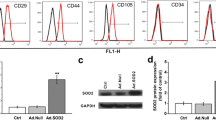
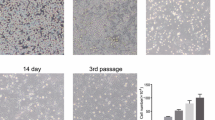
Abstract
Bone marrow stromal cells (MSCs) were used as cell therapy for various diseases in recent years. Some reports showed that transplanted MSCs promote functional recovery in animal models of brain trauma. But other studies indicate that tissue replacement by this method may not be the main source of therapeutic benefit. Neurotrophic factors such as brain-derived neurotrophic factor (BDNF) therapeutic potential may contribute to the recovery of function after trauma. Our previous study showed that BDNF–MSCs could promote the survival of neurons in neuronal injured models in vitro. The present study was undertaken to explore the therapeutic effects of MSCs transfected with BDNF in vivo. After intraventricular injection of MSCs–BDNF, BDNF levels were increased significantly in cerebrospinal fluid by ELISA. Further studies showed that treatment of traumatic brain injury with MSCs–BDNF could attenuate neuronal injury as measurement of biological behavior assessment. These studies demonstrate that by increasing the brain concentration of BDNF, intraventricularly transplanted MSCs–BDNF might play an important role in the treatment of traumatic brain injury and might be an optional therapeutic strategy.



Similar content being viewed by others
References
Assmus B, Schächinger V, Teupe C, Britten M, Lehmann R, Döbert N et al (2002) Transplantation of progenitor cells and regeneration enhancement in acute myocardial infarction. Circulation 106:3009–3017
Beni-Adani L, Gozes I, Cohen Y, Assaf Y, Steingart RA, Brenneman DE, Eizenberg O, Trembolver V, Shohami E. (2001). A peptide derived from activity-dependent neuroprotective protein (ADNP) ameliorates injury response in closed head injury in mice. J Pharmacol Exp Ther. 296.57-63.
Böker W, Yin Z, Drosse I, Haasters F, Rossmann O, Wierer M et al (2008) Introducing a single-cell-derived human mesenchymal stem cell line expressing hTERT after lentiviral gene transfer. J Cell Mol Med 12:1347–1359
Bolton MM, Pittman AJ, Lo DC (2000) Brain-derived neurotrophic factor differentially regulates excitatory and inhibitory synaptic transmission in hippocampal cultures. J Neurosci 20:3221–3232
Borlongan CV, Stahl CE, Cameron DF, Saporta S, Freeman TB, Cahill DW et al (1996) CNS immunological modulation of neural graft rejection and survival. Neurol Res 18:297–304
Brazelton TR, Rossi FM, Keshet GI, Blau HM (2000) From marrow to brain: expression of neuronal phenotypes in adult mice. Science 290:1775–1779
Chen J, Li Y, Wang L, Lu M, Zhang X, Chopp M (2001a) Therapeutic benefit of intracerebral transplantation of bone marrow stromal cells after cerebral ischemia in rats. J Neurol Sci 189:49–57
Chen X, Li Y, Wang L, Katakowski M, Zhang L, Chen J et al (2001b) Ischemic rat brain extracts induce human marrow stromal cell growth factor production. Neuropathology 22:275–279
Chen X, Katakowski M, Li Y, Lu D, Wang L, Zhang L et al (2002) Human bone marrow stromal cell cultures conditioned by traumatic brain tissue extracts: growth factor production. J Neurosci Res 69:687–691
Cheng SL, Yang JW, Rifas L, Zhang SF, Avioli LV (1994) Differentiation of human bone marrow osteogenic stromal cells in vitro: induction of the osteoblast phenotype by dexamethasone. Endocrinology 134:277–286
Chiaretti A, Piastra M, Polidori G (2003) Correlation between neurotrophic factor expression and outcome of children with severe traumatic brain injury. Intensive Care Med 29:1329–1338
Clark BR, Keating A (1995) Biology of bone marrow stroma. Ann N Y Acad Sci 770:70–78
Desai NS, Rutherford LC, Turrigiano GG (1999) Plasticity in the intrinsic excitability of cortical pyramidal neurons. Nat Neurosci 2:515–520
Diógenes MJ, Costenla AR, Lopes LV (2011) Enhancement of LTP in aged rats is dependent on endogenous BDNF. Neuropsychopharmacology 36:1823–1836
Dixon CE, Flinn P, Bao J, Venya R, Hayes RL (1997) Nerve growth factor attenuates cholinergic deficits following traumatic brain injury in rats. Exp Neurol 146:479–490
Fleming JO, Ting JYP, Stohlman SA, Weiner LP (1983) Improvements in obtaining and characterizing mouse cerebrospinal fluid. Application to mouse hepatitis virus-induced encephalomyelitis J Neuroimmunol 4:129–140
Hariri AR, Goldberg TE, Mattay VS (2003) Brain-derived neurotrophic factor val66met polymorphism affects human memory-related hippocampal activity and predicts memory performance. J Neurosci 23:6690–6694
Horwitz EM, Prockop DJ, Fitzpatrick LA, Koo WW, Gordon PL, Neel M et al (1999) Transplantability and therapeutic effects of bone marrow-derived mesenchymal cells in children with osteogenesis imperfecta. Nat Med 5:309–313
Jendelová P, Herynek V, DeCroos J, Glogarová K, Andersson B, Hájek M et al (2003) Imaging the fate of implanted bone marrow stromal cells labeled with superparamagnetic nanoparticles. Magn Reson Med 50:767–776
Kang H, Schuman EM (1996) A requirement for local protein synthesis in neurotrophin-induced hippocampal synaptic plasticity. Science 273:1402–1406
