via a novel structured SCCA approach. Med Image Anal 61:101656. https://doi.org/10.1016/j.media.2020.101656
Du L, Liu F, Liu K et al (2020b) Identifying diagnosis-specific genotype-phenotype associations via joint multitask sparse canonical correlation analysis and classification. Bioinformatics 36(Suppl_1):i371-i379. https://doi.org/10.1093/bioinformatics/btaa434
Du L, Liu F, Liu K et al (2020c) Associating multi-modal brain imaging phenotypes and genetic risk factors via a dirty multi-task learning method. IEEE Trans Med Imaging 39(11):3416–3428. https://doi.org/10.1109/TMI.2020.2995510
Du L, Liu K, Zhang T et al (2018) A novel SCCA approach via truncated ℓ1-norm and truncated group lasso for brain imaging genetics. Bioinformatics 34(2):278–285. https://doi.org/10.1093/bioinformatics/btx594
Du L, Zhang J, Liu F et al (2021) Identifying associations among genomic, proteomic and imaging biomarkers via adaptive sparse multi-view canonical correlation analysis [published correction appears in Med Image Anal 2021 May 16;72:102081]. Med Image Anal 70:102003. https://doi.org/10.1016/j.media.2021.102003
Galton CJ, Patterson K, Graham K et al (2001) Differing patterns of temporal atrophy in Alzheimer’s disease and semantic dementia. Neurology 57(2):216–225. https://doi.org/10.1212/wnl.57.2.216
Glahn DC, Thompson PM, Blangero J (2007) Neuroimaging endophenotypes: strategies for finding genes influencing brain structure and function. Hum Brain Mapp 28(6):488–501. https://doi.org/10.1002/hbm.20401
Gorski J, Pfeuffer F, Klamroth K (2007) Biconvex sets and optimization with biconvex functions: a survey and extensions. Math Methods Oper Res 66(3):373–407
Guo Z, Liu X, Hou H, Wei F, Liu J, Chen X (2016a) Abnormal degree centrality in Alzheimer’s disease patients with depression: a resting-state functional magnetic resonance imaging study. Exp Gerontol 79:61–66. https://doi.org/10.1016/j.exger.2016.03.017
Guo Z, Liu X, Hou H et al (2016b) (1)H-MRS asymmetry changes in the anterior and posterior cingulate gyrus in patients with mild cognitive impairment and mild Alzheimer’s disease. Compr Psychiatry 69:179–185. https://doi.org/10.1016/j.comppsych.2016.06.001
Hao X, Li C, Du L et al (2017) Mining outcome-relevant brain imaging genetic associations via three-way sparse canonical correlation analysis in Alzheimer’s disease. Sci Rep 7:44272. https://doi.org/10.1038/srep44272 (Published 2017 Mar 14)
Harris SE, Fox H, Wright AF et al (2007) A genetic association analysis of cognitive ability and cognitive ageing using 325 markers for 109 genes associated with oxidative stress or cognition. BMC Genet 8:43. https://doi.org/10.1186/1471-2156-8-43 (Published 2007 Jul 2)
Hotokezaka Y, Katayama I, van Leyen K, Nakamura T (2015) GSK-3β-dependent downregulation of γ-taxilin and αNAC merge to regulate ER stress responses. Cell Death Dis 6(4):e1719. https://doi.org/10.1038/cddis.2015.90 (Published 2015 Apr 16)
Hu W, Lin D, Cao S et al (2018) Adaptive sparse multiple canonical correlation analysis with application to imaging (epi)genomics study of schizophrenia. IEEE Trans Biomed Eng 65(2):390–399. https://doi.org/10.1109/TBME.2017.2771483
Ilic K, Mlinac-Jerkovic K, Jovanov-Milosevic N et al (2019) Hippocampal expression of cell-adhesion glycoprotein neuroplastin is altered in Alzheimer’s disease. J Cell Mol Med 23(2):1602–1607. https://doi.org/10.1111/jcmm.13998
Johanson CE, Duncan JA 3rd, Klinge PM, Brinker T, Stopa EG, Silverberg GD (2008) Multiplicity of cerebrospinal fluid functions: new challenges in health and disease. Cerebrospinal Fluid Res 5:10. https://doi.org/10.1186/1743-8454-5-10 (Published 2008 May 14)
Kendall A, Gal Y, Cipolla R (2018) Multi-task learning using uncertainty to weigh losses for scene geometry and semantics. In: Proceedings of the IEEE conference on computer vision and pattern recognition, p 7482–7491
Kuchtova B, Wurst Z, Mrzilkova J et al (2018) Compensatory shift of subcallosal area and paraterminal gyrus white matter parameters on DTI in patients with Alzheimer disease. Curr Alzheimer Res 15(6):590–599. https://doi.org/10.2174/1567205015666171227155510
Leyton CE, Landin-Romero R, Liang CT et al (2019) Correlates of anomia in non-semantic variants of primary progressive aphasia converge over time. Cortex 120:201–211. https://doi.org/10.1016/j.cortex.2019.06.008
Lin CH, Lin E, Lane HY (2017) Genetic biomarkers on age-related cognitive decline. Front Psychiatry 8:247. https://doi.org/10.3389/fpsyt.2017.00247 (Published 2017 Nov 21)
