Abstract
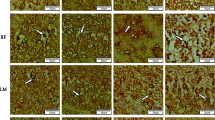
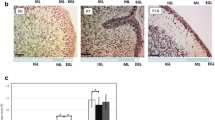
This study aimed to compare the regional distribution of insulin receptor in various portions of newborn rat hippocampus on postnatal days 0 (P0), 7 (P7), and 14 (P14) between male/female and right/left hippocampi. We found that the number of insulin receptor (InsR)-immunoreactive-positive (InsR+) cells in CA1 continued to increase until P7 and remained unchanged thereafter. A marked increase in distribution of InsR+ cells in CA3 from P0 to P14 was observed, although there was a significant decline in the number of InsR+ cells in dentate gyrus (DG) at the same time. No differences between the right/left and male/female hippocampi were detected at P0 (P > 0.05). Seven-day-old female rats showed a higher number of labeled cells in the left than in the right hippocampus. Moreover, the differences between the number of InsR+ cells in area CA1 and CA3 were statistically significant between males and females (P < 0.05). At P14, the number of InsR+ cells was significantly higher in CA1 and DG of males, especially in the right one (P < 0.05). These results indicate the existence of a differential distribution pattern of InsR between the left/right and male/female hippocampi. Together with other mechanisms, these differences may underlie sexual dimorphism and left/right asymmetry in the hippocampus.



Similar content being viewed by others
References
Agrawal R, Tyagi E, Shukla R, Nath C (2011) Insulin receptor signaling in rat hippocampus: a study in STZ (ICV) induced memory deficit model. Eur Neuropsychopharmacol 21(3):261–273. doi:10.1016/j.euroneuro.2010.11.009
Altman J, Das GD (1965) Autoradiographic and histological evidence of postnatal hippocampal neurogenesis in rats. J Comp Neurol 124(3):319–335
Baron-Van Evercooren A, Olichon-Berthe C, Kowalski A, Visciano G, Van Obberghen E (1991) Expression of IGF-I and insulin receptor genes in the rat central nervous system: a developmental, regional, and cellular analysis. J Neurosci Res 28(2):244–253. doi:10.1002/jnr.490280212
Baskin DG, Wilcox BJ, Figlewicz DP, Dorsa DM (1988) Insulin and insulin-like growth factors in the CNS. Trends Neurosci 11(3):107–111
Bayer SA (1980a) Development of the hippocampal region in the rat. I. Neurogenesis examined with 3H-thymidine autoradiography. J Comp Neurol 190(1):87–114. doi:10.1002/cne.901900107
Bayer SA (1980b) Development of the hippocampal region in the rat. II. Morphogenesis during embryonic and early postnatal life. J Comp Neurol 190(1):115–134. doi:10.1002/cne.901900108
Bondy CA, Bach MA, Lee WH (1992) Mapping of brain insulin and insulin-like growth factor receptor gene expression by in situ hybridization. Neuroprotocols 1(3):240–249
Bowers JM, Waddell J, McCarthy MM (2010) A developmental sex difference in hippocampal neurogenesis is mediated by endogenous oestradiol. Biol Sex Differ 1(1):8. doi:10.1186/2042-6410-1-8
Castle DJ, Murray RM (1991) The neurodevelopmental basis of sex differences in schizophrenia. Psychol Med 21(3):565–575
Chiu SL, Cline HT (2010) Insulin receptor signaling in the development of neuronal structure and function. Neural Dev 5:7. doi:10.1186/1749-8104-5-7
Cooke B, Hegstrom CD, Villeneuve LS, Breedlove SM (1998) Sexual differentiation of the vertebrate brain: principles and mechanisms. Front Neuroendocrinol 19(4):323–362. doi:10.1006/frne.1998.0171
