Abstract
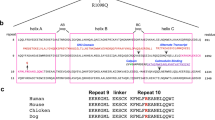
We use 1,2-diacetylbenzene (1,2-DAB) to probe molecular mechanisms of proximal giant neurofilamentous axonopathy (PGNA), a pathological hallmark of amyotrophic lateral sclerosis. The spinal cord proteome of rodents displaying 1,2-DAB PGNA suggests a reduction in the abundance of α-II spectrin (Spna2), a key protein in the maintenance of axonal integrity. Protein immunoblotting indicates that this reduction is due to Spna2 degradation. We investigated the importance of such degradation in 1,2-DAB PGNA. Spna2 mutant mice lacking a calpain- and/or caspase-sensitive domain (CSD), thus hypothetically resistant to 1,2-DAB, and wild-type littermates, were treated with 1,2-DAB, 35 mg/kg/day, or saline control, for 3 weeks. 1,2-DAB induced motor weakness and PGNA, irrespective of the genotype. Spna2–calpain breakdown products were not detected in mutant mice, which displayed a normal structure of the nervous system under saline treatment. Intriguingly, treatment with 1,2-DAB reduced the abundance of the caspase-specific 120-kDa Spna2 breakdown products. Our findings indicate that degradation of Spna2 by calpain- and/or caspase is not central to the pathogenesis of 1,2-DAB axonopathy. In addition, the Spna2-CSD seems to be not required for the maintenance of the cytoskeleton integrity. Our conceptual framework offers opportunities to study the role of calpain–caspase cross talk, including that of the protease degradomics, in models of axonal degeneration.





Similar content being viewed by others
References
Al-Chalabi A, Miller CC (2003) Neurofilaments and neurological disease. Bioessays 25:346–355
Banay-Schwartz M, Kenessey A, DeGuzman T, Lajtha A, Palkovits M (1992) Protein content of various regions of rat brain and adult and aging human brain. Age 15:51–54
Czogalla A, Sikorksi AF (2005) Spectrin and calpain: a ‘target’ and a ‘sniper’ in the pathology of neuronal cells. Cell Mol Life Sci 62:1913–1924
DeCaprio AP (1987) n-Hexane neurotoxicity: a mechanism involving pyrrole adduct formation in axonal cytoskeletal protein. Neurotoxicology 8:199–210
DeCaprio AP, Jackowski SJ, Regan KA (1987) Mechanism of formation and quantitation of imines, pyrroles, and stable nonpyrrole adducts in 2,5-hexanedione-treated protein. Mol Pharmacol 32:542–548
Delisle MB, Carpenter S (1984) Neurofibrillary axonal swellings and amyotrophic lateral sclerosis. J Neurol Sci 63:241–250
Genter St Clair MB, Amarnath V, Moody MA, Anthony DC, Anderson CW, Graham DG (1988) Pyrrole oxidation and protein cross-linking as necessary steps in the development of gamma-diketone neuropathy. Chem Res Toxicol 1:179–185
Hirano A, Donnenfeld H, Sasaki S, Nakano I (1984) Fine structural observations of neurofilamentous changes in amyotrophic lateral sclerosis. J Neuropathol Exp Neurol 43:461–470
Indraswari F, Wong PT, Yap E, Ng YK, Dheen ST (2009) Upregulation of Dpysl2 and Spna2 gene expression in the rat brain after ischemic stroke. Neurochem Int 55(4):235–242
Kim MS, Sabri MI, Miller VH, Kayton RJ, Dixon DA, Spencer PS (2001) 1,2-diacetylbenzene, the neurotoxic metabolite of a chromogenic aromatic solvent, induces proximal axonopathy. Toxicol Appl Pharmacol 177:121–131
Liu MC, Akle V, Zheng W, Dave JR, Tortella FC, Hayes RL, Wang KK (2006) Comparing calpain- and caspase-3-mediated degradation patterns in traumatic brain injury by differential proteome analysis. Biochem J 394:715–725
LoPachin RM, DeCaprio AP (2005) Protein adduct formation as a molecular mechanism of neurotoxicity. Toxicol Sci 86:214–225
Lynch G, Baudry M (1987) Brain spectrin, calpain and long-term changes in synaptic efficacy. Brain Res Bull 18(6):809–815
Meary F, Metral S, Ferreira C, Eladari D, Colin Y, Lecomte MC, Nicolas G (2007) A mutant αII-spectrin designed to resist calpain and caspase cleavage questions the functional importance of this process in vivo. J Biol Chem 282:14226–14237
Nath R, Raser KJ, Stafford D, Hajimohammadreza I, Posner A, Allen H, Talanian RV, Yuen P, Gilbertsen RB, Wang KKW (1996) Non-erythroid α-spectrin breakdown by calpain and interleukin 1β-converting-enzyme-like protease(s) in apoptotic cells: contributory roles of both protease families in neuronal apoptosis. Biochem J 319:683–690
