Abstract
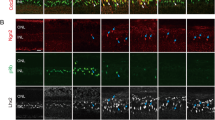
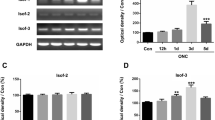
Transcription initiation factor IIB (TFIIB) is an ideal factor to localize core promoters and plays a central role in the assembly of the pre-initiation complex. Previous studies showed that the assembly of TFIIB played an important role in rat ischemic brain injury. To elucidate the expression and possible functions of TFIIB in retina lesion and repair, we performed an optic nerve crush (ONC) model in adult rats. Western blot analysis and immunohistochemistry showed a significant upregulation of TFIIB in retina after ONC. Immunofluorescent labeling indicated that TFIIB was localized mainly in the Müller glia cells (MGCs); colocalization of TFIIB and proliferating cell nuclear antigen (PCNA) in the injured retina suggested that TFIIB might participate in MGCs proliferation. In addition, we also examined the expression of the retinal progenitor markers (Nestin and Pax6) whose changes were correlated with the expression of TFIIB. In vitro, we induced MGCs differentiation with brain nerve growth factor (BNGF) and found that TFIIB expression was increased in the differentiated process, which was collected with the expression of PCNA, Nestin, and Pax6. Additionally, knocking TFIIB down with siRNA inhibited the expression of PCNA, Nestin, and Pax6. Collectively, we hypothesized ONC-induced upregulation of TFIIB in the retina was associated with MGCs activation and differentiation.







Similar content being viewed by others
References
Bagby S, Kim S, Maldonado E, Tong KI, Reinberg D, Ikura M (1995) Solution structure of the C-terminal core domain of human TFIIB: similarity to cyclin A and interaction with TATA-binding protein. Cell 82:857–867
Bernardos RL, Barthel LK, Meyers JR, Raymond PA (2007) Late-stage neuronal progenitors in the retina are radial Muller glia that function as retinal stem cells. J Neurosci 27:7028–7040
Blackshaw S, Harpavat S, Trimarchi J, Cai L, Huang H, Kuo WP, Weber G, Lee K, Fraioli RE, Cho SH, Yung R, Asch E, Ohno-Machado L, Wong WH, Cepko CL (2004) Genomic analysis of mouse retinal development. PLoS Biol 2:E247
Blattner C, Jennebach S, Herzog F, Mayer A, Cheung AC, Witte G, Lorenzen K, Hopfner KP, Heck AJ, Aebersold R, Cramer P (2011) Molecular basis of Rrn3-regulated RNA polymerase I initiation and cell growth. Genes Dev 25:2093–2105
Bodeutsch N, Thanos S (2000) Migration of phagocytotic cells and development of the murine intraretinal microglial network: an in vivo study using fluorescent dyes. Glia 32:91–101
Bringmann A, Pannicke T, Grosche J, Francke M, Wiedemann P, Skatchkov SN, Osborne NN, Reichenbach A (2006) Muller cells in the healthy and diseased retina. Prog Retin Eye Res 25:397–424
Bringmann A, Iandiev I, Pannicke T, Wurm A, Hollborn M, Wiedemann P, Osborne NN, Reichenbach A (2009) Cellular signaling and factors involved in Muller cell gliosis: neuroprotective and detrimental effects. Prog Retin Eye Res 28:423–451
Buratowski S, Zhou H (1993) Functional domains of transcription factor TFIIB. Proc Natl Acad Sci U S A 90:5633–5637
de Melo Reis RA, Ventura AL, Schitine CS, de Mello MC, de Mello FG (2008) Muller glia as an active compartment modulating nervous activity in the vertebrate retina: neurotransmitters and trophic factors. Neurochem Res 33:1466–1474
Fausett BV, Goldman D (2006) A role for alpha1 tubulin-expressing Muller glia in regeneration of the injured zebrafish retina. J Neurosci 26:6303–6313
Franklin CC, McCulloch AV, Kraft AS (1995) In vitro association between the Jun protein family and the general transcription factors, TBP and TFIIB. Biochem J 305(Pt 3):967–974
Friedman MJ, Shah AG, Fang ZH, Ward EG, Warren ST, Li S, Li XJ (2007) Polyglutamine domain modulates the TBP-TFIIB interaction: implications for its normal function and neurodegeneration. Nat Neurosci 10:1519–1528
Garcia-Valenzuela E, Sharma SC, Pina AL (2005) Multilayered retinal microglial response to optic nerve transection in rats. Mol Vis 11:225–231
Hadzic E, Desai-Yajnik V, Helmer E, Guo S, Wu S, Koudinova N, Casanova J, Raaka BM, Samuels HH (1995) A 10-amino-acid sequence in the N-terminal A/B domain of thyroid hormone receptor alpha is essential for transcriptional activation and interaction with the general transcription factor TFIIB. Mol Cell Biol 15:4507–4517
Jadhav AP, Roesch K, Cepko CL (2009) Development and neurogenic potential of Muller glial cells in the vertebrate retina. Prog Retin Eye Res 28:249–262
Jiang G, Zheng L, Pu J, Mei H, Zhao J, Huang K, Zeng F, Tong Q (2012) Small RNAs targeting transcription start site induce heparanase silencing through interference with transcription initiation in human cancer cells. PLoS One 7:e31379
