Abstract
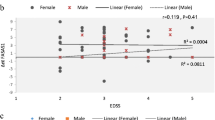
Recent studies have shown contribution of long non-coding RNAs (lncRNAs) in the pathogenesis of immune-related disorders including multiple sclerosis (MS). Based on the role of these transcripts in the regulation of immune response, peripheral levels of lncRNAs can reflect the level of immune activation. In the present study, we quantified expression of four lncRNAs namely SPRY4-IT1, HOXA-AS2, LINC-ROR, and MEG3 in venous blood of MS patients and controls using quantitative real-time PCR method. Relative expressions of SPRY4-IT1, HOXA-AS2, LINC-ROR, and MEG3 were significantly lower in female MS patients compared with female healthy subjects. For MEG3, this pattern of expression was also observed in male subjects. However, for other lncRNAs, no significant difference was detected between male patients and male controls. Expression of HOXA-AS2 was correlated with progression index (r = 0.36, P < 0.001). Besides, there was a significant correlation between expression of this lncRNA and expression of LINC-ROR in MS patients (r = 0.44, P < 0.0001). There was no other correlation between expression of lncRNAs and clinical data in MS patients. In control group, expressions of none of lncRNAs were correlated with age of persons. Notably, significant correlations were demonstrated between expression levels of all lncRNAs in healthy subjects with r values ranging from 0.23 to 0.42. The current investigation shows dysregulation of lncRNAs in MS patients in a sex-specific manner and warrants further studies to unravel the clinical and therapeutic implications of such dysregulation.






Similar content being viewed by others
References
Ahmed W, Liu ZF (2018) Long non-coding RNAs: novel players in regulation of immune response upon herpesvirus infection. Front Immunol 9:761
Cao S, Lin L, Xia X, Wu H (2019) lncRNA SPRY4-IT1 regulates cell proliferation and migration by sponging miR-101-3p and regulating AMPK expression in gastric cancer. Mol Ther Nucleic Acids 17:455–464
Chen J, Zhu J, Wang Z, Yao X, Wu X, Liu F, Zheng W, Li Z, Lin A (2017) MicroRNAs correlate with multiple sclerosis and neuromyelitis optica spectrum disorder in a Chinese population. Med Sci Monit 23:2565–2583
Cox MB, Cairns MJ, Gandhi KS, Carroll AP, Moscovis S, Stewart GJ, Broadley S, Scott RJ, Booth DR, Lechner-Scott J (2010) MicroRNAs miR-17 and miR-20a inhibit T cell activation genes and are under-expressed in MS whole blood. PLoS One 5:e12132
DE Santis G, Ferracin M, Biondani A, Caniatti L, Rosaria Tola M, Castellazzi M, Zagatti B, Battistini L, Borsellino G, Fainardi E, Gavioli R, Negrini M, Furlan R, Granieri E (2010) Altered miRNA expression in T regulatory cells in course of multiple sclerosis. J Neuroimmunol 226:165–171
Denaro N, Merlano MC, Lo Nigro C (2019) Long noncoding RNAs as regulators of cancer immunity. Mol Oncol 13:61–73
Fenoglio C, Oldoni E, Serpente M, DE Riz MA, Arcaro M, D'anca M, Pietroboni AM, Calvi A, Lecchi E, Goris A, Mallants K, Dubois B, Comi C, Cantello R, Scarpini E, Galimberti D (2018) LncRNAs expression profile in peripheral blood mononuclear cells from multiple sclerosis patients. J Neuroimmunol 324:129–135
Ghafouri-Fard S, Taheri M (2019) Maternally expressed gene 3 (MEG3): a tumor suppressor long non coding RNA. Biomed Pharmacother 118:109129
Ghafouri-Fard S, Taheri M (2020) A comprehensive review of non-coding RNAs functions in multiple sclerosis. Eur J Pharmacol 879:173127
Harbo HF, Gold R, Tintore M (2013) Sex and gender issues in multiple sclerosis. Ther Adv Neurol Disord 6:237–248
Hou L, Tu J, Cheng F, Yang H, Yu F, Wang M, Liu J, Fan J, Zhou G (2018) Long noncoding RNA ROR promotes breast cancer by regulating the TGF-beta pathway. Cancer Cell Int 18:142
Jia H-Y, Zhang K, Lu W-J, Xu G-W, Zhang J-F, Tang Z-L (2019) LncRNA MEG3 influences the proliferation and apoptosis of psoriasis epidermal cells by targeting miR-21/caspase-8. BMC molecular and cell biology 20:46–46
Jiang L, Wu Z, Meng X, Chu X, Huang H, Xu C (2019) LncRNA HOXA-AS2 facilitates tumorigenesis and progression of papillary thyroid cancer by modulating the miR-15a-5p/HOXA3 axis. Hum Gene Ther 30:618–631
Lee PW, Severin ME, Lovett-Racke AE (2017) TGF-beta regulation of encephalitogenic and regulatory T cells in multiple sclerosis. Eur J Immunol 47:446–453
