Abstract
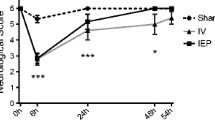
Stroke is the second leading reason for death worldwide and is one of the fundamental causes of long-term disabilities. The aim of this investigation was to assess the impact of combined administration progesterone (PROG) and melatonin (MEL) on stroke complications. Male Wistar rats (9–10 weeks) weighing 250–300 g were used as a part of this examination. They were randomly separated into eight groups (nine rats for every group). Common carotid arteries on the two sides clamped (BCCAO model) with non-traumatic clips for 20 min. At that point, the rats were treated with 8 mg/kg PROG, 10 mg/kg MEL, and vehicles (sesamoid and normal saline). Morris water maze testing was performed following 2 weeks. At that point, the rats were euthanized, and histological examination was directed. The outcome demonstrated that utilization of PROG and MEL in treatment groups essentially increases the quantity of pyramidal cells and enhances spatial memory compared to non-treatment groups (p < 0.05). Moreover, the neuroleptic factor gene expression and protein concentration were significantly enhanced in the treated groups (p < 0.05). As indicated by the outcomes, co-administration of PROG and MEL can enhance learning and memory by surviving the pyramidal neurons and diminishing neural death by means of increasing neuroleptic factors in the hippocampal CA1 zone.




Similar content being viewed by others
References
Chen TY, Lee MY, Chen HY, Kuo YL, Lin SC, Wu TS, Lee E (2006) Melatonin attenuates the postischemic increase in blood–brain barrier permeability and decreases hemorrhagic transformation of tissue-plasminogen activator therapy following ischemic stroke in mice. J Pineal Res 40:242–250
Chesler EJ, Juraska JM (2000) Acute administration of estrogen and progesterone impairs the acquisition of the spatial Morris water maze in ovariectomized rats. Horm Behav 38:234–242
Chu S-F, Zhang Z, Zhang W, Zhang M-J, Gao Y, Han N, Zuo W, Huang H-Y, Chen N-H (2017) Upregulating the expression of survivin-HBXIP complex contributes to the protective role of IMM-H004 in transient global cerebral ischemia/reperfusion. Mol Neurobiol 54:524–540
Cuzzocrea S, Costantino G, Gitto E, Mazzon E, Fulia F, Serraino I, Cordaro S, Barberi I, de Sarro A, Caputi AP (2000) Protective effects of melatonin in ischemic brain injury. J Pineal Res 29:217–227
Espinosa-García C, Aguilar-Hernández A, Cervantes M, Moralí G (2014) Effects of progesterone on neurite growth inhibitors in the hippocampus following global cerebral ischemia. Brain Res 1545:23–34
Gaignard P, Fréchou M, Schumacher M, Thérond P, Mattern C, Slama A, Guennoun R (2016) Progesterone reduces brain mitochondrial dysfunction after transient focal ischemia in male and female mice. J Cereb Blood Flow Metab 36:562–568
Harburger LL, Bennett JC, Frick KM (2007) Effects of estrogen and progesterone on spatial memory consolidation in aged females. Neurobiol Aging 28:602–610
Hassanzadeh P, Arbabi E (2012) The effects of progesterone on glial cell line-derived neurotrophic factor secretion from C6 glioma cells. Iran J Basic Med Sci 15:1046
Imbesi M, Uz T, Manev H (2008) Role of melatonin receptors in the effects of melatonin on BDNF and neuroprotection in mouse cerebellar neurons. J Neural Transm 115:1495–1499
Ishrat T, Sayeed I, Atif F, Hua F, Stein DG (2010) Progesterone and allopregnanolone attenuate blood–brain barrier dysfunction following permanent focal ischemia by regulating the expression of matrix metalloproteinases. Exp Neurol 226:183–190
Ishrat T, Sayeed I, Atif F, Hua F, Stein DG (2012) Progesterone is neuroprotective against ischemic brain injury through its effects on the phosphoinositide 3-kinase/protein kinase B signaling pathway. Neuroscience 210:442–450
Kondoh T, Uneyama H, Nishino H, Torii K (2002) Melatonin reduces cerebral edema formation caused by transient forebrain ischemia in rats. Life Sci 72:583–590
Kühl NM, Hoekstra D, de Vries H, de Keyser J (2003) Insulin-like growth factor-binding protein 6 inhibits survival and differentiation of rat oligodendrocyte precursor cells. Glia 44:91–101
Lee E, Lee MY, Chen HY, Hsu YS, Wu TS, Chen ST, Chang GL (2005) Melatonin attenuates gray and white matter damage in a mouse model of transient focal cerebral ischemia. J Pineal Res 38:42–52
Lee MY, Kuan YH, Chen HY, Chen TY, Chen ST, Huang CC, Yang I, Hsu YS, Wu TS, Lee E (2007) Intravenous administration of melatonin reduces the intracerebral cellular inflammatory response following transient focal cerebral ischemia in rats. J Pineal Res 42:297–309