de Lacoste MC, Adesanya T, Woodward DJ (1990) Measures of gender differences in the human brain and their relationship to brain weight. Biol Psychiatry 28(11):931–942
de Pablo F, de la Rosa EJ (1995) The developing CNS: a scenario for the action of proinsulin, insulin and insulin-like growth factors. Trends Neurosci 18(3):143–150
DeCarolis NA, Eisch AJ (2010) Hippocampal neurogenesis as a target for the treatment of mental illness: a critical evaluation. Neuropharmacology 58(6):884–893. doi:10.1016/j.neuropharm.2009.12.013
Diamond MC, Murphy GM Jr, Akiyama K, Johnson RE (1982) Morphologic hippocampal asymmetry in male and female rats. Exp Neurol 76(3):553–565
Diamond MC, Johnson RE, Young D, Singh SS (1983) Age-related morphologic differences in the rat cerebral cortex and hippocampus: male–female; right–left. Exp Neurol 81(1):1–13
Dou JT, Chen M, Dufour F, Alkon DL, Zhao WQ (2005) Insulin receptor signaling in long-term memory consolidation following spatial learning. Learn Mem 12(6):646–655. doi:10.1101/lm.88005
Eichenbaum H (1996) Is the rodent hippocampus just for ‘place’? Curr Opin Neurobiol 6(2):187–195
Exner C, Nehrkorn B, Martin V, Huber M, Shiratori K, Rief W (2008) Sex-dependent hippocampal volume reductions in schizophrenia relate to episodic memory deficits. J Neuropsychiatry Clin Neurosci 20(2):227–230. doi:10.1176/appi.neuropsych.20.2.227
Forster E, Zhao S, Frotscher M (2006) Laminating the hippocampus. Nat Rev Neurosci 7(4):259–267. doi:10.1038/nrn1882
Foster DJ, Knierim JJ (2012) Sequence learning and the role of the hippocampus in rodent navigation. Curr Opin Neurobiol 22(2):294–300. doi:10.1016/j.conb.2011.12.005
Frazier JA, Hodge SM, Breeze JL, Giuliano AJ, Terry JE, Moore CM, Kennedy DN, Lopez-Larson MP, Caviness VS, Seidman LJ, Zablotsky B, Makris N (2008) Diagnostic and sex effects on limbic volumes in early-onset bipolar disorder and schizophrenia. Schizophr Bull 34(1):37–46. doi:10.1093/schbul/sbm120
Fuhs MC, Touretzky DS (2007) Context learning in the rodent hippocampus. Neural Comput 19(12):3173–3215. doi:10.1162/neco.2007.19.12.3173
Galea LA (2008) Gonadal hormone modulation of neurogenesis in the dentate gyrus of adult male and female rodents. Brain Res Rev 57(2):332–341. doi:10.1016/j.brainresrev.2007.05.008
Galea LA, Spritzer MD, Barker JM, Pawluski JL (2006) Gonadal hormone modulation of hippocampal neurogenesis in the adult. Hippocampus 16(3):225–232. doi:10.1002/hipo.20154
Giap BT, Jong CN, Ricker JH, Cullen NK, Zafonte RD (2000) The hippocampus: anatomy, pathophysiology, and regenerative capacity. J Head Trauma Rehabil 15(3):875–894
Gilbert PE, Kesner RP (2002) Role of the rodent hippocampus in paired-associate learning involving associations between a stimulus and a spatial location. Behav Neurosci 116(1):63–71
Gundersen HJ, Bagger P, Bendtsen TF, Evans SM, Korbo L, Marcussen N, Møller A, Nielsen K, Nyengaard JR, Pakkenberg B et al (1988) The new stereological tools: disector, fractionator, nucleator and point sampled intercepts and their use in pathological research and diagnosis. APMIS 96(10):857–881
Haj-ali V, Mohaddes G, Babri SH (2009) Intracerebroventricular insulin improves spatial learning and memory in male Wistar rats. Behav Neurosci 123(6):1309–1314. doi:10.1037/a0017722
Hallschmid M, Benedict C, Born J, Kern W (2007) Targeting metabolic and cognitive pathways of the CNS by intranasal insulin administration. Expert Opin Drug Deliv 4(4):319–322. doi:10.1517/17425247.4.4.319