Kaplan GB, Vasterling JJ, Vedak PC (2010) Brain-derived neurotrophic factor in traumatic brain injury, post-traumatic stress disorder, and their comorbid conditions: role in pathogenesis and treatment. Behav Pharmacol 21:427–437
Kim HJ, Lee JH, Kim SH (2010) Therapeutic effects of human mesenchymal stem cells on traumatic brain injury in rats: secretion of neurotrophic factors and inhibition of apoptosis. J Neurotrauma 27:131–138
Koç ON, Gerson SL, Cooper BW, Dyhouse SM, Haynesworth SE, Caplan AI et al (2000) Rapid hematopoietic recovery after coinfusion of autologous-blood stem cells and culture-expanded marrow mesenchymal stem cells in advanced breast cancer patients receiving high-dose chemotherapy. J Clin Oncol 18:307–316
Kocher AA, Schuster MD, Szabolcs MJ, Takuma S, Burkhoff D, Wang J et al (2001) Neovascularization of ischemic myocardium by human bone-marrow-derived angioblasts prevents cardiomyocyte apoptosis, reduces remodeling and improves cardiac function. Nat Med 7:430–436
Li Y, Chopp M, Chen J, Wang L, Gautam SC, Xu YX et al (2000) Intrastriatal transplantation of bone marrow nonhematopoietic cells improves functional recovery after stroke in adult mice. J Cereb Blood Flow Metab 20:1311–1319
Li Y, Chen J, Chen XG, Wang L, Gautam SC, Xu YX et al (2002) Human marrow stromal cell therapy for stroke in rat: neurotrophins and functional recovery. Neurology 59:514–523
Lipsky RH, Marini AM (2007) Brain-derived neurotrophic factor in neuronal survival and behavior-related plasticity. Ann N Y Acad Sci 1122:130–143
Lu D, Li Y, Mahmood A, Wang L, Rafiq T, Chopp M (2002) Neural and marrow-derived stromal cell sphere transplantation in a rat model of traumatic brain injury. J Neurosurg 97:935–940
Mahmood A, Lu D, Wang L, Li Y, Lu M, Chopp M (2001a) Treatment of traumatic brain injury in female rats with intravenous administration of bone marrow stromal cells. Neurosurgery 49:1196–1203
Mahmood A, Lu D, Yi L, Chen JL, Chopp M (2001b) Intracranial bone marrow transplantation after traumatic brain injury improving functional outcome in adult rats. J Neurosurg 94:589–595
Mahmood A, Lu D, Wang L, Chopp M (2002) Intracerebral transplantation of marrow stromal cells cultured with neurotrophic factors promotes functional recovery in adult rats subjected to traumatic brain injury. J Neurotrauma 19:1609–1617
McIntosh TK, Smith DH, Meaney DF, Kotapka MJ, Gennarelli TA, Graham DI (1996) Neuropathological sequelae of traumatic brain injury: relationship to neurochemical and biomechanical mechanisms. Lab Invest 74:315–342
Mezey E, Chandross KJ, Harta G, Maki RA, McKercher SR (2000) Turning blood into brain: cells bearing neuronal antigens generated in vivo from bone marrow. Science 290:1779–1782
Mori T, Kiyono T, Imabayashi H, Takeda Y, Tsuchiya K, Miyoshi S et al (2005) Combination of hTERT and bmi-1, E6, or E7 induces prolongation of the life span of bone marrow stromal cells from an elderly donor without affecting their neurogenic potential. Mol Cell Biol 25:5183–5195
Mu JS, Li WP, Yao ZB, Zhou XF (1999) Deprivation of endogenous brain-derived neurotrophic factor results in impairment of spatial learning and memory in adult rats. Brain Res 835:259–265
Philips MF, Mattiasson G, Wieloch T, Bjorklund A, Johansson BB, Tomasevic G et al (2001) Neuroprotective and behavioral efficacy of nerve growth factor-transfected hippocampal progenitor cell transplants after experimental traumatic brain injury. J Neurosurgs 94:765–774
Sinson G, Perri BR, Trojanowski JQ, Flamm ES, McIntosh TK (1997) Improvement of cognitive deficits and decreased cholinergic neuronal cell loss and apoptotic cell death following neurotrophin infusion after experimental traumatic brain injury. J Neurosurg 86:511–518
Terada N, Hamazaki T, Oka M, Hoki M, Mastalerz DM, Nakano Y, Meyer EM, Morel L, Petersen BE, Scott EW (2002) Bone marrow cells adopt the phenotype of other cells by spontaneous cell fusion. Nature 416:542–545
Thoenen H (1995) Neurotrophins and neuronal plasticity. Science 270:593–598
Wang Z, Deng Q, Zhang X et al (2009) Treatment of injured neurons with bone marrow stem cells cotransfected by hTERT and Ad-BDNF in vitro. J Mol Neurosci 38:265–272
Williams LR, Varon S, Peterson GM, Wictorin K, Fischer W, Bjorklund A et al (1986) Continuous infusion of nerve growth factor prevents basal forebrain neuronal death after fimbria fornix transection. Proc Natl Acad Sci USA 83:9231–9235
Yang K, Perez-Polo JR, Mu XS (1996) Increased expression of brain-derived neurotrophic factor but not neurotrophin-3 mRNA in rat brain after cortical impact injury. J Neurosci Res 44:157–164
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, Z., Yao, W., Deng, Q. et al. Protective Effects of BDNF Overexpression Bone Marrow Stromal Cell Transplantation in Rat Models of Traumatic Brain Injury. J Mol Neurosci 49, 409–416 (2013). https://doi.org/10.1007/s12031-012-9908-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-012-9908-0