Luxenberg JS, Haxby JV, Creasey H, Sundaram M, Rapoport SI (1987) Rate of ventricular enlargement in dementia of the Alzheimer type correlates with rate of neuropsychological deterioration. Neurology 37(7):1135–1140. https://doi.org/10.1212/wnl.37.7.1135
Mu Y, Li Y, Zhang Q et al (2020) Amplitude of low-frequency fluctuations on Alzheimer’s disease with depression: evidence from resting-state fMRI. Gen Psychiatr 33(4):e100147. https://doi.org/10.1136/gpsych-2019-100147 (Published 2020 Jul 9)
Muñoz-Neira C, Tedde A, Coulthard E, Thai NJ, Pennington C (2019) Neural correlates of altered insight in frontotemporal dementia: a systematic review. Neuroimage Clin 24:102066. https://doi.org/10.1016/j.nicl.2019.102066
Novikova G, Kapoor M, Tcw J et al (2021) Integration of Alzheimer’s disease genetics and myeloid genomics identifies disease risk regulatory elements and genes. Nat Commun 12(1):1610. https://doi.org/10.1038/s41467-021-21823-y (Published 2021 Mar 12)
Parkhomenko E, Tritchler D, Beyene J (2009) Sparse canonical correlation analysis with application to genomic data integration. Stat Appl Genet Mol Biol 8:1. https://doi.org/10.2202/1544-6115.1406
Penniello MJ, Lambert J, Eustache F et al (1995) A PET study of the functional neuroanatomy of writing impairment in Alzheimer’s disease The role of the left supramarginal and left angular gyri. Brain 118(Pt 3):697–706. https://doi.org/10.1093/brain/118.3.697
Purcell S, Neale B, Todd-Brown K et al (2007) PLINK: a tool set for whole-genome association and population-based linkage analyses. Am J Hum Genet 81(3):559–575. https://doi.org/10.1086/519795
Qadar MA, Seghouane AK (2019) A projection CCA method for effective fMRI data analysis. IEEE Trans Biomed Eng 66(11):3247–3256. https://doi.org/10.1109/TBME.2019.2902876
Rana P, Franco EF, Rao Y et al (2019) Evaluation of the common molecular basis in Alzheimer’s and Parkinson’s diseases. Int J Mol Sci 20(15):3730. https://doi.org/10.3390/ijms20153730 (Published 2019 Jul 30)
Ritchie ME, Phipson B, Wu D et al (2015) limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res 43(7):e47. https://doi.org/10.1093/nar/gkv007
Ruan Z, Delpech JC, Venkatesan Kalavai S et al (2020) P2RX7 inhibitor suppresses exosome secretion and disease phenotype in P301S tau transgenic mice. Mol Neurodegener 15(1):47. https://doi.org/10.1186/s13024-020-00396-2 (Published 2020 Aug 18)
Ryan NS, Shakespeare TJ, Lehmann M et al (2014) Motor features in posterior cortical atrophy and their imaging correlates. Neurobiol Aging 35(12):2845–2857. https://doi.org/10.1016/j.neurobiolaging.2014.05.028
Saykin AJ, Shen L, Foroud TM et al (2010) Alzheimer’s Disease Neuroimaging Initiative biomarkers as quantitative phenotypes: genetics core aims progress and plans. Alzheimers Dement 6(3):265–273. https://doi.org/10.1016/j.jalz.2010.03.013
Sekar S, McDonald J, Cuyugan L et al (2015) Alzheimer’s disease is associated with altered expression of genes involved in immune response and mitochondrial processes in astrocytes. Neurobiol Aging 36(2):583–591. https://doi.org/10.1016/j.neurobiolaging.2014.09.027
Shen L, Thompson PM, Potkin SG et al (2014) Genetic analysis of quantitative phenotypes in AD and MCI: imaging, cognition and biomarkers. Brain Imaging Behav 8(2):183–207. https://doi.org/10.1007/s11682-013-9262-z
Shinohara M, Tachibana M, Kanekiyo T, Bu G (2017) Role of LRP1 in the pathogenesis of Alzheimer’s disease: evidence from clinical and preclinical studies. J Lipid Res 58(7):1267–1281. https://doi.org/10.1194/jlr.R075796
Silver M, Montana G, Alzheimer’s Disease Neuroimaging Initiative, (2012) Fast identification of biological pathways associated with a quantitative trait using group lasso with overlaps. Stat Appl Genet Mol Biol 11(1):Article-7. https://doi.org/10.2202/1544-6115.1755 (Published 2012 Jan 6)
Suga K, Saito A, Mishima T, Akagawa K (2015) ER and Golgi stresses increase ER-Golgi SNARE Syntaxin5: implications for organelle stress and βAPP processing. Neurosci Lett 604:30–35. https://doi.org/10.1016/j.neulet.2015.07.017
Tuschl K, Meyer E, Valdivia LE et al (2016) Mutations in SLC39A14 disrupt manganese homeostasis and cause childhood-onset parkinsonism-dystonia. Nat Commun 7:11601. https://doi.org/10.1038/ncomms11601 (Published 2016 May 27)
Wang K, Li M, Hakonarson H (2010) ANNOVAR: functional annotation of genetic variants from high-throughput sequencing data. Nucleic Acids Res 38(16):e164. https://doi.org/10.1093/nar/gkq603