Hami J, Sadr-Nabavi A, Sankian M, Haghir H (2012) Sex differences and left–right asymmetries in expression of insulin and insulin-like growth factor-1 receptors in developing rat hippocampus. Brain Struct Funct 217(2):293–302. doi:10.1007/s00429-011-0358-1
Han X, Ma Y, Liu X, Wang L, Qi S, Zhang Q, Du Y (2012) Changes in insulin-signaling transduction pathway underlie learning/memory deficits in an Alzheimer's disease rat model. J Neural Transm 119(11):1407–1416. doi:10.1007/s00702-012-0803-1
Hayman LA, Fuller GN, Cavazos JE, Pfleger MJ, Meyers CA, Jackson EF (1998) The hippocampus: normal anatomy and pathology. AJR Am J Roentgenol 171(4):1139–1146. doi:10.2214/ajr.171.4.9763010
Heidenreich KA (1991) Insulin in the brain what is its role? Trends Endocrinol Metab 2(1):9–12
Hill JM, Lesniak MA, Pert CB, Roth J (1986) Autoradiographic localization of insulin receptors in rat brain: prominence in olfactory and limbic areas. Neuroscience 17(4):1127–1138
Hine RJ, Das GD (1974) Neuroembryogenesis in the hippocampal formation of the rat. An autoradiographic study. Z Anat Entwicklungsgesch 144(2):173–186
Humphrey T (1967) The development of the human hippocampal fissure. J Anat 101(Pt 4):655–676
Hussain RJ, Carpenter DO (2005) A comparison of the roles of protein kinase C in long-term potentiation in rat hippocampal areas CA1 and CA3. Cell Mol Neurobiol 25(3–4):649–661. doi:10.1007/s10571-005-4045-8
Kappy MS, Raizada MK (1982) Adult-level insulin binding is present in term fetal rat CNS membranes. Brain Res 249(2):390–392
Kappy M, Sellinger S, Raizada M (1984) Insulin binding in four regions of the developing rat brain. J Neurochem 42(1):198–203
Kar S, Chabot JG, Quirion R (1993) Quantitative autoradiographic localization of [125I]insulin-like growth factor I, [125I]insulin-like growth factor II, and [125I]insulin receptor binding sites in developing and adult rat brain. J Comp Neurol 333(3):375–397. doi:10.1002/cne.903330306
Knowles WD (1992) Normal anatomy and neurophysiology of the hippocampal formation. J Clin Neurophysiol 9(2):252–263
Kohl MM, Shipton OA, Deacon RM, Rawlins JN, Deisseroth K, Paulsen O (2011) Hemisphere-specific optogenetic stimulation reveals left–right asymmetry of hippocampal plasticity. Nat Neurosci 14(11):1413–1415. doi:10.1038/nn.2915
Kumazawa-Manita N, Hama H, Miyawaki A, Iriki A (2013) Tool use specific adult neurogenesis and synaptogenesis in rodent (Octodon degus) hippocampus. PLoS One 8(3):e58649. doi:10.1371/journal.pone.0058649
Lancaster FE (1994) Gender differences in the brain: implications for the study of human alcoholism. Alcohol Clin Exp Res 18(3):740–746
Lister JP, Tonkiss J, Blatt GJ, Kemper TL, DeBassio WA, Galler JR, Rosene DL (2006) Asymmetry of neuron numbers in the hippocampal formation of prenatally malnourished and normally nourished rats: a stereological investigation. Hippocampus 16(11):946–958. doi:10.1002/hipo.20221
Marks JL, Porte D Jr, Stahl WL, Baskin DG (1990) Localization of insulin receptor mRNA in rat brain by in situ hybridization. Endocrinology 127(6):3234–3236
Martin SJ, Clark RE (2007) The rodent hippocampus and spatial memory: from synapses to systems. Cell Mol Life Sci 64(4):401–431. doi:10.1007/s00018-007-6336-3
McBain CJ (2008) Differential mechanisms of transmission and plasticity at mossy fiber synapses. Prog Brain Res 169:225–240. doi:10.1016/S0079-6123(07)00013-1
