Abstract
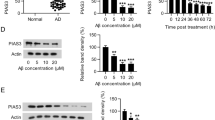
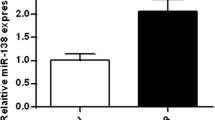
Alzheimer’s disease (AD), the most common form of senile dementia, is associated with neurodegeneration. The development of Alzheimer’s disease is related to abnormalities of cell cycle regulation. Preliminary work showed that a novel gene, CAC1, was highly expressed in tumors and had an oncogene-like function related to cell cycle regulation. The pathogenesis of AD is still incompletely understood. In this study, we measured the expression level of CAC1 in the hippocampus of AD patients to explore the involvement of CAC1 in the development of AD. Our findings showed that the expression level of CAC1 in the hippocampus of AD patients was significantly lower than that of normal controls. The reduction of CAC1 expression did not affect tau/p-tau-396, amyloid precursor protein or apolipoprotein E4 in the in vitro model. A reduction of cyclin E was detected after a CAC1-knockdown. Interestingly, we found that the knockdown of CAC1 by RNAi led to an increase in oxidative stress and the level of p53 protein in SHSY-5Y cells. The expression of CAC1 in SHSY-5Y cells protected the cells from apoptosis induced by Aβ toxicity or oxidative stress. These results established that CAC1 is an important factor for the protection of cells against Aβ toxicity and oxidative stress.






Similar content being viewed by others
References
Arendt T (2002) Dysregulation of neuronal differentiation and cell cycle control in Alzheimer's disease. J Neural Transm Suppl 62:77–85
Arendt T, Bruckner MK (2007) Linking cell-cycle dysfunction in Alzheimer's disease to a failure of synaptic plasticity. Biochim Biophys Acta 1772(4):413–421
Ashford JW, Mortimer JA (2002) Non-familial Alzheimer's disease is mainly due to genetic factors. J Alzheimers Dis 4(3):169–177
Behl C, Davis JB, Lesley R, Schubert D (1994) Hydrogen peroxide mediates amyloid beta protein toxicity. Cell 77:817–827
Bertram L (2009) Alzheimer's disease genetics current status and future perspectives. Int Rev Neurobiol 84:167–184
Bertram L, Tanzi RE (2004) Alzheimer's disease: one disorder, too many genes? Hum Mol Genet 13(Spec No 1):R135–R141
Bettens K, Sleegers K, Van Broeckhoven C (2010) Current status on Alzheimer disease molecular genetics: from past, to present, to future. Hum Mol Genet 19(R1):R4–R11
Bonda DJ, Evans TA, Santocanale C, Llosá JC, Viña J, Bajic VP, Castellani RJ, Siedlak SL, Perry G, Smith MA, Lee HG (2009) Evidence for the progression through S-phase in the ectopic cell cycle re-entry of neurons in Alzheimer disease. Aging 1(4):382–388
Bonda DJ, Bajić VP, Spremo-Potparevic B, Casadesus G, Zhu X, Smith MA, Lee HG (2010) Cell cycle aberrations and neurodegeneration. Neuropathol Appl Neurobiol 36(2):157–163
Bowser R, Smith MA (2002) Cell cycle proteins in Alzheimer's disease: plenty of wheels but no cycle. J Alzheimers Dis 4(3):249–254
Browne SE, Ferrante RJ, Beal MF (1999) Oxidative stress in Huntington's disease. Brain Pathol 9:147–163
Caricasole A, Copani A, Caruso A, Caraci F, Iacovelli L, Sortino MA, Terstappen GC, Nicoletti F (2003) The Wnt pathway, cell-cycle activation and beta-amyloid: novel therapeutic strategies in Alzheimer's disease? Trends Pharmacol Sci 24(5):233–238
Chen Y, Zhang YZ, Zhou ZG, Wang G, Yi ZN (2006) Identification of differently expressed genes in human colorectal adenocarcinoma. World J Gastroenterol 12(7):1025–1032
