Abstract
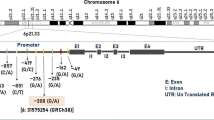
Transient receptor potential vanilloid type 1 (TRPV1) receptors activated by heat and capsaicin are expressed in trigeminal nociceptive neurons and implicated in the generation of migraine pain. Genetic studies suggested that single-nucleotide polymorphism (SNP) 1911A>G (rs8065080), leading to amino acid substitution Ile585Val, in the TRPV1 gene affects functional activity of TRPV1 receptors and is involved in different pain conditions. However, this polymorphism has not been tested in migraine patients. The objective of this pilot study was to investigate genetic factors of migraine susceptibility. We evaluated frequency distribution of AA, AG, and GG variants of SNP 1911A>G in the TRPV1 gene in patients with episodic and chronic migraine compared with healthy individuals. The study included 46 patients diagnosed with migraine (27 episodic and 19 chronic) and 50 healthy individuals as a control group. DNA from peripheral blood was used to test TRPV1 SNP using allele-specific PCR combined with gel electrophoresis. The genotype frequency distribution in episodic migraine was comparable with that in controls (AA 33%, AG 56%, GG 11% and AA 34%, AG 46%, GG 20%, respectively). On the contrary, in chronic migraine, the distribution differed significantly (p < 0.05) (AA 68%, AG 32%, GG 0%). This are first indications for a distinctive genotype frequency distribution of TRPV1 1911A>G in chronic migraine patients compared with episodic migraine patients and controls. Our data confirm a different predisposition to chronic pain in migraine and give a prerequisite for a new look at the nature of chronification of migraine, proposing that the absence of GG genotype may be considered as possible risk biomarker of episodic migraine evolution to chronic form.


Similar content being viewed by others
Data Availability
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
References
Akerman S, Kaube H, Goadsby PJ (2003) Vanilloid type 1 receptors (VR1) on trigeminal sensory nerve fibres play a minor role in neurogenic dural vasodilatation, and are involved in capsaicin-induced dural dilation. Br J Pharmacol 140:718–724. https://doi.org/10.1038/sj.bjp.0705486
Bernardini N, Neuhuber W, Reeh PW, Sauer SK (2004) Morphological evidence for functional capsaicin receptor expression and calcitonin gene-related peptide exocytosis in isolated peripheral nerve axons of the mouse. Neuroscience 126:585–590
Binder A, May D, Baron R, Maier C, Tölle TR, Treede RD, Berthele A, Faltraco F, Flor H, Gierthmühlen J, Haenisch S, Huge V, Magerl W, Maihöfner C, Richter H, Rolke R, Scherens A, Uçeyler N, Ufer M, Wasner G, Zhu J, Cascorbi I (2011) Transient receptor potential channel polymorphisms are associated with the somatosensory function in neuropathic pain patients. PLoS One 6(3):e17387. https://doi.org/10.1371/journal.pone.0017387
Buse DC, Greisman JD, Baigi K, Lipton RB (2019) Migraine progression: a systematic review. An editorial comment. Headache 59(7):974–976. https://doi.org/10.1111/head.13573
Cantero-Recasens G, Gonzalez JR, Fandos C, Duran-Tauleria E, Smit LA, Kauffmann F, Antó JM, Valverde MA (2010) Loss of function of transient receptor potential vanilloid 1 (TRPV1) genetic variant is associated with lower risk of active childhood asthma. J Biol Chem 285(36):27532–27535. https://doi.org/10.1074/jbc.C110.159491
Cardillo G (2007) MyFisher23: a very compact routine for Fisher’s exact test on 2x3 matrix. http://www.mathworks.com/matlabcentral/fileexchange/15399. Accessed 26 September 2019
Carreno O, Corominas R, Fernandez-Morales J et al (2012) SNP variants within the vanilloid TRPV1 and TRPV3 receptor genes are associated with migraine in the Spanish population. Am J Med Genet B Neuropsychiatr Genet 159B(1):94–103
Caterina MJ, Schumacher MA, Tominaga M, Rosen TA, Levine JD, Julius D (1997) The capsaicin receptor: a heat-activated ion channel in the pain pathway. Nature 389:816–824