Moosavi M, Naghdi N, Maghsoudi N, Zahedi Asl S (2006) The effect of intrahippocampal insulin microinjection on spatial learning and memory. Horm Behav 50(5):748–752. doi:10.1016/j.yhbeh.2006.06.025
Morris JA, Jordan CL, Breedlove SM (2004) Sexual differentiation of the vertebrate nervous system. Nat Neurosci 7(10):1034–1039. doi:10.1038/nn1325nn1325
Mudd LM, Masters BA, Raizada MK (1988) Insulin and related growth factors: effects on the nervous system and mechanism for neurite growth and regeneration. Understanding brain development and repair of cns injury. Neurochem Int 12(4):415–417
Muramatsu R, Ikegaya Y, Matsuki N, Koyama R (2007) Neonatally born granule cells numerically dominate adult mice dentate gyrus. Neuroscience 148(3):593–598. doi:10.1016/j.neuroscience.2007.06.040
Nakae J, Kido Y, Accili D (2001) Distinct and overlapping functions of insulin and IGF-I receptors. Endocr Rev 22(6):818–835
Nalloor R, Bunting KM, Vazdarjanova A (2012) Encoding of emotion-paired spatial stimuli in the rodent hippocampus. Front Behav Neurosci 6:27. doi:10.3389/fnbeh.2012.00027
Navarro I, Leibush B, Moon TW, Plisetskaya EM, Banos N, Mendez E, Planas JV, Gutierrez J (1999) Insulin, insulin-like growth factor-I (IGF-I) and glucagon: the evolution of their receptors. Comp Biochem Physiol B Biochem Mol Biol 122(2):137–153
Needleman LA, McAllister AK (2008) Seeing the light: insulin receptors and the CNS. Neuron 58(5):653–655. doi:10.1016/j.neuron.2008.06.001
Nelson TJ, Sun MK, Hongpaisan J, Alkon DL (2008) Insulin, PKC signaling pathways and synaptic remodeling during memory storage and neuronal repair. Eur J Pharmacol 585(1):76–87. doi:10.1016/j.ejphar.2008.01.051
Ormerod BK, Lee TT, Galea LA (2004) Estradiol enhances neurogenesis in the dentate gyri of adult male meadow voles by increasing the survival of young granule neurons. Neuroscience 128(3):645–654. doi:10.1016/j.neuroscience.2004.06.039
Ormerod BK, Palmer TD, Caldwell MA (2008) Neurodegeneration and cell replacement. Philos Trans R Soc Lond B Biol Sci 363(1489):153–170. doi:10.1098/rstb.2006.2018
Pawluski JL, Brummelte S, Barha CK, Crozier TM, Galea LA (2009) Effects of steroid hormones on neurogenesis in the hippocampus of the adult female rodent during the estrous cycle, pregnancy, lactation and aging. Front Neuroendocrinol 30(3):343–357. doi:10.1016/j.yfrne.2009.03.007
Paxinos G, Watson C (2006) The rat brain in stereotaxic coordinates. Elsevier, New York
Perfilieva E, Risedal A, Nyberg J, Johansson BB, Eriksson PS (2001) Gender and strain influence on neurogenesis in dentate gyrus of young rats. J Cereb Blood Flow Metab 21(3):211–217. doi:10.1097/00004647-200103000-00004
Plum L, Schubert M, Bruning JC (2005) The role of insulin receptor signaling in the brain. Trends Endocrinol Metab 16(2):59–65. doi:10.1016/j.tem.2005
Ragbetli MC, Aydinlioglu A, Kaplan S (2002) Sex differences and right–left asymmetries in rat hippocampal components. Int J Neurosci 112(1):81–95
Ramachandra R, Thyagarajan S (2011) Atlas of the neonatal rat brain. CRC Press, Bosa Roca
Reagan LP (2007) Insulin signaling effects on memory and mood. Curr Opin Pharmacol 7(6):633–637. doi:10.1016/j.coph.2007.10.012
Roof RL, Havens MD (1992) Testosterone improves maze performance and induces development of a male hippocampus in females. Brain Res 572(1–2):310–313
Schechter R, Whitmire J, Holtzclaw L, George M, Harlow R, Devaskar SU (1992) Developmental regulation of insulin in the mammalian central nervous system. Brain Res 582(1):27–37