Letechipía-Vallejo G, López-Loeza E, ESPINOZA-GONZÁLEZ V, González-Burgos I, Olvera-Cortés ME, Moralí G, Cervantes M (2007) Long-term morphological and functional evaluation of the neuroprotective effects of post-ischemic treatment with melatonin in rats. J Pineal Res 42:138–146
Lin H-W, Lee E-J (2009) Effects of melatonin in experimental stroke models in acute, sub-acute, and chronic stages. Neuropsychiatr Dis Treat 5:157–162
Manev H, Uz T, Kharlamov A, Joo J (1996) Increased brain damage after stroke or excitotoxic seizures in melatonin-deficient rats. FASEB J 10:1546–1551
Melchiorri D, Reiter R, Sewerynek E, Chen L, Nistico G (1995) Melatonin reduces kainate-induced lipid peroxidation in homogenates of different brain regions. FASEB J 9:1205–1210
Mori MA, Meyer E, Soares LM, Milani H, Guimarães FS, de Oliveira RMW (2017) Cannabidiol reduces neuroinflammation and promotes neuroplasticity and functional recovery after brain ischemia. Prog Neuro-Psychopharmacol Biol Psychiatry 75:94–105
Movassaghi S, Sharifi ZN, Soleimani M, Joghataii MT, Hashemi M, Shafaroodi H, Mehdizadeh M (2012) Effect of pentoxifylline on ischemia-induced brain damage and spatial memory impairment in rat. Iran J Basic Med Sci 15:1083
Nibuya M, Morinobu S, Duman RS (1995) Regulation of BDNF and trkB mRNA in rat brain by chronic electroconvulsive seizure and antidepressant drug treatments. J Neurosci 15:7539–7547
Reiter RJ (2000) Melatonin: lowering the high price of free radicals. Physiology 15:246–250
Sayeed I, Stein DG (2009) Progesterone as a neuroprotective factor in traumatic and ischemic brain injury. Prog Brain Res 175:219–237
Sharifi Z-N, Abolhassani F, Zarrindast MR, Movassaghi S, Rahimian N, Hassanzadeh G (2011) Effects of FK506 on hippocampal CA1 cells following transient global ischemia/reperfusion in Wistar rat. Stroke Res Treat 2012
Shinozuka K, Staples M, Borlongan CV (2013) Melatonin-based therapeutics for neuroprotection in stroke. Int J Mol Sci 14:8924–8947
Tao R-R, Ji Y-L, Lu Y-M, Fukunaga K, Han F (2012) Targeting nitrosative stress for neurovascular protection: new implications in brain diseases. Curr Drug Targets 13:272–284
Tao RR, Huang JY, Shao XJ, YE WF, Tian Y, Liao MH, Fukunaga K, Lou YJ, Han F, Lu YM (2013) Ischemic injury promotes Keap1 nitration and disturbance of antioxidative responses in endothelial cells: a potential vasoprotective effect of melatonin. J Pineal Res 54:271–281
Tütüncüler F, Eskiocak S, Başaran ÜN, Ekuklu G, Ayvaz S, Vatansever Ü (2005) The protective role of melatonin in experimental hypoxic brain damage. Pediatr Int 47:434–439
Watson N, Diamandis T, Gonzales-Portillo C, Reyes S, Borlongan CV (2016) Melatonin as an antioxidant for stroke neuroprotection. Cell Transplant 25:883–891
Wong R, Gibson CL, Kendall DA, Bath PM (2014) Evaluating the translational potential of progesterone treatment following transient cerebral ischaemia in male mice. BMC Neurosci 15:131
Xu Q, Ji X-F, Chi T-Y, Liu P, Jin G, Gu S-L, Zou L-B (2015) Sigma 1 receptor activation regulates brain-derived neurotrophic factor through NR2A-CaMKIV-TORC1 pathway to rescue the impairment of learning and memory induced by brain ischaemia/reperfusion. Psychopharmacology 232:1779–1791
Yang Y, Jiang S, Dong Y, Fan C, Zhao L, Yang X, Li J, Di S, Yue L, Liang G (2015) Melatonin prevents cell death and mitochondrial dysfunction via a SIRT1-dependent mechanism during ischemic-stroke in mice. J Pineal Res 58:61–70
Yao X-L, Liu J, Lee E, Ling GS, Mccabe JT (2005) Progesterone differentially regulates pro-and anti-apoptotic gene expression in cerebral cortex following traumatic brain injury in rats. J Neurotrauma 22:656–668
Younan N, El-Attar S & Sabry D (2012). Progesterone as a neuroprotective treatment in brain ischemic injury in Ovariectomized rats: relation to brain derived neurotrophic factor, Synaptic Plasticity and Astrocytes The Medical Journal of Cairo University, 80
Yousuf S, Atif F, Sayeed I, Wang J, Stein DG (2013) Post-stroke infections exacerbate ischemic brain injury in middle-aged rats: immunomodulation and neuroprotection by progesterone. Neuroscience 239:92–102
Yousuf S, Sayeed I, Atif F, Tang H, Wang J, Stein DG (2014) Delayed progesterone treatment reduces brain infarction and improves functional outcomes after ischemic stroke: a time-window study in middle-aged rats. J Cereb Blood Flow Metab 34:297–306
Zhang HM, Zhang Y (2014) Melatonin: a well-documented antioxidant with conditional pro-oxidant actions. J Pineal Res 57:131–146
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Hedayatpour, A., Shiasi, M., Famitafreshi, H. et al. Co-Administration of Progesterone and Melatonin Attenuates Ischemia-Induced Hippocampal Damage in Rats. J Mol Neurosci 66, 251–260 (2018). https://doi.org/10.1007/s12031-018-1163-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-018-1163-6