Kassen SC, Ramanan V, Montgomery JE, Burket CT, Liu CG, Vihtelic TS, Hyde DR (2007) Time course analysis of gene expression during light-induced photoreceptor cell death and regeneration in albino zebrafish. Dev Neurobiol 67:1009–1031
Liu Z, Wang D, Shao B, Wu X, Xu J, Lu Q, Wang Y, Li C, Shen A, Wu Q (2011) Increased expression of transcription initiation factor IIB after rat traumatic brain injury. J Mol Histol 42:265–271
Lubiniski W, Palacz O, Karczewicz D (2006) Electrophysiological tests in early and differential diagnosis of some hereditary retinal and optic nerve diseases. Klin Ocz 108:93–98
Luna G, Lewis GP, Banna CD, Skalli O, Fisher SK (2010) Expression profiles of nestin and synemin in reactive astrocytes and Muller cells following retinal injury: a comparison with glial fibrillar acidic protein and vimentin. Mol Vis 16:2511–2523
Nikolov DB, Chen H, Halay ED, Usheva AA, Hisatake K, Lee DK, Roeder RG, Burley SK (1995) Crystal structure of a TFIIB-TBP-TATA-element ternary complex. Nature 377:119–128
Ooto S, Akagi T, Kageyama R, Akita J, Mandai M, Honda Y, Takahashi M (2004) Potential for neural regeneration after neurotoxic injury in the adult mammalian retina. Proc Natl Acad Sci U S A 101:13654–13659
Orphanides G, Lagrange T, Reinberg D (1996) The general transcription factors of RNA polymerase II. Genes Dev 10:2657–2683
Osborne NN, Wood JP, Chidlow G, Bae JH, Melena J, Nash MS (1999) Ganglion cell death in glaucoma: what do we really know? Br J Ophthalmol 83:980–986
Panagis L, Thanos S, Fischer D, Dermon CR (2005) Unilateral optic nerve crush induces bilateral retinal glial cell proliferation. Eur J Neurosci 21:2305–2309
Quigley HA (1993) Open-angle glaucoma. N Engl J Med 328:1097–1106
Ramírez M, Lamas M (2009) NMDA receptor mediates proliferation and CREB phosphorylation in postnatal Müller glia-derived retinal progenitors. Mol Vis 15:713–721
Roberts SG, Ha I, Maldonado E, Reinberg D, Green MR (1993) Interaction between an acidic activator and transcription factor TFIIB is required for transcriptional activation. Nature 363:741–744
Sauer F, Fondell JD, Ohkuma Y, Roeder RG, Jackle H (1995) Control of transcription by Kruppel through interactions with TFIIB and TFIIE beta. Nature 375:162–164
Sun J, Li R (2010) Human negative elongation factor activates transcription and regulates alternative transcription initiation. J Biol Chem 285:6443–6452
Sun H, Taneja R (2000) Stra13 expression is associated with growth arrest and represses transcription through histone deacetylase (HDAC)-dependent and HDAC-independent mechanisms. Proc Natl Acad Sci U S A 97:4058–4063
Tansey WP, Herr W (1997) Selective use of TBP and TFIIB revealed by a TATA-TBP-TFIIB array with altered specificity. Science 275:829–831
Thummel R, Kassen SC, Montgomery JE, Enright JM, Hyde DR (2008) Inhibition of Muller glial cell division blocks regeneration of the light-damaged zebrafish retina. Dev Neurobiol 68:392–408
Wu WH, Hampsey M (1999) An activation-specific role for transcription factor TFIIB in vivo. Proc Natl Acad Sci U S A 96:2764–2769
Yang J, Cao J, Wang Y, Xu J, Zhou Z, Gu X, Liu X, Wen H, Wu H, Cheng C (2012) Transcription initiation factor IIB involves in Schwann cell differentiation after rat sciatic nerve crush. J Mol Neurosci. doi:10.1007/s12031-012-9865-7
Yurco P, Cameron DA (2005) Responses of Muller glia to retinal injury in adult zebrafish. Vis Res 45:991–1002
Zeng Q, Xia XB (2010) Study on the differentiation of retinal ganglion cells from rat Müller cells in vitro. Zhonghua Yan Ke Za Zhi 46(7):615–620
Zhang C, Tso MO (2003) Characterization of activated retinal microglia following optic axotomy. J Neurosci Res 73:840–845
Acknowledgments
The authors would like to thank Dr. Zhifeng Gu for technical assistance. This work was supported by Health Department Research Projects in Jiangsu Province (grant no. Z200807), Science and Technology Projects in Nantong (grant no. S2010020), Graduate Student Innovation of Science and Technology Projects in Jiangsu Province, and Graduate Student Innovation of Science and Technology Projects in Nantong University (grant no. 13025045).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Yue Xu and Chen Chen contributed equally to this work.
Rights and permissions
About this article
Cite this article
Xu, Y., Chen, C., Jin, N. et al. Müller Glia Cells Activation in Rat Retina After Optic Nerve Injury: Spatiotemporal Correlation with Transcription Initiation Factor IIB. J Mol Neurosci 51, 37–46 (2013). https://doi.org/10.1007/s12031-012-9941-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-012-9941-z