Schlessinger AR, Cowan WM, Gottlieb DI (1975) An autoradiographic study of the time of origin and the pattern of granule cell migration in the dentate gyrus of the rat. J Comp Neurol 159(2):149–175. doi:10.1002/cne.901590202
Takahashi R, Ishii K, Kakigi T, Yokoyama K (2011) Gender and age differences in normal adult human brain: voxel-based morphometric study. Hum Brain Mapp 32(7):1050–1058. doi:10.1002/hbm.21088
Thompson CL, Pathak SD, Jeromin A, Ng LL, MacPherson CR, Mortrud MT, Cusick A, Riley ZL, Sunkin SM, Bernard A, Puchalski RB, Gage FH, Jones AR, Bajic VB, Hawrylycz MJ, Lein ES (2008) Genomic anatomy of the hippocampus. Neuron 60(6):1010–1021. doi:10.1016/j.neuron.2008.12.008
Toga AW, Thompson PM (2003) Mapping brain asymmetry. Nat Rev Neurosci 4(1):37–48. doi:10.1038/nrn1009nrn1009
von Wilmsdorff M, Sprick U, Bouvier ML, Schulz D, Schmitt A, Gaebel W (2010) Sex-dependent behavioral effects and morphological changes in the hippocampus after prenatal invasive interventions in rats: implications for animal models of schizophrenia. Clinics (Sao Paulo) 65(2):209–219. doi:10.1590/S1807-59322010000200014
Werther GA, Hogg A, Oldfield BJ, McKinley MJ, Figdor R, Allen AM, Mendelsohn FA (1987) Localization and characterization of insulin receptors in rat brain and pituitary gland using in vitro autoradiography and computerized densitometry. Endocrinology 121(4):1562–1570
Wozniak M, Rydzewski B, Baker SP, Raizada MK (1993) The cellular and physiological actions of insulin in the central nervous system. Neurochem Int 22(1):1–10
Wrighten SA, Piroli GG, Grillo CA, Reagan LP (2009) A look inside the diabetic brain: contributors to diabetes-induced brain aging. Biochim Biophys Acta 1792(5):444–453. doi:10.1016/j.bbadis.2008.10.013
Xiao L, Jordan CL (2002) Sex differences, laterality, and hormonal regulation of androgen receptor immunoreactivity in rat hippocampus. Horm Behav 42(3):327–336
Zalutsky RA, Nicoll RA (1990) Comparison of two forms of long-term potentiation in single hippocampal neurons. Science 248(4963):1619–1624
Zhang JM, Konkle AT, Zup SL, McCarthy MM (2008) Impact of sex and hormones on new cells in the developing rat hippocampus: a novel source of sex dimorphism? Eur J Neurosci 27(4):791–800. doi:10.1111/j.1460-9568.2008.06073.x
Zhao WQ, Alkon DL (2001) Role of insulin and insulin receptor in learning and memory. Mol Cell Endocrinol 177(1–2):125–134
Zhao W, Chen H, Xu H, Moore E, Meiri N, Quon MJ, Alkon DL (1999) Brain insulin receptors and spatial memory. Correlated changes in gene expression, tyrosine phosphorylation, and signaling molecules in the hippocampus of water maze trained rats. J Biol Chem 274(49):34893–34902
Zhao WQ, Chen H, Quon MJ, Alkon DL (2004) Insulin and the insulin receptor in experimental models of learning and memory. Eur J Pharmacol 490(1–3):71–81. doi:10.1016/j.ejphar.2004.02.045
Acknowledgments
This paper is based on the results of the thesis of Javad Hami Ph.D. (no. A-299) which was financially supported by the Mashhad University of Medical Sciences (MUMS) grant (no. 88463).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hami, J., Kheradmand, H. & Haghir, H. Sex Differences and Laterality of Insulin Receptor Distribution in Developing Rat Hippocampus: an Immunohistochemical Study. J Mol Neurosci 54, 100–108 (2014). https://doi.org/10.1007/s12031-014-0255-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-014-0255-1