Wei K, Kong W, Wang S (2021) An improved multi-task sparse canonical correlation analysis of imaging genetics for detecting biomarkers of Alzheimer’s disease. IEEE Access 9:30528–30538
Weiner MW, Aisen PS, Jack CR Jr et al (2010) The Alzheimer’s Disease Neuroimaging Initiative: progress report and future plans. Alzheimers Dement 6(3):202–211. https://doi.org/10.1016/j.jalz.2010.03.007
Witten DM, Tibshirani RJ (2009) Extensions of sparse canonical correlation analysis with applications to genomic data. Stat Appl Genet Mol Biol 8(1):Article28. https://doi.org/10.2202/1544-6115.1470
Witten DM, Tibshirani R, Hastie T (2009) A penalized matrix decomposition, with applications to sparse principal components and canonical correlation analysis. Biostatistics 10(3):515–534. https://doi.org/10.1093/biostatistics/kxp008
Xu S, Duan P, Li J et al (2016) Zinc finger and X-linked factor (ZFX) binds to human SET transcript 2 promoter and transactivates SET expression. Int J Mol Sci 17(10):1737. https://doi.org/10.3390/ijms17101737 (Published 2016 Oct 20)
Yan J, Du L, Kim S et al (2014) Transcriptome-guided amyloid imaging genetic analysis via a novel structured sparse learning algorithm. Bioinformatics 30(17):i564–i571. https://doi.org/10.1093/bioinformatics/btu465
Yan J, Risacher SL, Nho K, Saykin AJ, Shen LI (2017) Identification of discriminative imaging proteomics associations in Alzheimer’s disease via a novel sparse correlation model. Pac Symp Biocomput 22:94–104. https://doi.org/10.1142/9789813207813_0010
Yan S, Zheng C, Cui B et al (2020) Multiparametric imaging hippocampal neurodegeneration and functional connectivity with simultaneous PET/MRI in Alzheimer’s disease. Eur J Nucl Med Mol Imaging 47(10):2440–2452. https://doi.org/10.1007/s00259-020-04752-8
Yang J, Pan P, Song W et al (2012) Voxelwise meta-analysis of gray matter anomalies in Alzheimer’s disease and mild cognitive impairment using anatomic likelihood estimation. J Neurol Sci 316(1–2):21–29. https://doi.org/10.1016/j.jns.2012.02.010
Yu Y, Li Z, Lin Y et al (2019) Depression affects intrinsic brain activity in patients with mild cognitive impairment. Front Neurosci 13:1333. https://doi.org/10.3389/fnins.2019.01333 (Published 2019 Dec 17)
Zhao Q, Sang X, Metmer H, Swati ZNNK, Lu J, Alzheimer’s Disease NeuroImaging Initiative, (2019) Functional segregation of executive control network and frontoparietal network in Alzheimer’s disease. Cortex 120:36–48. https://doi.org/10.1016/j.cortex.2019.04.026
Zhou M, Zhang F, Zhao L, Qian J, Dong C (2016) Entorhinal cortex: a good biomarker of mild cognitive impairment and mild Alzheimer’s disease. Rev Neurosci 27(2):185–195. https://doi.org/10.1515/revneuro-2015-0019
Zhu XC, Wang HF, Jiang T et al (2017) Effect of CR1 genetic variants on cerebrospinal fluid and neuroimaging biomarkers in healthy mild cognitive impairment and Alzheimer’s disease cohorts. Mol Neurobiol 54(1):551–562. https://doi.org/10.1007/s12035-015-9638-8
Funding
This work was supported in part by the Natural Science Foundation of Shanghai (No. 18ZR1417200) and National Natural Science Foundation of China (No. 61803257).
Author information
Authors and Affiliations
Contributions
Conception and design of the research: Wenbo Wang and Wei Kong. Acquisition, analysis, and interpretation of data: Wenbo Wang, Shuaiqun Wang, and Wei Kong. Statistical analysis: Wenbo Wang. Drafting the manuscript: Wenbo Wang. Manuscript revision for important intellectual content: Wei Kong. All authors have read and approved the manuscript.
Corresponding author
Ethics declarations
Ethics Approval and Consent to Participate
This article does not contain any studies with human participants or animals performed by any of the authors; therefore, the ethical approval and consent to participate are not applicable.
Consent for Publication
The authors have consented to the publication of this article.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Wenbo Wang and Wei Kong should be regarded as joint first authors.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, W., Kong, W., Wang, S. et al. Detecting Biomarkers of Alzheimer’s Disease Based on Multi-constrained Uncertainty-Aware Adaptive Sparse Multi-view Canonical Correlation Analysis. J Mol Neurosci 72, 841–865 (2022). https://doi.org/10.1007/s12031-021-01963-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-021-01963-y