Copani A, Caraci F, Hoozemans JJ, Calafiore M, Sortino MA, Nicoletti F (2007) The nature of the cell cycle in neurons: focus on a "non-canonical" pathway of DNA replication causally related to death. Biochim Biophys Acta 1772(4):409–412
Crews L, Masliah E (2010) Molecular mechanisms of neurodegeneration in Alzheimer's disease. Hum Mol Genet 19(R1):R12–R20
Dubois B, Feldman HH, Jacova C, Dekosky ST, Barberger-Gateau P, Cummings J, Delacourte A, Galasko D, Gauthier S, Jicha G, Meguro K, O'brien J, Pasquier F, Robert P, Rossor M, Salloway S, Stern Y, Visser PJ, Scheltens P (2007) Research criteria for the diagnosis of Alzheimer's disease: revising the NINCDS-ADRDA criteria. Lancet Neurol 6(8):734–746
Erol A (2010) Are paradoxical cell cycle activities in neurons and glia related to the metabolic theory of Alzheimer's disease? J Alzheimers Dis 19(1):129–135
Harris PL, Zhu X, Pamies C, Rottkamp CA, Ghanbari HA, McShea A, Feng Y, Ferris DK, Smith MA (2000) Neuronal polo-like kinase in Alzheimer disease indicates cell cycle changes. Neurobiol Aging 21(6):837–841
Herrup K, Yang Y (2001) Pictures in molecular medicine: contemplating Alzheimer's disease as cancer: a loss of cell-cycle control. Trends Mol Med 7(11):527
Hornsby PJ (2007) Senescence as an anticancer mechanism. J Clin Oncol 25(14):1852–1857
Huang X, Atwood CS, Hartshorn MA, Multhaup G, Goldstein LE, Scarpa RC, Cuajungco MP, Gray DN, Lim J, Moir RD, Tanzi RE, Bush AI (1999) The A beta peptide of Alzheimer's disease directly produces hydrogen peroxide through metal ion reduction. Biochemistry 38:7609–7616
Kamboh MI (2004) Molecular genetics of late-onset Alzheimer’s disease. Ann Hum Genet 68:381–404
Kong Y, Nan K, Yin Y (2009) Identification and characterization of CAC1 as a novel CDK2-associated cullin. Cell Cycle 8(21):3544–3553
Lendon C, Craddock N (2001) Susceptibility gene(s) for Alzheimer’s disease on chromosome 10. Trends Neurosci 24:557–559
McShea A, Wahl AF, Smith MA (1999) Re-entry into the cell cycle: a mechanism for neurodegeneration in Alzheimer disease. Med Hypotheses 52(6):525–527
Sultana R, Perluigi M, Butterfield DA (2009) Oxidatively modified proteins in Alzheimer's disease (AD), mild cognitive impairment and animal models of AD: role of Abeta in pathogenesis. Acta Neuropathol 118(1):131–150
Waring SC, Rosenberg RN (2008) Genome-wide association studies in Alzheimer disease. Arch Neurol 65(3):329–334
Acknowledgments
We appreciate the work of American Journal Experts (AJE) in the correction of the English text. This work was supported by funds from the National Natural Science Foundation of China (81000865), the science and technology projects of Xi'an city (SF09024-5), and the science and technology projects of Shaanxi Province (2009K12-02). We thank Prof. Yonglie Chu of Xi'an Jiaotong University for proofreading, Prof. Yuxin Yin of Columbia University Medical Center for technical assistance and the members of the Department of Oncology, First Hospital of Xi'an Jiaotong University for advice and suggestions.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kong, Y., Bai, Ps., Sun, H. et al. Expression of the Newly Identified Gene CAC1 in the Hippocampus of Alzheimer’s Disease Patients. J Mol Neurosci 47, 207–218 (2012). https://doi.org/10.1007/s12031-012-9717-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-012-9717-5