Neumar RW, Xu YA, Gada H, Guttmann RP, Siman R (2002) Cross-talk between calpain and caspase proteolytic systems during neuronal apoptosis. J Biol Chem 278(16):14162–14167
Okamoto K, Hirai S, Shoji M, Senoh Y, Yamazaki T (1990) Axonal swellings in the corticospinal tracts in amyotrophic lateral sclerosis. Acta Neuropathol 80:222–226
Overall CM, Tam EM, Kappelhoff R, Connor A, Ewart T, Morrison CJ, Puente X, López-Otín C, Seth A (2004) Protease degradomics: mass spectrometry discovery of protease substrates and the CLIP-CHIP, a dedicated DNA microarray of all human proteases and inhibitors. Biol Chem 385(6):493–504
Pike BR, Flint J, Dutta S, Johnson E, Wang KKW, Hayes RL (2001) Accumulation of non-erythroid αII-spectrin breakdown products in cerebrospinal fluid after traumatic brain injury in rats. J Neurochem 78:1297–1306
Sikorski AF, Sangerman J, Goodman SR, Critz SD (2000) Spectrin (betaSpIIsigma1) is an essential component of synaptic transmission. Brain Res 852:161–166
Siman R, McIntosh TK, Soltesz KM, Chen Z, Neumar RW, Roberts VL (2004) Proteins released from degenerating neurons are surrogate markers for acute brain damage. Neurobiol Dis 16(2):311–320
Simonovic M, Zhang Z, Cianci CD, Steitz TA, Morrow JS (2006) Structure of the calmodulin αII-spectrin complex provides insight into the regulation of cell plasticity. J Biol Chem 281:34333–34340
Spencer PS, Kim MS, Sabri MI (2002) Aromatic as well as aliphatic hydrocarbon solvent axonopathy. Int J Hyg Environ Health 205:131–136
Susuki K, Rasband MN (2008) Spectrin and ankyrin-based cytoskeletons at polarized domains in myelinated axons. Exp Biol Med (Maywood) 233:394–400
Tshala-Katumbay DD, Palmer VS, Kayton RJ, Sabri MI, Spencer PS (2005) A new murine model of giant proximal axonopathy. Acta Neuropathol 109:405–410
Tshala-Katumbay D, Monterroso V, Kayton R, Lasarev M, Sabri M, Spencer P (2008) Probing mechanisms of axonopathy. Part I: protein targets of 1,2-diacetylbenzene, the neurotoxic metabolite of aromatic solvent 1,2-diethylbenzene. Toxicol Sci 105:134–141
Tshala-Katumbay D, Monterroso V, Kayton R, Lasarev M, Sabri M, Spencer P (2009) Probing mechanisms of axonopathy. Part II: protein targets of 2,5-hexanedione, the neurotoxic metabolite of the aliphatic solvent n-hexane. Toxicol Sci 107:482–489
Wang KKW (2000) Calpain and caspase: can you tell the difference? Trends Neurosci 23:20–26
Warren MW, Kobeissy FH, Liu MC, Hayes RL, Gold MS, Wang KKW (2005) Concurrent calpain and caspase-3 mediated proteolysis of αII-spectrin and tau in rat brain after methamphetamine exposure: a similar profile to traumatic brain injury. Life Sci 78:301–309
Warren MW, Zheng W, Kobeissy FH, Liu MC, Hayes RL, Gold MS, Larner SF, Wang KKW (2007) Calpain- and caspase-mediated αII-spectrin and tau proteolysis in rat cerebrocortical neuronal cultures after ecstasy or methamphetamine exposure. Int J Neuropsychopharmacol 10:479–489
Zhang SX, Bondada V, Geddes JW (2003) Evaluation of conditions for calpain inhibition in the rat spinal cord: effective postinjury inhibition with intraspinal MDL28170 microinjection. J Neurotrauma 20(1):59–67
Zhao X, Pike BR, Newcomb JK, Wang KKW, Posmantur RM, Hayes RL (1999) Maitotoxin induces calpain but not caspase-3 activation and necrotic cell death in primary septo-hippocampal cultures. Neurochem Res 24:371–382
Acknowledgments
We thank Nicole Desmarais and Eli Magnum, OHSU, for their technical assistance. These studies were supported by the NS052183 grant from the NIH-National Institutes of Neurological Disorders and Stroke, Bethesda, MD, USA.
Author information
Authors and Affiliations
Corresponding author
Additional information
Kassa R. and Monterroso V. have equally contributed to the study.
Rights and permissions
About this article
Cite this article
Kassa, R., Monterroso, V., Wentzell, J. et al. Proximal Giant Neurofilamentous Axonopathy in Mice Genetically Engineered to Resist Calpain and Caspase Cleavage of α-II Spectrin. J Mol Neurosci 47, 631–638 (2012). https://doi.org/10.1007/s12031-011-9699-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-011-9699-8