Li J, Chen Y, Chen Z, He A, Xie H, Zhang Q, Cai Z, Liu Y, Huang W (2017) SPRY4-IT1: a novel oncogenic long non-coding RNA in human cancers. Tumour Biol 39:1010428317711406
Li R, Fang L, Pu Q, Bu H, Zhu P, Chen Z, Yu M, Li X, Weiland T, Bansal A, Ye SQ, Wei Y, Jiang J, Wu M (2018) MEG3-4 is a miRNA decoy that regulates IL-1beta abundance to initiate and then limit inflammation to prevent sepsis during lung infection. Sci Signal 11
Lin CC, Edelson BT (2017) New insights into the role of IL-1beta in experimental autoimmune encephalomyelitis and multiple sclerosis. J Immunol 198:4553–4560
Liu YR, Yang L, Xu QQ, Lu XY, Ma TT, Huang C, Li J (2019) Long noncoding RNA MEG3 regulates rheumatoid arthritis by targeting NLRC5. J Cell Physiol 234:14270–14284
Lorenzi JC, Brum DG, Zanette DL, De Paula Alves Souza A, Barbuzano FG, Dos Santos AC, Barreira AA, Da Silva WA (2012) miR-15a and 16-1 are downregulated in CD4+ T cells of multiple sclerosis relapsing patients. Int J Neurosci 122:466–471
Mahad DJ, Ransohoff RM (2003) The role of MCP-1 (CCL2) and CCR2 in multiple sclerosis and experimental autoimmune encephalomyelitis (EAE). Semin Immunol 15:23–32
Meares GP, Qin H, Liu Y, Holdbrooks AT, Benveniste EN (2013) AMP-activated protein kinase restricts IFN-gamma signaling. J Immunol 190:372–380
Paintlia AS, Paintlia MK, Mohan S, Singh AK, Singh I (2013) AMP-activated protein kinase signaling protects oligodendrocytes that restore central nervous system functions in an experimental autoimmune encephalomyelitis model. Am J Pathol 183:526–541
Quintana E, Ortega FJ, Robles-Cedeno R, Villar ML, Buxo M, Mercader JM, Alvarez-Cermeno JC, Pueyo N, Perkal H, Fernandez-Real JM, Ramio-Torrenta L (2017) miRNAs in cerebrospinal fluid identify patients with MS and specifically those with lipid-specific oligoclonal IgM bands. Mult Scler J 23:1716–1726
Santos AS, Neto EC, Fukui RT, Ferreira LRP, Silva MER (2019) Increased expression of circulating microRNA 101-3p in type 1 diabetes patients: new insights into miRNA-regulated pathophysiological pathways for type 1 diabetes. Frontiers in Immunology 10
Shaker OG, Mahmoud RH, Abdelaleem OO, Ibrahem EG, Mohamed AA, Zaki OM, Abdelghaffar NK, Ahmed TI, Hemeda NF, Ahmed NA, Mansour DF (2019) LncRNAs, MALAT1 and lnc-DC as potential biomarkers for multiple sclerosis diagnosis. Biosci Rep 39
Shull MM, Ormsby I, Kier AB, Pawlowski S, Diebold RJ, Yin M, Allen R, Sidman C, Proetzel G, Calvin D et al (1992) Targeted disruption of the mouse transforming growth factor-beta 1 gene results in multifocal inflammatory disease. Nature 359:693–699
Sukma Dewi I, Hollander Z, Lam KK, Mcmanus JW, Tebbutt SJ, Ng RT, Keown PA, Mcmaster RW, Mcmanus BM, Gidlof O, Ohman J (2017) Association of serum MiR-142-3p and MiR-101-3p levels with acute cellular rejection after heart transplantation. PLoS One 12:e0170842
Wang Y, Zhang R, Cheng G, Xu R, Han X (2018) Long non-coding RNA HOXA-AS2 promotes migration and invasion by acting as a ceRNA of miR-520c-3p in osteosarcoma cells. Cell Cycle 17:1637–1648
Yang X, Wu Y, Zhang B, Ni B (2018) Noncoding RNAs in multiple sclerosis. Clin Epigenetics 10:149
Zampetaki A, Albrecht A, Steinhofel K (2018) Long non-coding RNA structure and function: is there a link? Front Physiol 9:1201
Zhang Y, Li X-J, He R-Q, Wang X, Zhang T-T, Qin Y, Zhang R, Deng Y, Wang H-L, Luo D-Z (2018) Upregulation of HOXA1 promotes tumorigenesis and development of non-small cell lung cancer: a comprehensive investigation based on reverse transcription-quantitative polymerase chain reaction and bioinformatics analysis. Int J Oncol 53:73–86
Funding
The current study was supported by a grant from Shahid Beheshti University of Medical Sciences.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The study protocol was approved by the Ethical Committee of Shahid Beheshti University of Medical Sciences. Written informed consents were obtained from all participants.
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Safa, A., Taheri, M., Fallah, H. et al. Downregulation of Cancer-Associated lncRNAs in Peripheral Blood of Multiple Sclerosis Patients. J Mol Neurosci 70, 1533–1540 (2020). https://doi.org/10.1007/s12031-020-01646-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-020-01646-0