Chasman DI, Schürks M, Anttila V, de Vries B, Schminke U, Launer LJ, Terwindt GM, van den Maagdenberg AM, Fendrich K, Völzke H, Ernst F, Griffiths LR, Buring JE, Kallela M, Freilinger T, Kubisch C, Ridker PM, Palotie A, Ferrari MD, Hoffmann W, Zee RY, Kurth T (2011) Genome-wide association study reveals three susceptibility loci for common migraine in the general population. Nat Genet 43(7):695–698. https://doi.org/10.1038/ng.856
Chatchaisak D, Srikiatkhachorn A, Grand SM, Govitrapong P, Chetsawang B (2012) The role of calcitonin gene-related peptide on the increase in transient receptor potential vanilloid-1 levels in trigeminal ganglion and trigeminal nucleus caudalis activation of rat. J Chem Neuroanat 47:50–56. https://doi.org/10.1016/j.jchemneu.2012.09.005
Clarkson DB, Fan Y, Joe H (1993) A remark on algorithm 643: FEXACT: an algorithm for performing Fisher’s exact test in rxc contingency tables. ACM Trans Math Softw 19:484–488. https://doi.org/10.1145/168173.168412
Cortright DN, Szallasi A (2009) TRP channels and pain. Curr Pharm Des 15:1739–1749
Edvinsson L, Jansen I, Kingman TA, McCulloch J (1990) Cerebrovascular responses to capsaicin in vitro and in situ. Br J Pharmacol 100:312–318. https://doi.org/10.1111/j.1476-5381.1990.tb15801.x
Ensembl 98. Available from: http://jan2020.archive.ensembl.org/. Accessed 15 January 2020
Forstenpointner J, Förster M, May D, Hofschulte F, Cascorbi I, Wasner G, Gierthmühlen J, Baron R (2007) Short report: TRPV1-polymorphism 1911 A>G alters capsaicin-induced sensory changes in healthy subjects. PLoS One 12(8):e0183322. https://doi.org/10.1371/journal.pone.0183322
Goadsby P, Edvinsson L (1993) The trigeminovascular system and migraine: studies characterizing cerebrovascular and neuropeptide changes seen in humans and cats. Ann Neurol 33:48–56
Hayes P, Meadows HJ, Gunthorpe MJ, Harries MH, Duckworth DM, Cairns W, Harrison DC, Clarke CE, Ellington K, Prinjha RK, Barton AJ, Medhurst AD, Smith GD, Topp S, Murdock P, Sanger GJ, Terrett J, Jenkins O, Benham CD, Randall AD, Gloger IS, Davis JB (2000) Cloning and functional expression of a human orthologue of rat vanilloid receptor-1. Pain 88:205–215
Headache Classification Committee of the International Headache Society (IHS) The international classification of headache disorders, 3rd edition (2018) Cephalalgia 38: 1–211. https://doi.org/10.1177/0333102417738202
Hoffmann J, Wecker S, Neeb L, Dirnagl U, Reuter U (2012) Primary trigeminal afferents are the main source for stimulus-induced CGRP release into jugular vein blood and CSF. Cephalalgia 32:659–667. https://doi.org/10.1177/0333102412447701
Jansen-Olesen I, Mortensen A, Edvinsson L (1996) Calcitonin gene-related peptide is released from capsaicin-sensitive nerve fibres and induces vasodilatation of human cerebral arteries concomitant with activation of adenylyl cyclase. Cephalalgia 6:310–316
Kamshilin A, Volynsky M, Khayrutdinova O, Nurkhametova D, Babayan L, Amelin AV, Mamontov OV, Giniatullin R (2018) Novel capsaicin-induced parameters of microcirculation in migraine patients revealed by imaging photoplethysmography. J Headache Pain 19:43. https://doi.org/10.1186/s10194-018-0872-0
Kara I, Sazci A, Ergul E, Kaya G, Kilic G (2003) Association of the C677T and A1298C polymorphisms in the 5,10 methylenetetrahydrofolate reductase gene in patients with migraine risk. Mol Brain Res 111(1–2):84–90. https://doi.org/10.1016/S0169-328X(02)00672-1
Kim H, Neubert JK, San Miguel A, Xu K, Krishnaraju RK, Iadarola MJ, Goldman D, Dionne RA (2004) Genetic influence on variability in human acute experimental pain sensitivity associated with gender, ethnicity and psychological temperament. Pain. 2004;109:488–496
Kim H, Mittal DP, Iadarola MJ, Dionne RA (2006) Genetic predictors for acute experimental cold and heat pain sensitivity in humans. J Med Genet 43(8):e40
Kirkman TW (1996) Statistics to Use. http://www.physics.csbsju.edu/stats/.
Kroonenberg PM, Verbeek A (2018) The tale of Cochran’s rule: my contingency table has so many expected values smaller than 5, what am I to do? Am Stat 72:2,175–2,183. https://doi.org/10.1080/00031305.2017.1286260
van den Maagdenberg AMJM, Nyholt DR, Anttila V (2019) Novel hypotheses emerging from GWAS in migraine? J Headache Pain 20(1):5. https://doi.org/10.1186/s10194-018-0956-x
McDonald JH (2014) Handbook of biological statistics, 3rd edn. Sparky House Publishing, Baltimore, Maryland
Meents JE, Neeb L, Reuter U (2012) TRPV1 in migraine pathophysiology. Trends Mol Med 16:153–159. https://doi.org/10.1016/j.molmed.2010.02.004
Nicoletti P, Trevisani M, Manconi M, Gatti R, De Siena G, Zagli G, Benemei S, Capone JA, Geppetti P, Pini LA (2008) Ethanol causes neurogenic vasodilation by TRPV1 activation and CGRP release in the trigeminovascular system of the guinea pig. Cephalalgia 28:9–17
Okamoto N, Okumura M, Tadokoro O, Sogawa N, Tomida M, Kondo E (2018) Effect of single nucleotide polymorphisms in TRPV1 on burning pain and capsaicin sensitivity in Japanese adults. Mol Pain 14:1–8. https://doi.org/10.1177/1744806918804439
Özge A, Uluduz D, Selekler M, Öztürk M, Baykan B, Çınar N, Domaç FM, Zarifoğlu M, Inan LE, Akyol A, Bolay H, Uzuner GT, Erdemoğlu AK, Oksuz N, Temel GO (2015) Chronic migraine in older adults. Geriatr Gerontol Int 15:652–658. https://doi.org/10.1111/ggi.12314
Piane M, Lulli P, Farinelli I, Simeoni S, De Filippis S, Patacchioli FR, Marteletti P (2007) Genetics of migraine and pharmacogenomics: some considerations. J Headache Pain 8(6):334–339. https://doi.org/10.1007/s10194-007-0427-2
Stucky CL, Dubin AE, Jeske NA, Malin SA, McKemy DD, Story GM (2009) Roles of transient receptor potential channels in pain. Brain Res Rev 60:2–23. https://doi.org/10.1016/j.brainresrev.2008
Szallasi A, Blumberg PM (1999) Vanilloid (capsaicin) receptors and mechanisms. Pharmacol Rev 51(2):159–212
Tominaga M, Caterina MJ (2004) Thermosensation and pain. J Neurobiol 61:3–12
Tominaga M, Caterina MJ, Malmberg AB, Rosen TA, Gilbert H, Skinner K, Raumann BE, Basbaum AI, Julius D (1998) The cloned capsaicin receptor integrates multiple pain producing stimuli. Neuron 21:531–543
Wang S, Joseph J, Diatchenko L, Ro JY, Chung MK (2016) Agonist-dependence of functional properties for common nonsynonymous variants of human transient receptor potential vanilloid 1. Pain 157(7):1515–1524. https://doi.org/10.1097/j.pain.0000000000000556
World Health Organization, Lifting The Burden (2011) Atlas of headache disorders and resources in the world 2011. WHO, Geneva
Zakharov AV, Vitale K, Kilinc E, Koroleva K, Fayuk D, Shelukhina I, Naumenko N, Skorinkin A, Khazipov R, Giniatullin R (2015) Hunting for origins of migraine pain: cluster analysis of spontaneous and capsaicin-induced firing in meningeal trigeminal nerve fibers. Front Cell Neurosci 9:287. https://doi.org/10.3389/fncel.2015.00287
Acknowledgments
We thank Prof. Arn M. J. M. van den Maagdenberg for the manuscript reading and providing valuable advice.
Code Availability
Not applicable.
Funding
The study was supported by the Russian Science Foundation (grant 15–15-20012) in the part of study design, organization, and carrying out the experiments. A.R. was supported by state assignments 20.5175.2017/6.7 and 17.9783.2017/8.9 of the Ministry of Science and Higher Education of Russian Federation in the part of carrying out theoretical work. JT was supported by the Academy of Finland (grant 316258 to J.T.) in the part of carrying out theoretical work. This study was supported by the Russian Government Program of Competitive Growth of Kazan Federal University and ITMO University.
Author information
Authors and Affiliations
Contributions
A.Y. performed genotyping experiments, analyzed data, and wrote the manuscript. Y.D. designed and performed the experiment and represented it in the manuscript. J.T. performed statistical analysis of data and represented it in the manuscript. A.Y., O.K., I.K., and D.N. contributed to patients’ enrolment and sample collection. A.K. and R.G. conceived the idea of the study. R.G. and A.R. discussed the results and revised the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethics Approval
The study was conducted in Kazan in accordance with ethical standards presented in the Declaration of Helsinki. The Ethics Committees of the Kazan State Medical University prior the research approved the protocol of this study (permission protocol number 7 from 25.09.2018).
Consent to Participate
Informed consent was obtained from all individual participants included in the study.
Consent for Publication
Not applicable.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yakubova, A., Davidyuk, Y., Tohka, J. et al. Searching for Predictors of Migraine Chronification: a Pilot Study of 1911A>G Polymorphism of TRPV1 Gene in Episodic Versus Chronic Migraine. J Mol Neurosci 71, 618–624 (2021). https://doi.org/10.1007/s12031-020-01683-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-020-01683-9